Y10 Chemistry MOCKS
1/76
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
77 Terms
In covalent bonding, electrons are _______
Shared
In ionic bonding, electrons are _______
Lost/Gain
Ionic = A ______ and a _______ reacting
Metal, Non-Metal
Ionic compounds are held together by forces of _______
Electrostatic attraction
Metal = _ Charge
+
Non-metal = _ Charge
-
What are ionic compounds made up of?
A giant structure of ions arranged in a lattice
Giant ionic structures are held by electrostatic attraction meaning it takes ____________
A lot of energy to overcome
Solid ionic compounds can/cannot conduct electricity → Ions aren’t/are fixed
Cannot → Ions are fixed
All ionic compounds will dissolve in water - True or False?
False → Many can but not all
Covalent = ___ _________ reacting
2+ non-metals
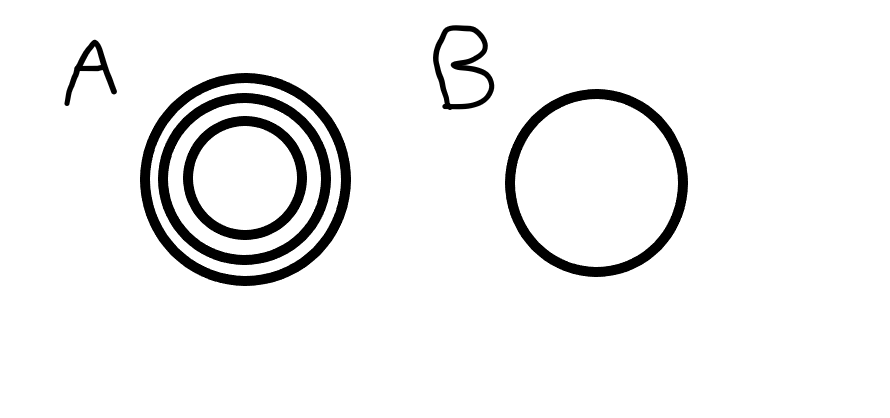
Which diagram is an example of covalent bonding?
B → Only shows outer shell
What is a giant covalent structure?
A compound made of a large network of covalent bonds
Diamond is a giant covalent structure, it is made of carbon with each atoning having _ bonds with other carbons
4
What holds simple molecule substances together?
Weak intermolecular forces → Low melting + boiling point
What holds individual molecules together?
Strong covalent bonds
What are polymers?
Very large molecules with very strong intermolecular forces
Compounds made of simple molecules cannot/can conduct electricity
Cannot → Covalent bonds stop this + no delocalised electrons
Giant covalent structures have strong bonds, this means it is _______ in water and ____ conduct electricity
Insoluble, can’t
Examples of giant covalent structures:
Diamond → Made of _____, each _____ is bonded to another _
Graphite → Made of _____, each _____ is bonded to another _, _____ conduct electricity
Carbon, Carbon, 4
Carbon, Carbon, 3, Can
All (except for graphite) giant covalent structures don’t have delocalised electrons - True or False
True → Can’t conduct, each carbon in graphite has 1 unbonded electron
Graphite will form giant layers of _____
Hexagons
Graphite layers are held together by ________
Intermolecular Forces
What is an allotrope?
Different arrangement of the same elements e.g. Diamond and graphite → Carbon
What is a fullerene?
Hollow shaped molecule of carbon. Based on hexagonal rings (6 carbons) but can be Pentagonal (5) or Heptagonal (7)
In a fullerene, each carbon is bonded to another _ and ___ carry a charge
3, can
What is a nanotube?
A very thin tube with a much greater length than diameter
What are some properties of a nanotube?
High tensile strength → Can withstand a lot
High electrical and thermal conductivity
What is graphene?
Single layer of graphite that is a very good conductor. Low density and the most reactive form of carbon.
Metallic = 2 ______ reacting
Metals
Metal cations are arranged in a _______. Outershell electrons of each metal atom are in a _________________.
Regularly arranged lattice. Sea of delocalised electrons from outershells.
What is the regular structure in metallic bonding called?
Giant metallic lattice
Metal cations are ___________ the electrons → _________ holds lattice tight and makes it strong
Attracted to, electrostatic attraction
More delocalised electrons = More ________ + ______
Electrostatic Attraction, stronger
Conductivity depends on the number of ____________
Delocalised electrons donated → More electrons able to carry the charge
Pure metals have layers that can easily _____________ meaning they’re soft and can be easily _____
Slide over each other, bent
When atoms are added to metals, it makes the layers ______
Distorted
In metals, there is _________________ attraction between cations and _________ electrons
Strong electrostatic, delocalised
Metal + Water → Metal ___________ + ________
Hydroxide, Hydrogen
Metal + Acid → _____ + _________
Salt, Hydrogen
Name the reactivity series using “Please Stop Calling Me A Careless Zebra Instead Try Learning How Copper Saves Gold”
Potassium, Sodium, Calcium, Magnesium, Aluminium, Carbon, Zinc, Iron, Tin, Lead, Hydrogen, Copper, Silver, Gold
What’s an observation of iron in acid?
Little effervescence/bubbling
What’s an observation of copper in acid?
No reaction
What’s an observation of magnesium in acid?
Got warmer and foamed/bubbled
When can a solution carry a charge?
When aqueous or molten
In a displacement reaction, which metal displaces the other?
More reactive displaces the less reactive one
When a metal reacts with acid, the _______ in the acid is displaced. Metal must be ____ reactive than _______.
Hydrogen, more, hydrogen
To separate metals from their oxides, _____ must be used. The metal must be _____ reactive than it.
Carbon, less
Oxidation is ______. Reduction is _______
Loss (of electrons). Gain (of electrons).
Metals are found as ______ with another element.
Ores
To collect pure metal, you must _______ the oxide → Method depends on ________
Reduce, reactivity
For metals more reactive than carbon, you must use _________
Electrolysis
To do electrolysis, the compound must be ______ or ________
Molten, aqueous
Complete this (Acid name → Salt name/ending):
Hydrochloric → ___________
__________ → Sulphate
Nitric → _________
Chloride, sulphuric, nitrate
Neutralisation - Acid + _____ → ____ + Water
Base, salt
Write the steps when making a salt (ABCDEEF)
Acid → Gently Heat
Base → Combine with acid
Crystallisation and Cool
Dab dry → Filter paper
Excess → Solid base seen at the bottom = all acid has reacted
Evaporate → Gently over water bath
Filter → Funnel + Filter paper
A concentrated acid has ____ solute particles in a given volume than a dilute
More
In a strong acid it _____ ionises meaning ______ molecules will dissociate
Fully, all
In a weak acid it _____ ionises meaning _____ molecules will dissociate
Partially, some
What do electrodes do?
Carry electricity into a solution to pull it apart
What charge is the anode?
Positive
What charge is the cathode?
Negative
What charge is the anion?
Negative
What charge is the cation?
Positive
What 2 elements are electrodes mostly made from?
Graphite or platinum
What is electroylsis?
Splitting a substance using an electrical current
What are the charges of OH and NO3 ?
-1
What are the charges of SO4 and CO3 ?
-2
At the anode, what is the rule?
If a halide (Group 7), it will form the halide, otherwise, OH- will form water + oxygen
At the cathode, what is the rule?
Least reactive forms
What are some uses of aluminium?
Pans, drink cans, aeroplanes
How is bauxite collected?
Collected by open cast mining
What does bauxite contain and is purified to?
Aluminium oxide (Al2 O3)
What is aluminium oxide’s melting point?
High → 20500C
Aluminium Oxide’s melting point can be lowered when mixed with molten _______
Cryolite
Electrodes are made of hot _____ which react with oxygen to form _____ _______
Carbon, carbon dioxide
Anodes get ___ ____ meaning they need to get ______ regularly
Worn away, replaced