Honors Chapter 3: Matter and Energy
5.0(1)
Studied by 6 peopleCard Sorting
1/34
Earn XP
Description and Tags
Last updated 2:08 AM on 10/31/22
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
35 Terms
1
New cards
matter
anything that has volume and mass
2
New cards
element
A substance that cannot be broken down into simpler substances
3
New cards
nucleus
Center of an atom made of protons and neutrons
4
New cards
proton
A subatomic particle that has a positive charge and that is found in the nucleus of an atom.
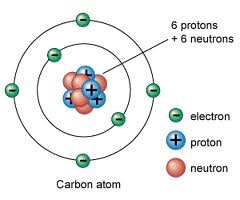
5
New cards
neutron
A subatomic particle that has no charge and that is found in the nucleus of an atom
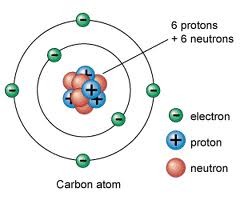
6
New cards
Electron
A subatomic particle that has a negative charge that surround a nucleus. It has almost no mass
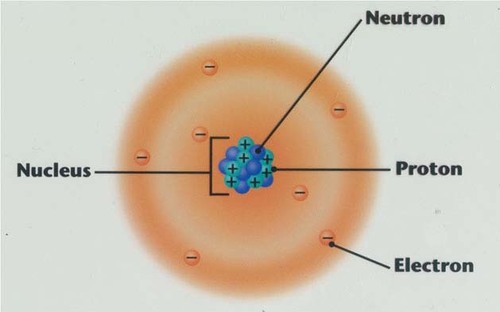
7
New cards
atomic number
the number of protons in the nucleus of an atom
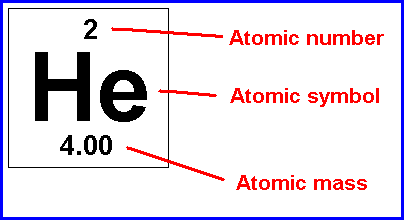
8
New cards
mass number
the sum of the number of neutrons and protons in an atomic nucleus
9
New cards
isotope
Atoms of the same element that have different numbers of neutrons
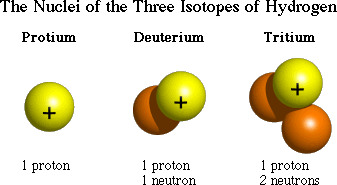
10
New cards
ion
An atom or group of atoms that has a positive or negative charge. Positive are called cations, negative are anions.
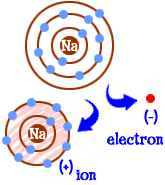
11
New cards
valence electrons
electrons in the outermost shell
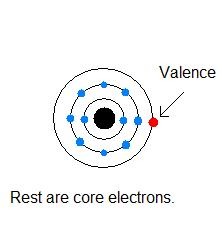
12
New cards
hydrogen and helium
The 2 most ABUNDANT ELEMENTS in the chemical composition of the universe are:
13
New cards
oxygen and silicon
two most abundant elements in the earth's crust
14
New cards
Compound
A substance made up of atoms of two or more different elements joined by chemical bonds that changes the properties of each
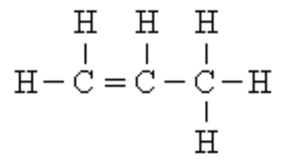
15
New cards
chemical bond
the attractive force that holds atoms or ions together
16
New cards
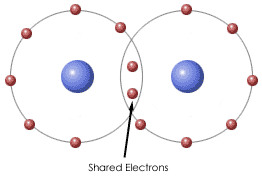
covalent bond
A chemical bond that involves sharing a pair of electrons between atoms in a molecule
17
New cards
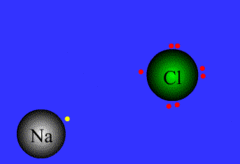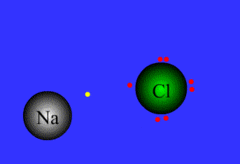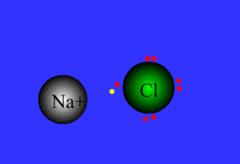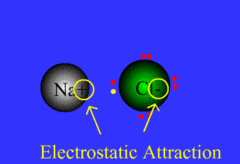
ionic bond
Formed when one or more electrons are transferred from one atom to another
18
New cards
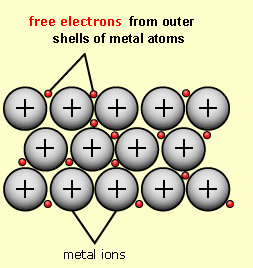
metallic bond
a bond formed by the attraction between positively charged metal ions and negative electrons shared between the atoms
19
New cards
Molecule
two or more atoms held together by covalent bonds
20
New cards
chemical reaction
process that changes one set of chemicals into another set of chemicals
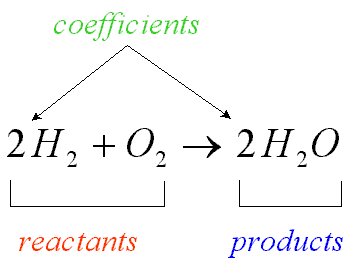
21
New cards
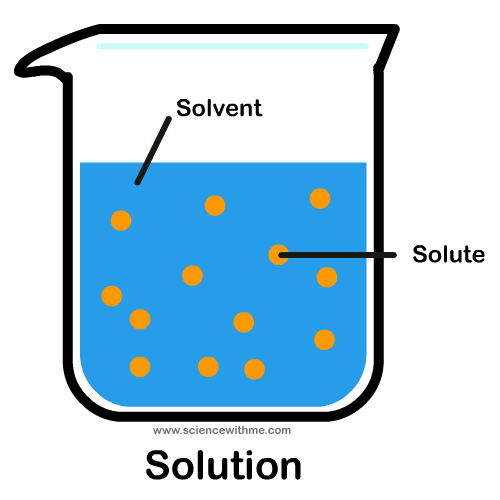
solution
A homogeneous mixture of two or more substances
22
New cards
Mixture
material composed of two or more elements or compounds that are physically mixed together but not chemically combined

23
New cards
Acid
compound that forms hydrogen ions (H+) in solution
24
New cards
base
a compound that produces hydroxide ions (HO-) in solution
25
New cards
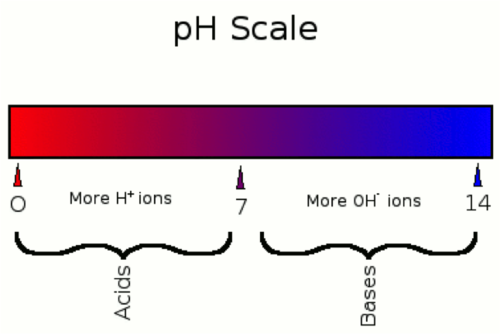
pH scale
scale with values from 0 to 14, used to measure the concentration of H+ ions in a solution; a pH of 0 to 7 is acidic, a pH of 7 is neutral, and a pH of 7 to 14 is basic
26
New cards
Plasma
hot, highly ionized, electrically conducting gas

27
New cards
Evaporation
The change of a substance from a liquid to a gas by an increase in thermal energy

28
New cards
Condensation
The change of state from a gas to a liquid by a release of thermal energy

29
New cards
Sublimation
A change directly from the solid to the gaseous state slowly without becoming liquid

30
New cards
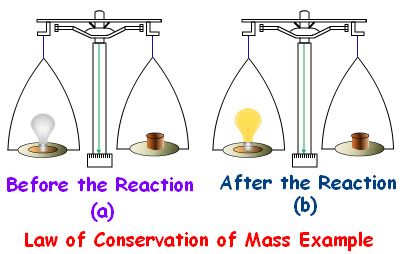
Law of Conservation of Energy
Energy cannot be created or destroyed
31
New cards
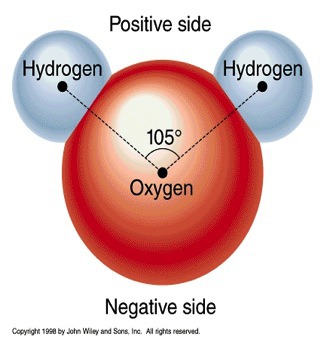
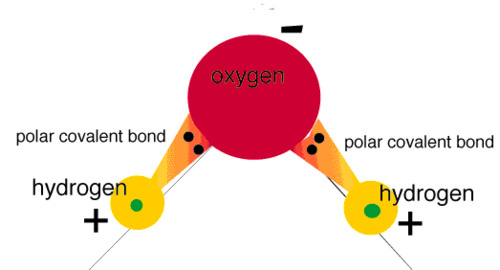
polar molecule
A neutral molecule that has electrically charged areas.
32
New cards
endothermic reaction
A reaction that ABSORBS energy in the form of heat

33
New cards
exothermic reaction
A reaction that releases energy in the form of heat

34
New cards
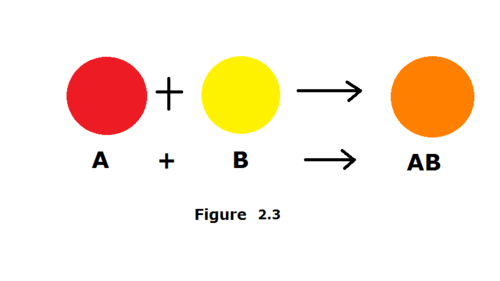
synthesis reaction
a reaction in which two or more substances combine to form a new compound
35
New cards
decomposition reaction
a reaction in which a single compound breaks down to form two or more simpler substances