Diels-Alder reaction
1/11
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
12 Terms
What is the purpose?
Purpose is to provide a diels-alder cycloaddition rxn.
→An e- rich diene reacts with an e- poor alkene to from a cyclohexene ring
Explain the reaction
-The reaction is between a diene with an electron-
deficient alkene (dienophile) that produces a cyclohexene ring
-Known as a [4+2] cycloaddition bc a ring is formed by the interaction of 4 π e- of the alkene with 2 π e- of the alkene/alkyne
What are the stereochemical requirements?
-Diene must be in s-cis conformation.
-Diene’s C1 and C4 p orbitals must overlap with dienophile’s p orbitals to form new sigma bonds.
-Both sigma bonds are on same face of the diene: syn stereochemistry.
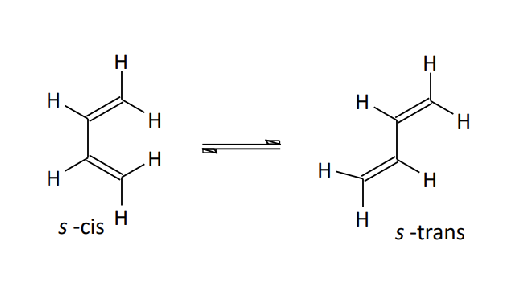
Explain the S-Cis Conformation of the Diene
→s-cis conformation: Can rotate around the C-C single bond to get the more STABLE s-trans conformation.
→s-trans conformation: Its more stable (12 kJ/mol) than the s-cis
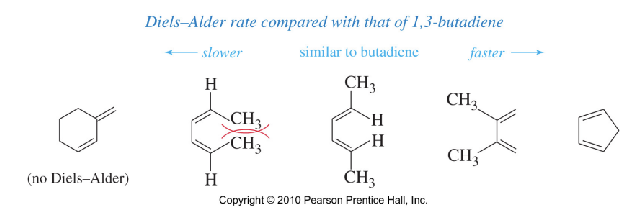
Explain the Diels–Alder Rate for Dienes
→The cyclopentadiene undergoes Diels-Alder rxn bc of its fixed s-cis conformation.
→When the diene is sterically hindered, the reaction
slows down even though if it has s-cis conformation.
→S-trans dienes do NOT undergo the Diels–Alder reaction
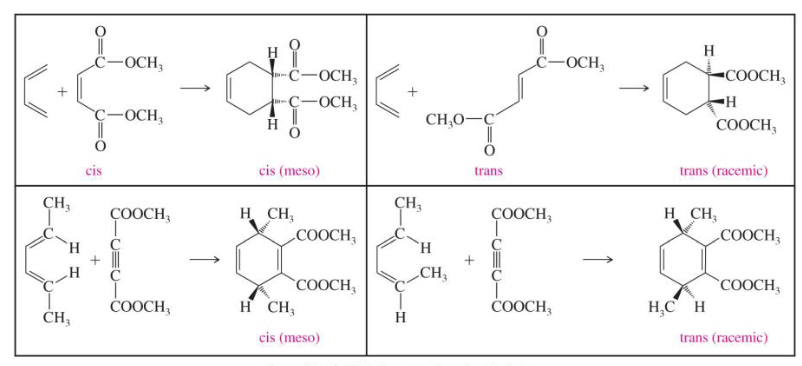
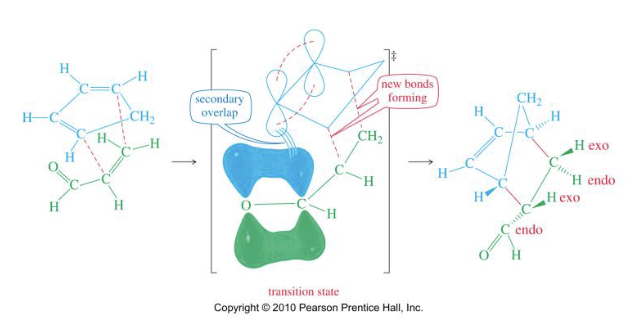
What is the Endo rule?
The p orbitals of the EWG on the dienophile have a secondary overlap with the p orbitals of C2 and C3 in the diene.
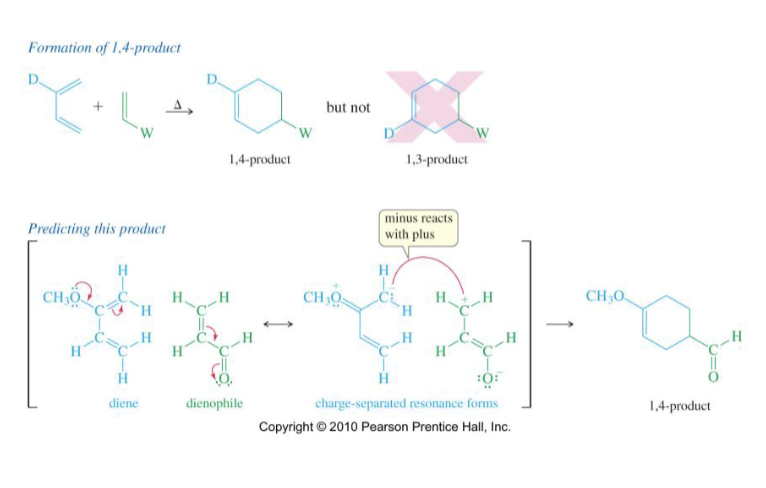
Draw mechanism of a 1,4 product
→A 1,4 product can form but a 1,3 product CANNOT
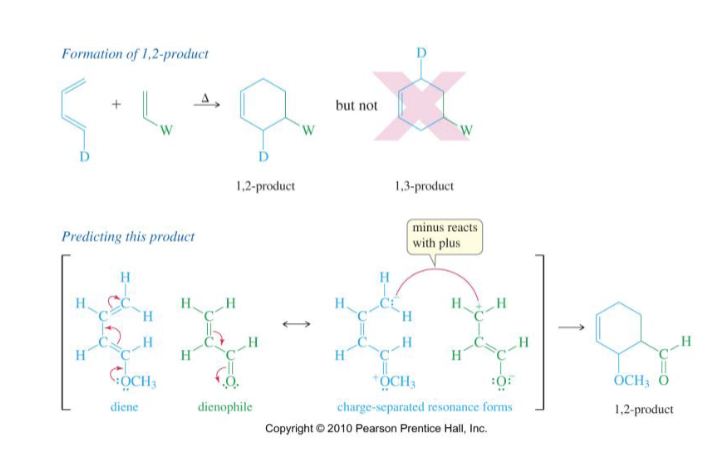
Draw mechanism of a 1,2 product
→A 1,2 product can form but a 1,3 product CANNOT
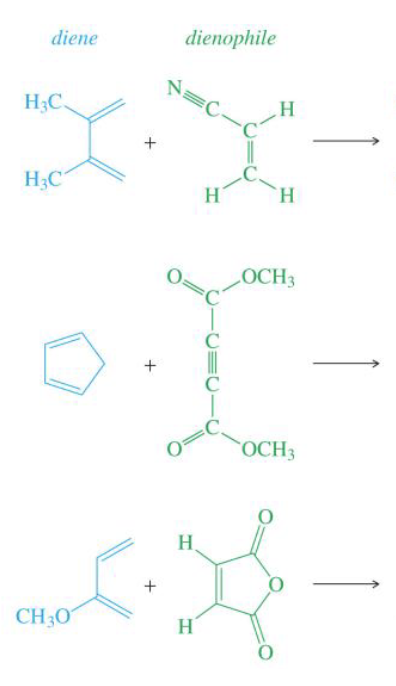
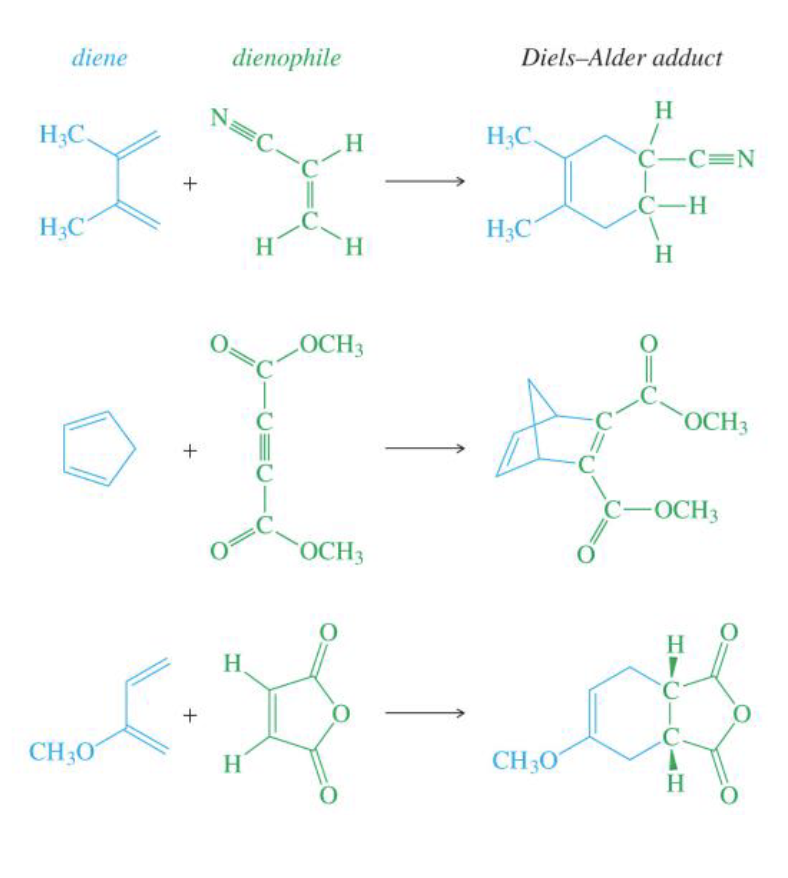
Draw the products of these of diels-alder reactions
Draw the mechanism of Diels-Alder reaction
-state which is the diene and dienophile
→one-step, concerted mechanism
→A diene reacts with an electron-poor alkene (dienophile) to give cyclohexene or cyclohexadiene rings.
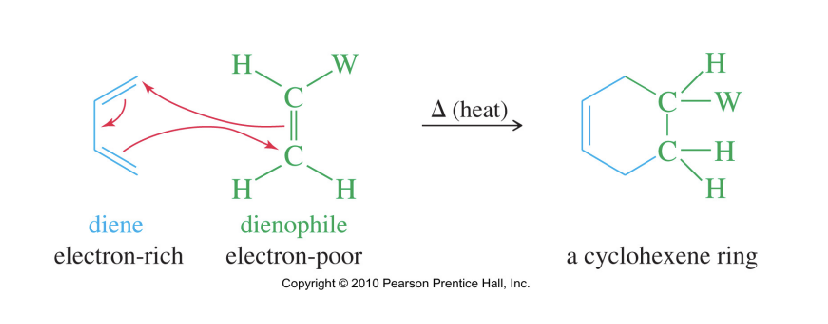
Draw the flow chart of Diels-Alder reaction
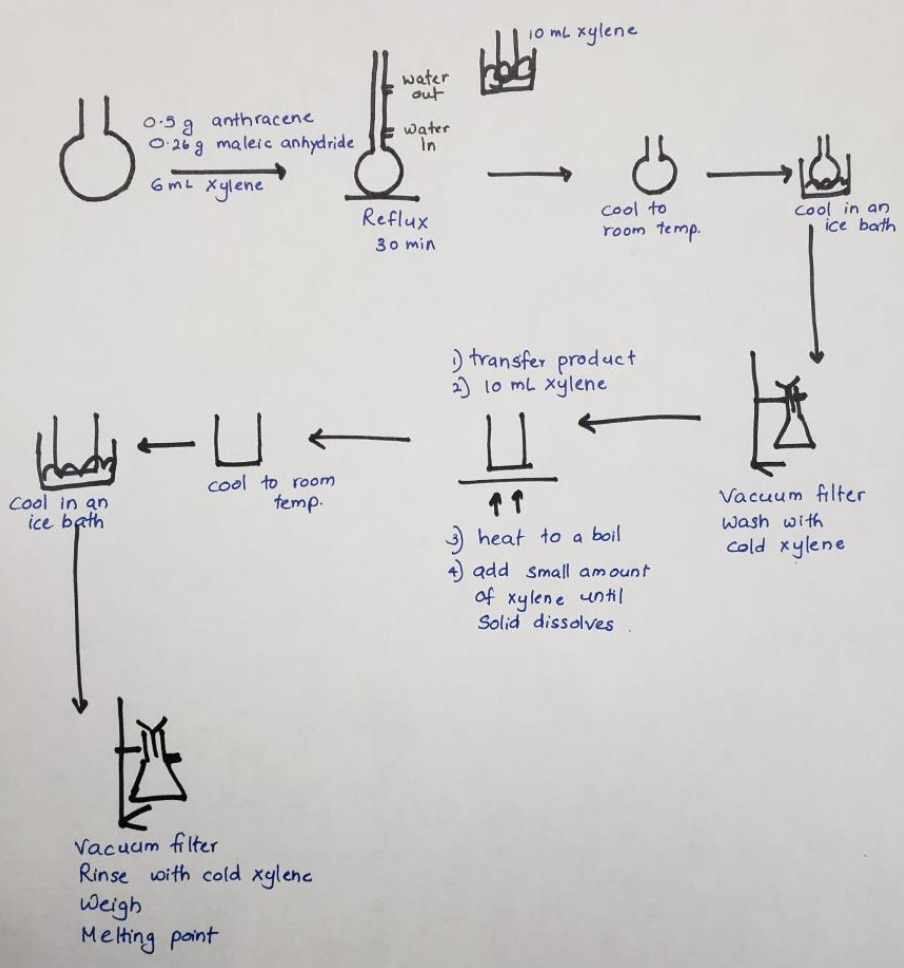
What are the 3 steps in this reaction
Add 0.5g of anthracene, 0.26g of maleic anhydride, 6mL of xylene and boiling chips into a round bottom flask for a 30min reflux.
Cool solution to room temp. Place solution & 10mL of xylene in ice-bath.
Vacuum filter & rinse with xylene. Check mp of dry crystals.