chemistry exam
1/83
Earn XP
Description and Tags
content through all of the booklets
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
84 Terms
atomic model: greek
principal characteristics: atoms are indivisible and indestructible
evidence: thoughts
atomic model: daltons
description of experiment:
law of conservation of mass compared to total mass before and after experiments
law of define proportions - measured mass of elements in the same compound
gas behaviour experiments
patterns in reactions
key findings:
atoms are re-arranged not destroyed
compounds form in fixed ratios of atoms
atoms combine in whole numbers
matter must be made of tiny particles
elements have unique identical atoms
atomic model: thomsons model
description of experiment:
Thomson discovered the electron by experimenting with cathode ray tubes, finding that rays were composed of negatively charged particles with very high charge - to - mass ratio.
using vacuum tube, electric plates, and magnetic fields, he determined these particles over 1000 times lighter than hydrogen proving atoms are divisible.
key findings:
atoms consist of positively charged ‘pudding’ or sphere, which spread throughout atom.
negatively charged electrons embedded randomly within this positive sphere
total positive charge are equal, making atom equally neutral
atomic model: rutherford
description of experiment:
beam of alpha particles from lead block containing radium (the radioactive alpha particle emitting source) targeted at very thin sheet of gold foil. the scattered alpha particles observed through microscope as cause flash light when strike the ZnS coated screen. most alpha particles in beam pass through gold foil undetected, some show slight deflection and very small deflected backwards.
key findings:
the nucleus incredibly small compared to total size of atom
proposed electron orbit nucleus at large distance with electrostatic attractive providing stability.
atomic model: bohr
description of experiment:
shined light through an element (gas) through a triangular prism giving off colours of light where he was able to measure wavelengths.
analysed the hydrogen emission spectrum to determine that electrons orbit the nucleus in specific quantised energy levels
key findings:
electrons orbit a positive nucleus in specific discrete circular orbits associated with fixed energy levels and electrons don’t radiate energy while occupying “stationary states”
explained spectral lines of hydrogen by introducing the concept of quantised energy levels.
atomic model: schrondinger
description of experiment:
any moving particle is associated with wave character
the schrondingers cat experiment absurdity of quantum super position.
key findings:
electrons behave as waves rather than particles.
replaced boars fixed orbits with orbitals three-dimensional mathematically defined regions of space representing probability of finding electron rather than specific path.
atomic model: Chadwick
description of experiment:
James Chadwick discovered the neutron by repeating a popular experiment that produced unknown radiation. placed polonium in a vacuum - sealed box which radiated alpha particles through a piece of beryllium, caused unknown radiation to accelerate out.
key findings:
neutron present in nucleus
neutrons and protons make nucleus mass
explanation of isotopes atoms of same element with same protons but different neutrons therefore unstable.
isotope
the number of neutrons of an element can vary, producing atoms that are slightly different.
isotopes have some chemical properties which are determined by their electron configuration eg. combustibility, flammability, reactivity.
different physical properties due to different mass eg. density melting boiling point.
forces in atom
electrostatic attraction: between the negative electrons and positive protons
electrostatic repulsion between the positive charged protons together in the nucleus. counteracted by the strong nuclear force acting between all nuclear particles
stable atoms has all these forces in balance
electron arrangement
electrons arranged in energy levels which are further organised in s p d and f orbitals
noble gases
unreactive inert gases found in group 18 full outer shell.
electron dot diagram
shows the number of valence electrons. known as lewis structures.
emission spectra and AAS
heating element vigorously can cause electron to absorb energy jump to higher energy state
as electron falls lower shell emits energy in form light corresponding to exact difference in energy between higher and lower energy.
electron may return directly to ground state or may move energy levels before returning.
as energy levels represent distinct quantised energies, each transition the electron makes is association with emission of a different quantity of energy in form different colours.
individual colour of light emitted by hydrogen atoms known as emission lines and collectively form emission spectrum.
atomic absorption spectroscopy is analytical technique that uses light absorption to measure the concentration of a metal in a sample.
AAS adresses limitation of flame test
AAS provides both quantitative and qualitative test
more than 70 elements can be analysed by AAS
AAS can detect elements in concentration as low as micrograms per litre
AAS highly selective and regularly used with mixtures
use of AAS
used in particularly for deflecting the concentrations of metal ions in solutions.
mining- metal concentration in mineral samples
water analysis- have metals, metal ions
biological tissues or fluids- heavy metals, toxicity levels
agriculture- trace elements in soil
determining concentration in AAS
create calibration curve by measuring absorbance of standard solutions of known concentration (absorbance y axis concentration on x axis) the standard solution should produce a straight line.
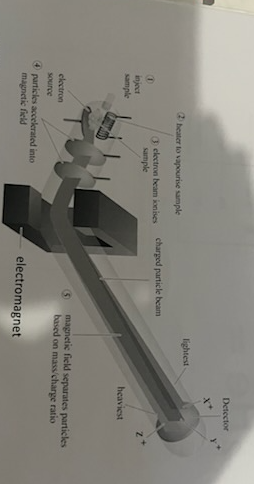
mass spectrometry
heat vaporising the sample- separating the individual isotopes in the sample
ionisation: positive ions are formed as the vapour passes through a high energy electron beam
acceleration: an electric field accelerates the ions to the same high kinetic energy
trends in periodic table
arranged by increasing atomic number (protons) so that similar electron configurations and hence chemical properties are together in columns and the same energy levels (shells) are together in a row.
valency
describes the combining power of an atom – how many atoms or groups that it would be capable of chemically bonding with.
atomic radius
represents the size of an atom usually measured in pm (10-12m). It is difficult to measure the radius of atoms in a consistent manner. Their size will depend on their state and whether they are an isolated or bonded atom.
ionisation energy
refers to the amount of energy required to remove an electron from an atom (in the gaseous state). It is measured in kJ/mol (electron removing energy).
electronegativity
describes the ability of an atom to attract the electrons of another atom in a bond (electron attracting power).
core charge
is an expression of the attractive force experienced by the valence electrons to the core (nucleus) of an atom. Core charge = number of protons – number of inner electrons
explain the trends of the atomic radium within a periodic table
Radius increases down a group:
extra energy level/shell is added which is further from the nucleus the radius increases
Radius decreases across a period:
within an energy level or shell as more protons are added the core charge increases. This increases the electrostatic attraction between the nucleus and valence electrons pulling the valence electrons closer and decreasing the radius
explain the trends of the ionisation energy within a periodic table
Ionisation energy decreases down a group:
valence electrons further from nucleus so experience a weaker attractive force. easier to remove as they are not held as tightly so ionisation energy is lower
Ionisation energy increases across a period:
atomic radii gets smaller across a period and the attractive forces are stronger due to the increased core charge (protons). Because the valence electrons are closer and held more tightly more energy required to overcome force and remove them. The ionisation energy increases.
Metals have low ionisation energies and non-metals have high ionisation energies.
explain the trends of electronegativity within a periodic table
Electronegativity increases across a period:
Across a period atoms smaller, have a more positive nucleus (core charge). able to exert greater attractive force on the electrons shared with another atom, pulling the electrons towards the more electronegative atom.
Electronegativity decreases down a group:
Down a group the radius increases so the attractive force of the nucleus must act over a larger distance. This results in a decreased attractive force acting on the shared electrons so a lower electronegativity.
Metals have low electronegativities and nonmetals have high electronegativities.
explain the trends of the core charge within a periodic table
larger core charge occurs across a period indicating a larger attractive force is acting to pull the outer electrons closer decreasing atomic radius.
also means that ionisation energy is increasing as more energy is needed to overcome this attractive force to remove an electron.
increase in electronegativity as the power to attract other electrons is also increasing.
groups within periodic table
alkali metals: group 1 highly reactive, soft, low density
alkali earth metals: group 2 shiny, highly reactive low density high electrical conductivity, high melting boiling point.
transitional metals: group 3-12 high melting/boiling points, high densities, and strong, hard, malleable structures
metals: group 13-16 high electrical and thermal conductivity, metallic lustre (shininess), high density, and high melting points
halogens: group 17 highly reactive
noble gases: group 18 odourless, colourless, nonflammable, and monatomic gases with extremely low chemical reactivity due to their complete valence electron shells
different types of bonding
metallic
ionic
covalent molecular
covalent network
allotropes of carbon
graphite
structure: covalent network, each carbon bonded to three other carbons, one delocalised electron per carbon atom.
properties: conductive, slippery, soft, greasy material.
uses: lubricant, pencils
diamond
structure: covalent network lattice, each carbon surrounded by four other carbon atoms in a tetrahedral arrangement.
properties: very hard, sublimes, non-conductor, brittle.
uses: jewellery, cutting tools, drills.
amorphous
structure: no crystalline structure. Amorphous carbon materials may be stabilized by terminating dangling-π bonds with hydrogen. As with other amorphous solids, some short-range order can be observed.
properties: conductive, non-crystalline, cheap.
uses: printing ink, carbon black filler, activated charcoal, photocopying
buckyball
structure: hollow, spherical, cage like structure. hexagonal lattice. 3 covalent bonds to each carbon atom. delocalised electrons.
properties: dark brown to black, highly symmetrical, stable, strong, incompressible.
uses: radical scavenger and antioxidant, used in organic solar cells.
(fullerene) nanotubes
structure: hollow cylindrical structure, with walls that are only one atom thick. hexagonal lattice, similar to graphene.
properties: highly elastic, high thermal conductivity and low density.
uses: batteries, solar panels, LED’s, sensors.
graphene
structure: single layer of carbon atoms, tightly bound in a hexagonal honeycomb lattice “one” instead of “its” indicates presence of double bonds within carbon structure.
properties: tough, flexible, light with a high resistance
uses: replace silicon as the basis for computer chips and circuits due to its high conductivity
can be used in organic photovoltaic cells
ionic compounds
ionic compounds can be represented using electron dot diagrams. ions tend to form based on the octet rule, that is most atoms will lose or gain electrons to achieve a more stable valence electron configuration of 8.
covalent compounds
covalent compounds form between non-metal atoms as the atoms have high electronnegativities. atoms will share electrons in order to fulfil the octet rule.lectrone
electron dot diagram for ionic compounds
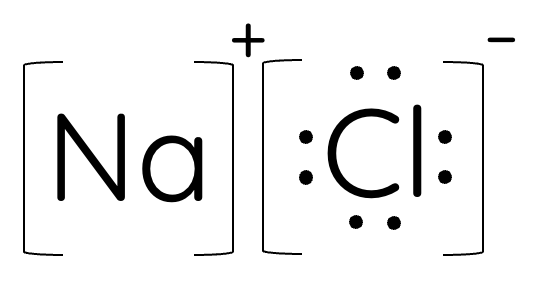
electron dot diagram for covalent molecular compounds
electron dot diagram for metallic bonding
just the symbol
non octet compounds
there are some compounds that do not follow the octet rule. these occur in compounds when there are an odd number of valence electrons, where there are too few valence electrons or when there are too many valence electrons.
define nanomaterials
a substance containing particles which range in size from 1 to 100 nanometres (nm)
1nm= 10 to the power of -9 metres ( a billionth of a metre )
examples of nano materials
spider silk
carbon fullerens
graphene
concerns of using nano materials
health hazards: inhaled nanoparticles may reach deep in the lung and travel to other organs
environmental persistence: many engineered nano materials do not easily degrade
workplace exposure: production of dry powders pose high risk for inhalation
uncertain toxicology: their small size allows them to pass through biological membranes
materials
a substance that has mass/ occupies space and is used for a purpose due to its particular qualities.
pure substance
made only of 1 type of particle ie. elements and compounds
mixtures
matter that contains 2 or more different materials or substances with varying composition
homogeneous
having uniform composition aka not a mixture
heterogenous
having non-uniform/ varying composition aka bread
elements
a pure substance which is only composed of atoms of the same atomic number
compounds
a pure substance composes of more than one type of atom chemically combined in fixed proportions. ie. has a set formula
physical change
change to the physical properties so chemical composition remains the same
physical properties
properties observed without changing the chemical identity eg. hardness, density, colour, boiling point.
chemical change
involves a chemical reaction so the chemical composition (formula) changes.
chemical properties
properties observed by preforming a chemical reaction eg. flammability, oxidation, heat of a reaction, reaction with acids.
what characteristics determine how matter is classified
particle type present- one type or multiple types
arrangement of particles- uniform or varied
physical properties- fixed or varied/ range
key ideas about chemical and physical properties and changes
properties help determine if a substance is pure or a mixture
pure substances have properties that don’t vary
the properties of a mixture will vary depending on the mixture’s specific composition
mixtures can be separated into their components using physical changes. these are known as separation techniques including processes such as filtration, distillation and evaporation.
electrostatic seperation
method of separation
properties used in separation- difference in electrical charge
explanation/description- charged particles attracted to charged plates and uncharged particles move past
example- separating waste gas products from chimney. particles are attached to charged plates
magnetic seperation
method of seperation
properties used in separation- magnetic susceptibility (force of attraction to a magnetic field)
explanation/description- a process that uses powerful magnets to separate magnetic, ferocious materials form non-magnetic materials
examples- recovery of steel or iron, such as crushed food cans, scrap metal.
filtration and serving
method of separation
property used in separation- difference in particles size
explanation/description- used to seperate a mixture of solid particles form a liquid or gas. passing mixtures through the mesh or filter papers.
example- pool filters, coffee, pasta sieve/ strainers.
vaporisation (evaporating/boiling)
property used in separation- boiling point
explanation/ description- the compound with lower Bp evaporates off and it is lost to the atmosphere solid residue remains (solvent is evaporated off)
example- salt from water, production from crystals, distillation
distillation and fractional distillation
seperation method
properties used in sepration- boiling point
description/ explanation- the component with the lowest Bp evaporates first then re-condenses and is collected
example- canola oil, alcohol
decantation/ sedimentation
separation method
properties used in separation- density
description/ explanation- more dense component sticks to the bottom
example- stock preparation
separating funnel/ solvent extraction
separation method
properties used in separation- solubility in 2 immiscible liquids
explanation/ description- one component of the mixture dissolves into the top layer, another component into the bottom layer
example- oil and water
centrifuge
separation method
properties used in separation- density (faster sedimentation)
explanation- centrifuge force pushes the dense component to the bottom of the tube
example- blood cells
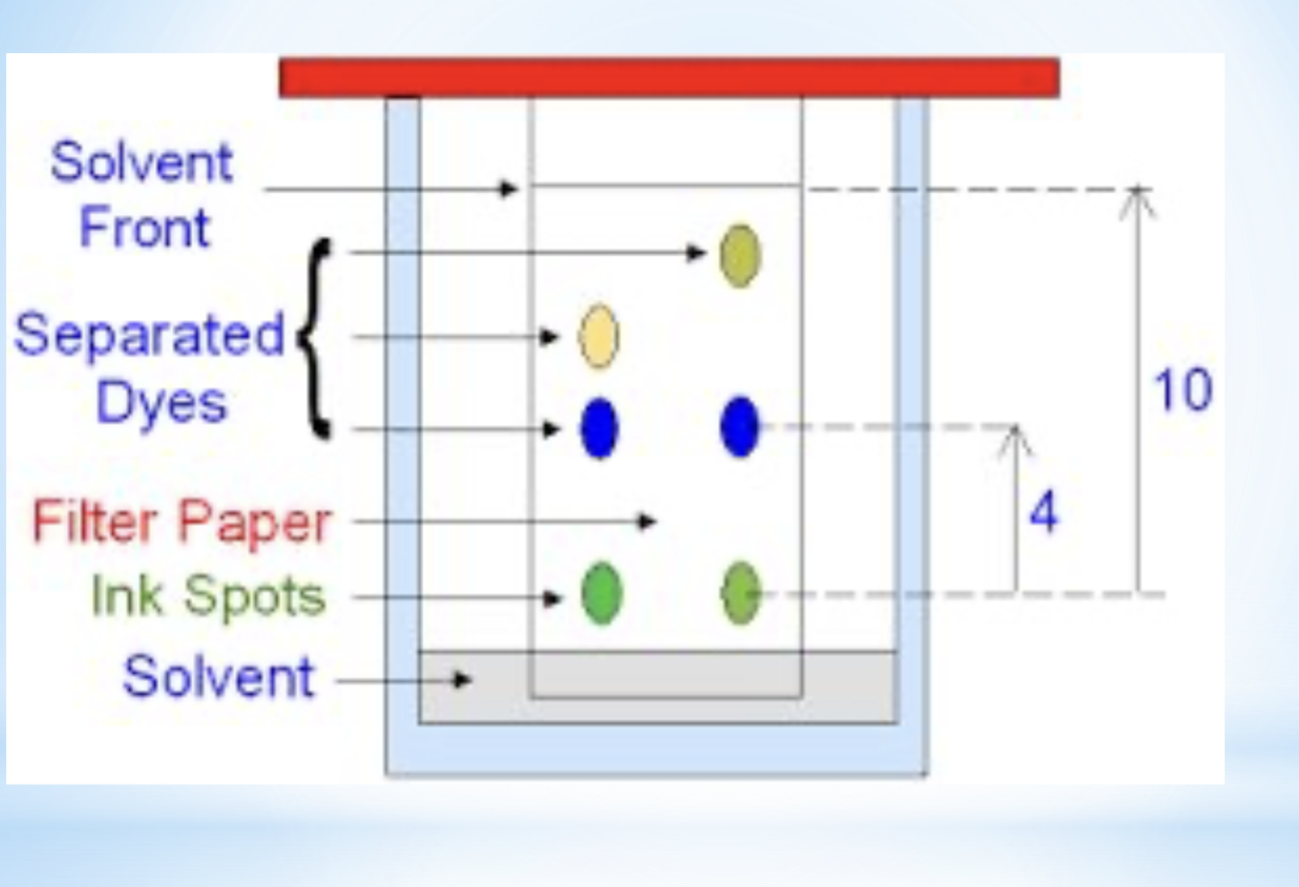
chromatography
seperation method
properties used in separation- adhesion (how well it sticks to the surface)
explanation- component that adheres more than one substance/ paper faster.
example- seperate dyes in ink
moles calculation
a mole is a convenient quantity for counting particles. the mole is given the symbol n and unit mol.
one mole is defined as the amount of substance that contains the same number of (specified) particles as there are atoms in 12g of carbon-12
the number of particles in 1 mol given symbol “N”. this is known as abagadros number and has the numerical value 6.022 × 10 to the power of 23.
the molar mass of an element or compound is the mass, in grams, of 1 mole of that element or compound. Molar mass is given the symbol M and the unit gmol-1
the molar mass of an element or compound has the same numerical value as the relative mass of the element or compound.
formula for the mass of moles in grams
m= n x M
formula for the number of moles
n= m/M
formula for the number of moles from the number of particles given
n= N/abagadros number
formula for the number of particles
N= n x abagadros number
interpreting formula
the chemical formula tells us what atoms are needed and how many. we can also work in moles rather than individual atoms. the formula tells us the proportion of each element requires in the compound.
what is stociochemistry
derived from the greek word “Stoicheion” or element
it is about being able to predict quantities of a product or reactant in a chemical reaction
to do this a balanced equation is required and a quantity needs to be known in moles
a conversion factor known as a more ratio enables on e quantity in a reaction to be predicted from another
balancing chemical equations
follow the law of conservation of mass ( equal on both sides)
the number of atoms of each type are equal on each side of the reaction
the formulae cannot be changed only coefficients can be added
it represents how many of each molecule or atom reacts
to determine the moles of one compound (unknown) when given the moles of another (known) what is the formula

stochiochemistry steps
write a balanced equation
calculate the moles of the known compound using n = m/M
determine the moles of the unknown compound using the mole ratio
answer question by converting mass m= n x M and rounding the correct number
percentage composition
the percentage composition of a compound gives the percentage by mass of each element in the compound.
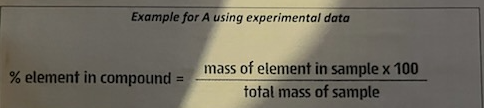
what is the flame test
qualitative, analytical chemistry procedure used to detect the presence of specific metal ions based on the characteristic colors they emit when heated in a Bunsen burner flame
how can the flame test be used
identify an element/ which particular metal ion is present
why is distilled rather than tap water used
contamination so no other metal ions are present
relative atomic mass calculation
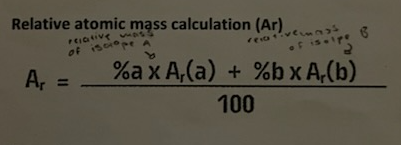
diagram for the separation technique electrostatic separation
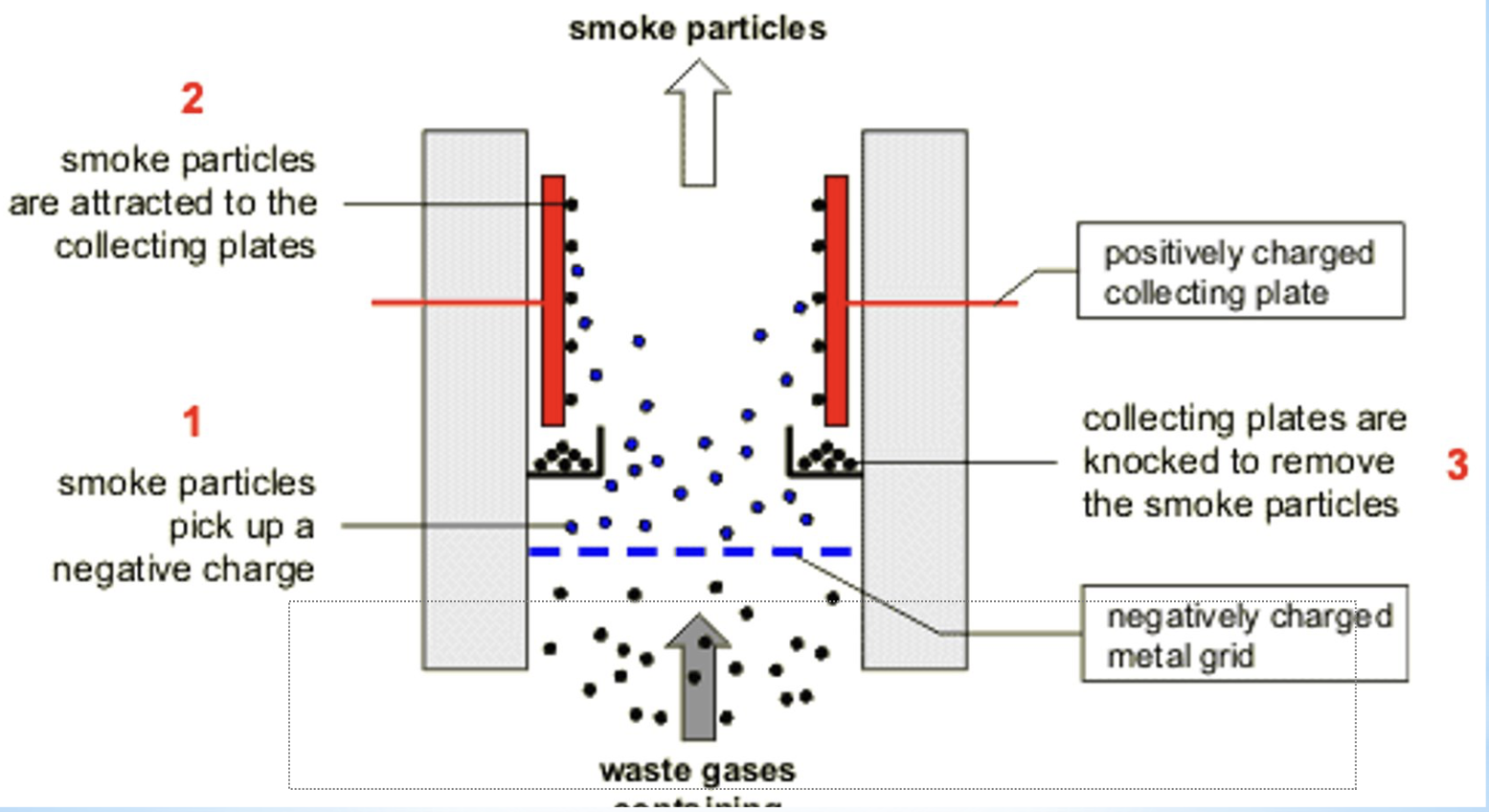
diagram for the separation technique magnetic separation
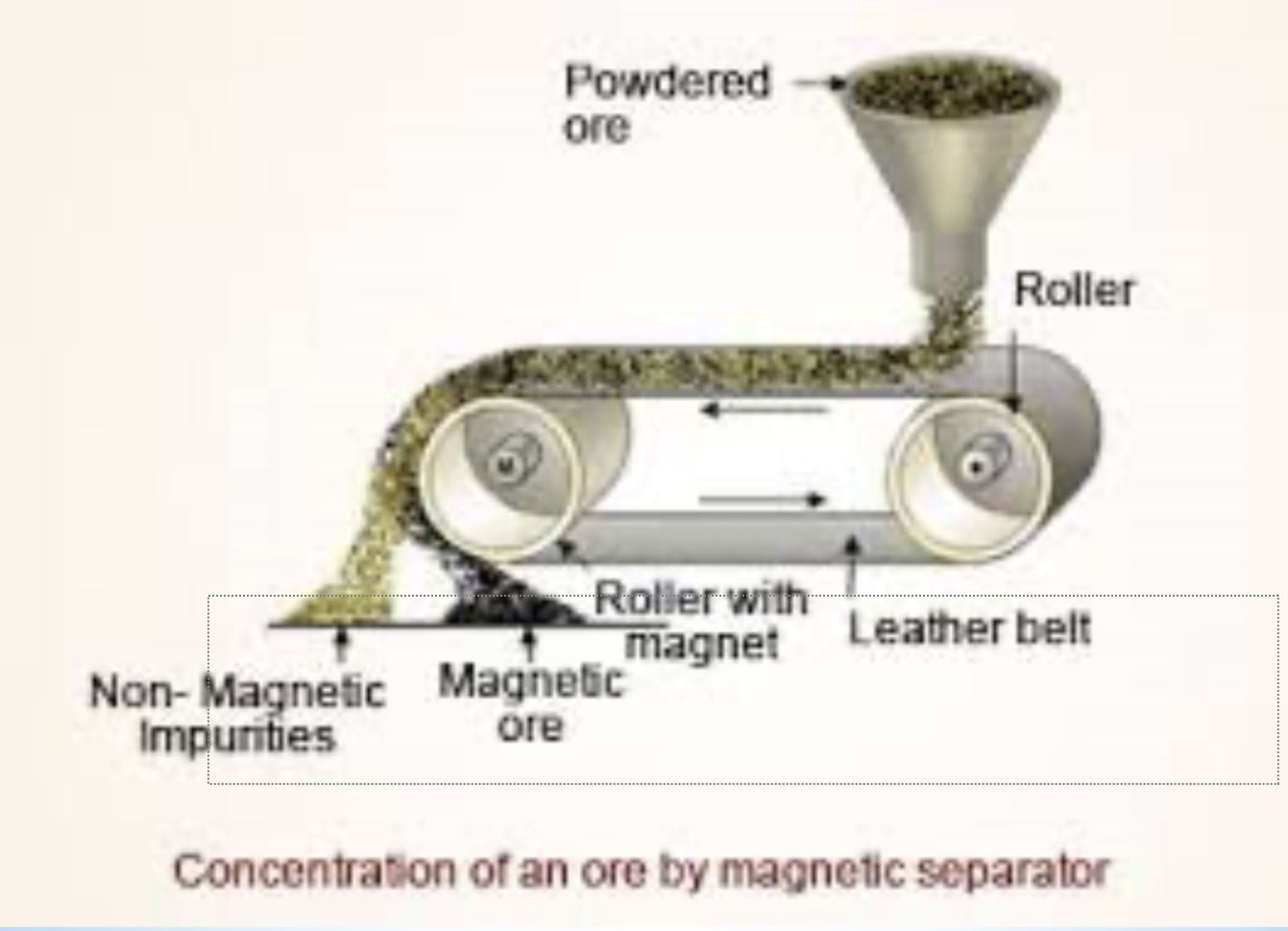
diagram for the separation technique filtration and serving
diagram for the separation technique vaporisation
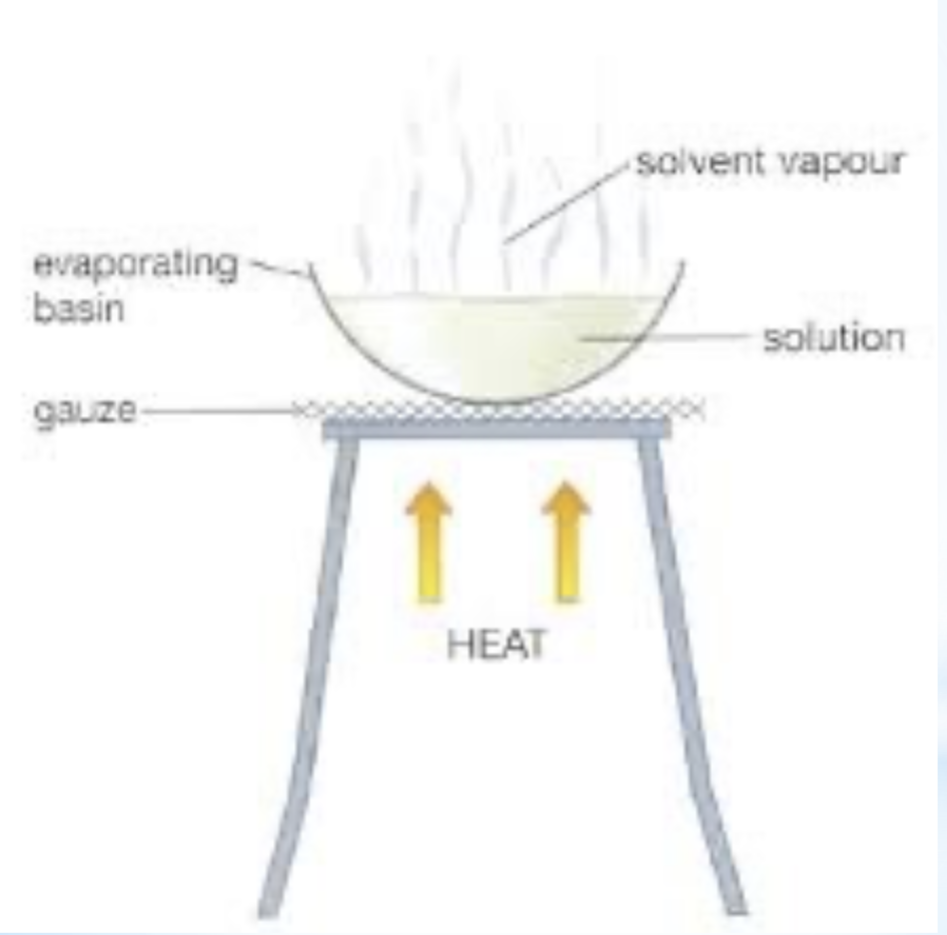
diagram for the separation technique distillation and fraction distillation
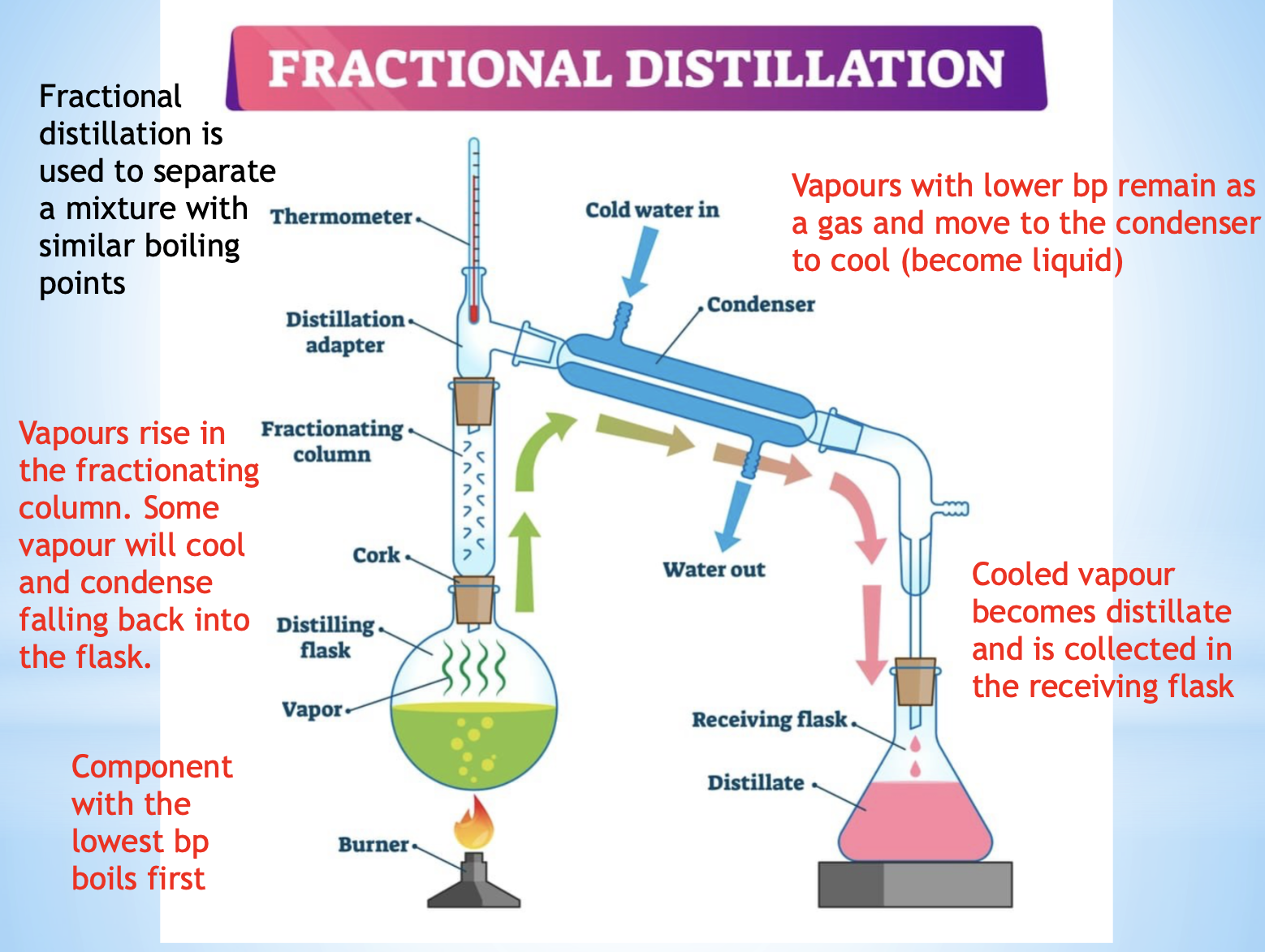
diagram for the separation technique decantation/ sedimentation
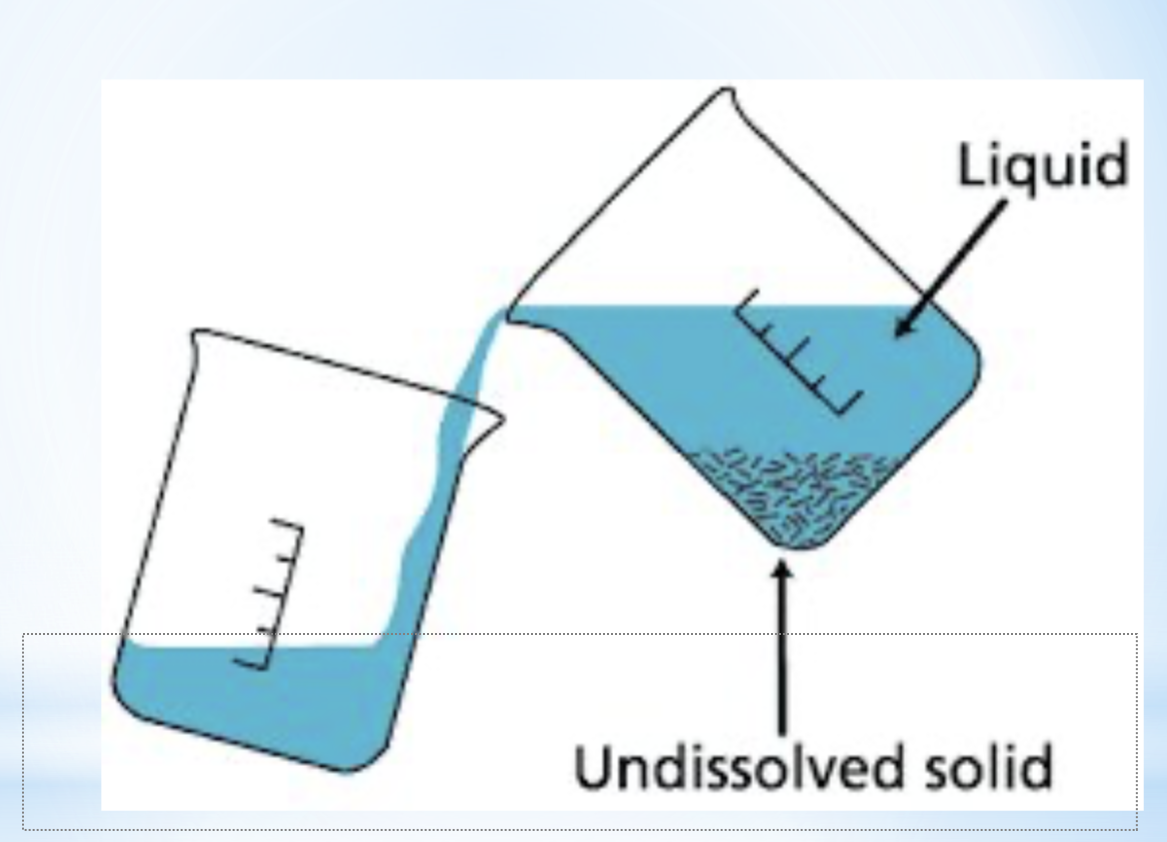
diagram for the separation technique funnel/ solvent extraction
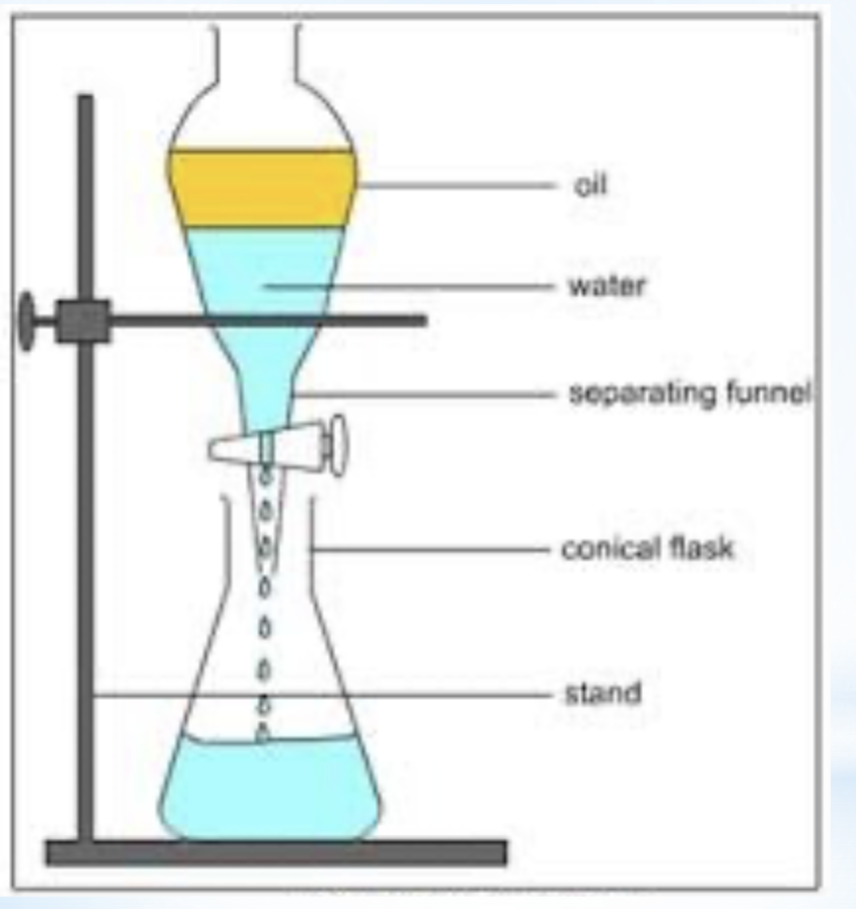
diagram for the separation technique centrifuge
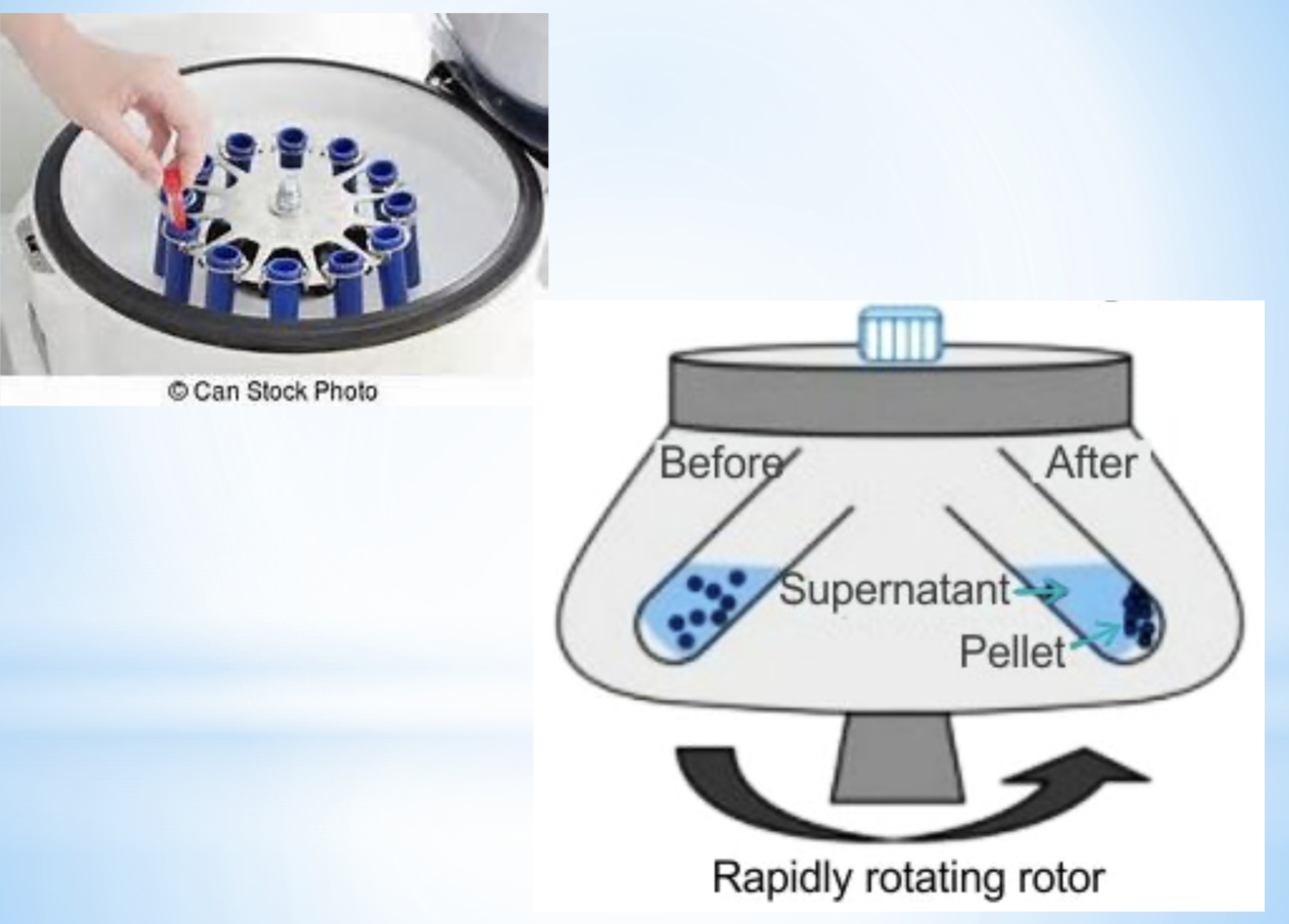
diagram for the separation technique chromatography