Amines & Amides: Reactions
1/8
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
9 Terms
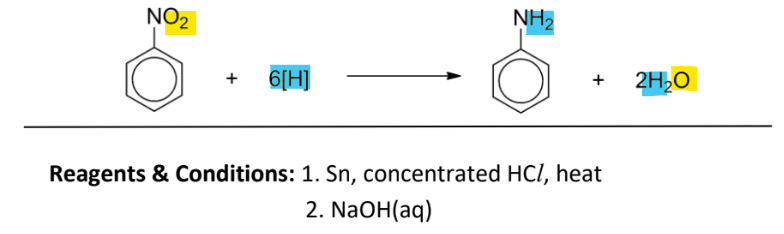
Reduction of nitrobenzene to produce amine
RnCs:
Sn, concnetrated HCl, heat
(after salt is formed) NaOH(aq)
Note:
product first forms the salt C6H5NH3+Cl-, then excess NaOH is added to liberate phenylamine
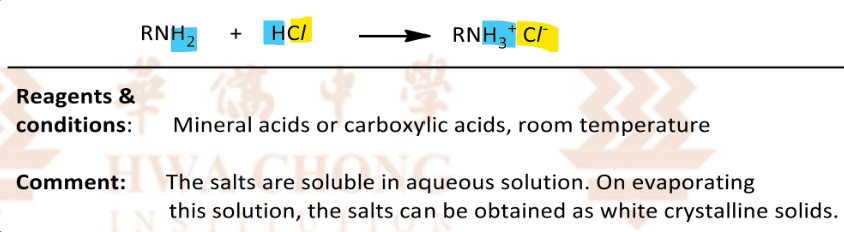
Reaction of amines with acids
RnCs: mineral acids or carboxylic acids, room temperature
Note:
soluble ionic salts are formed through acid-base reactions involving the insoluble amines
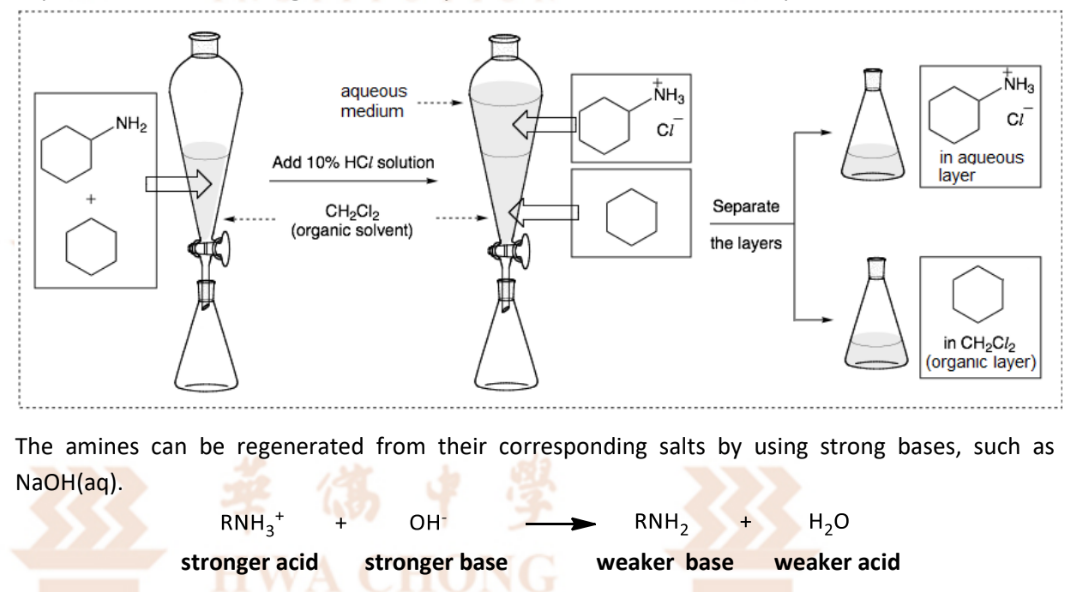
Regenerating amine through acid-base reactions
in an organic solvent in a separating funnel, first react with mineral acid/carboxylic acid
and obtain soluble ionic salt as it is insoluble in the solvent
react salt with strong base eg NaOH
Nucleophilic substitution to form primary, secondary and tertiary amines
RnCs: NH3 OR amine in ethanol, halogenoalkane heated under high pressure
Note:
lone pair on nitrogen(amine acts as a nucleophile) causes further reactions ot occur to form more substituted amines

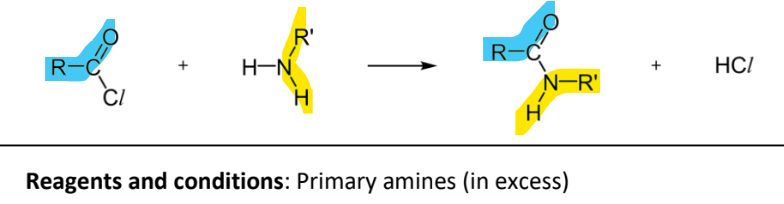
Condensation to form amides
RnCs: acyl chlorides, primary/secondary amines in excess
Note:
in excess to ensure complete reaction as some amines wil react with HCl in acid-base reaction, having excess will neutralise the HCl and ensure there is still amines to react with unreacted acyl chloride
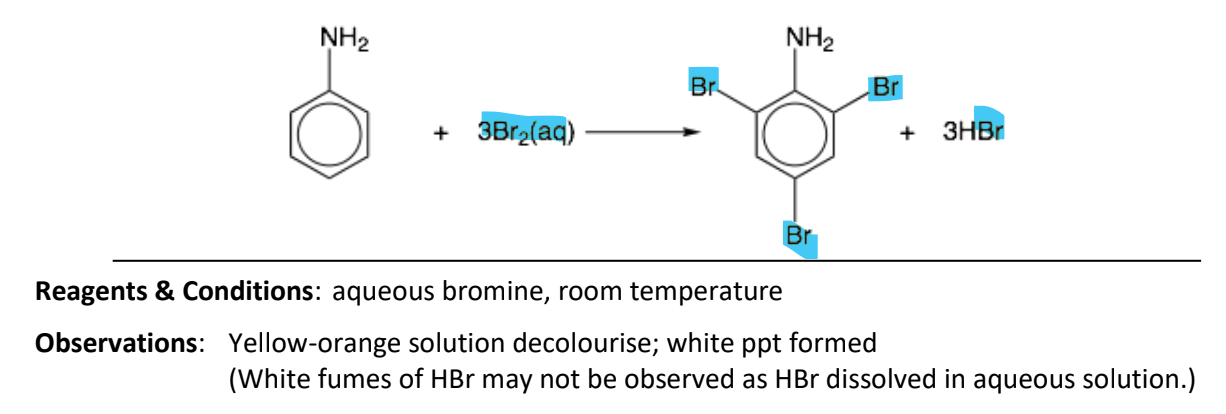
Electrophilic substitution of phenylamine to form 2,4,6-tribromophenylamine
RnCs: aqueous bromine, room temperature
Observation: yellow-orange solution declourise, white ppt formed (white fumes of HBr may nto be observed as HBr aqueous solution)
Notes:
-NH2 group is 2,4-directing
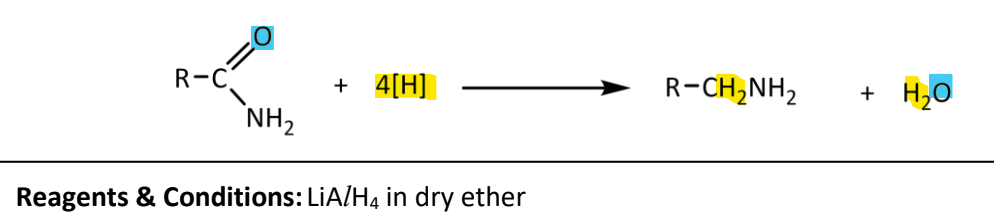
Reduction of amides
RnCs: LiAlH4 in dry ether
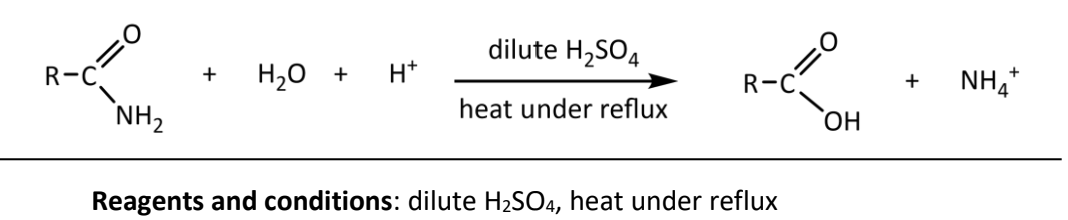
acidic hydrolysis of amides
RnCs: dilute H2SO4, heat under reflux
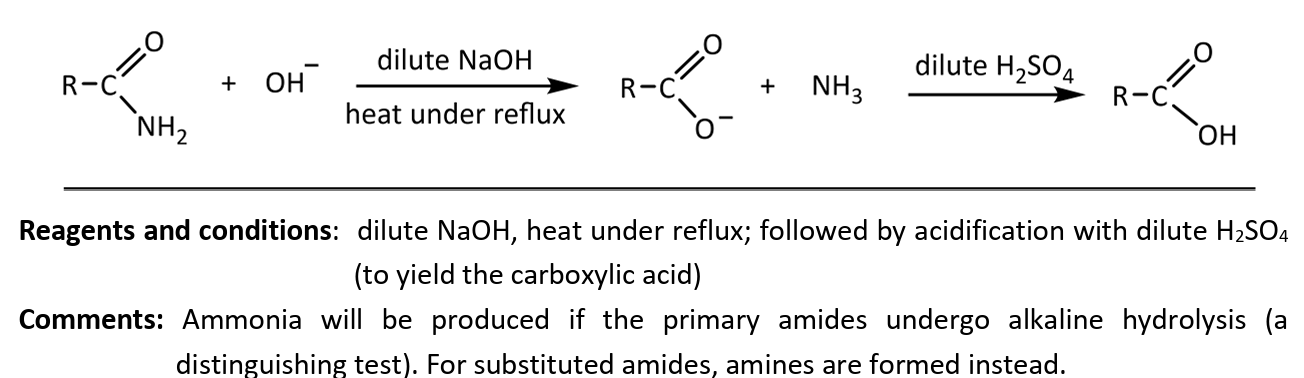
alkaline hydrolysis (followed by acidification)
RnCs: dilute NaOH, heat under reflux, followed by acidification with dilute H2SO4 (to yield caroxylic acid)
Note:
distinguishing test for amines as ammonia will be produced upom undergoing alkaline hydrolysis