ignore chemistry ch4
1/62
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
63 Terms
elements
pure substances from which other things are build
cannot be broken down into simpler substances
chemical symbols
1-2 letter abbreviations for elements name
periodic table is organized by groups w —- propeties , and in order of increasing —- —-
similar
atomic mass
who created the periodic table?
dmitri mendeleev
groups are…
down a periodic table, vertical
periods are …
across a periodic table, horizontal
7 rows
where are the representative element?
signified by “a”
group 1-2, 13-`18
what are the transition elements
stuff in the middle
“b”
group 3-12
from left to right the atomic number ===
increases
alkali metals
Lithium (Li)
sodium (Na)
potassium (K)
halogens
chlorine (Cl2)
bromine (Br2)
iodine (I2)
metals are located where on the periodic table?
to the left of staircase
metalloids are where?
on the zigzag line, starting with Boron (B)
excluding Al
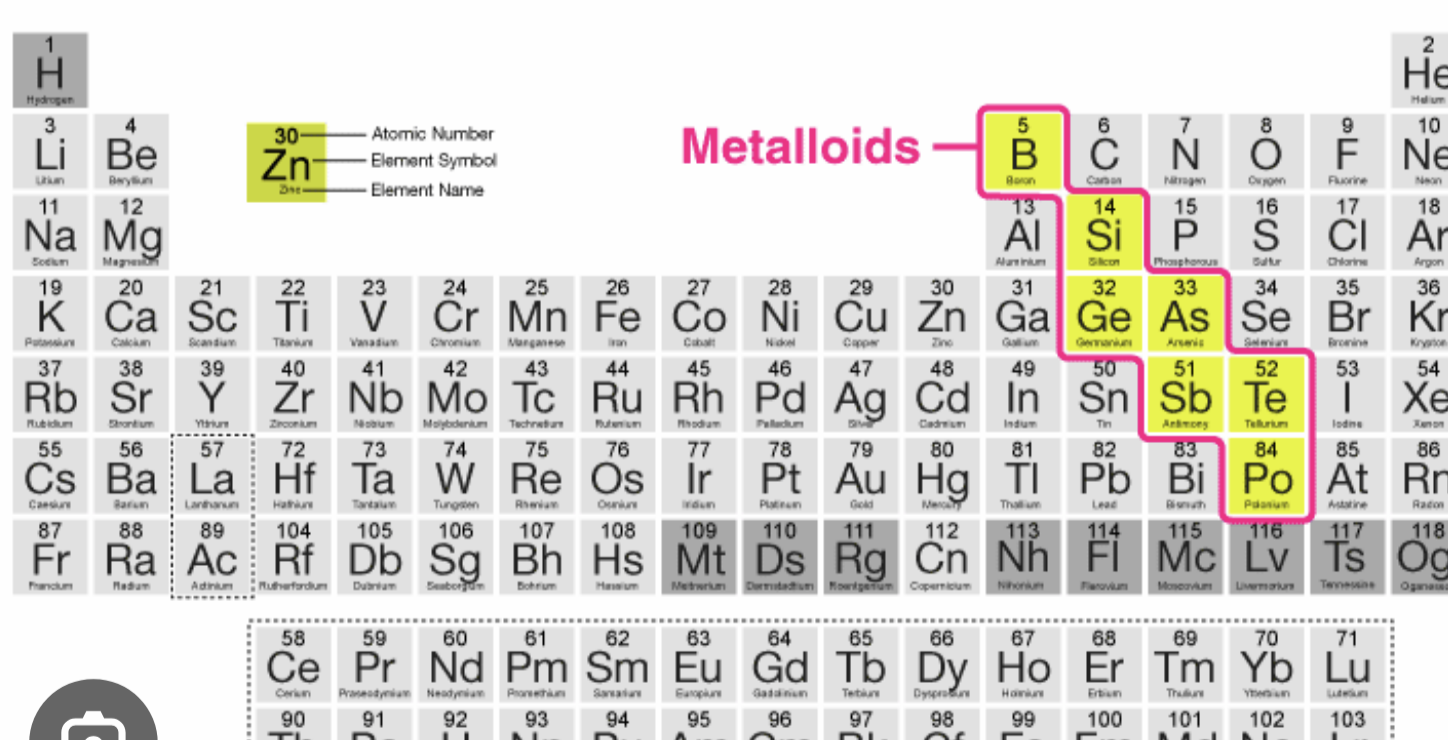
what are the macrominerals from the periodic table
needed in the body in large amounts
Ca, P, K, Cl, S, Na, Mg
blue= macro
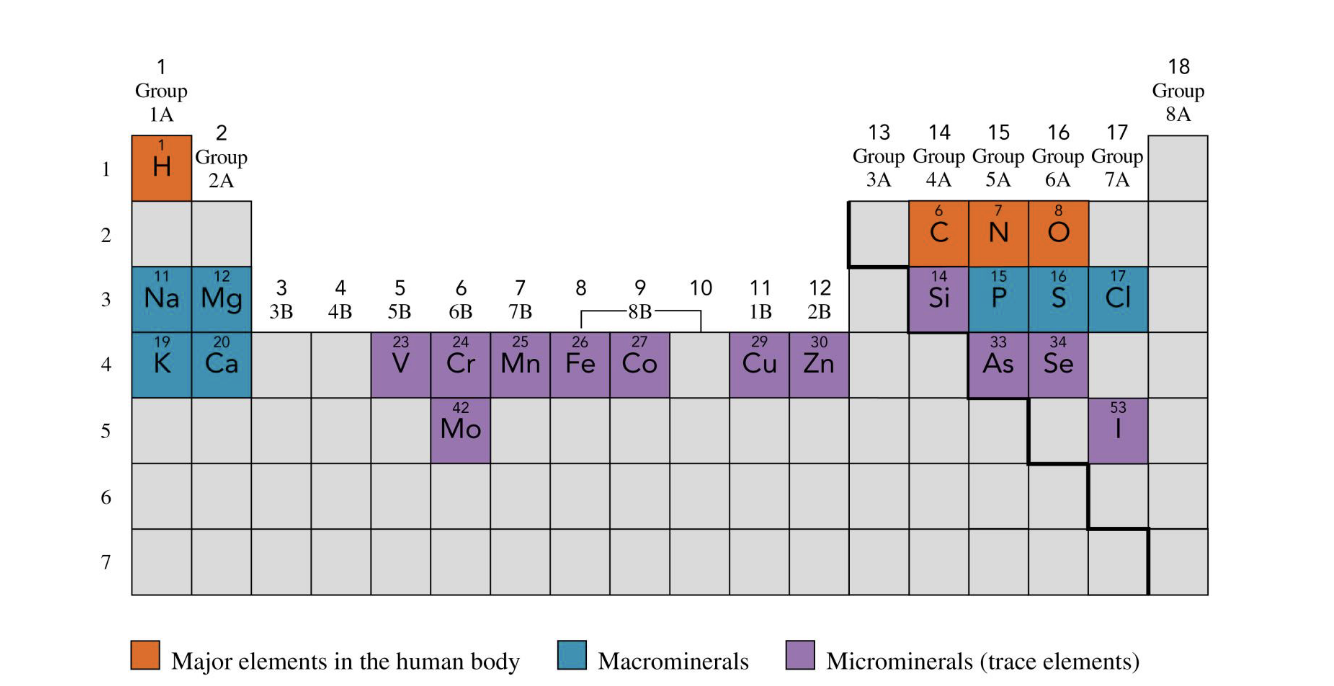
what are the microminerals from the periodic table
V, Cr, Mn, Mo, Fe, Co, Cu, Zn, Si, As, Se, I
purple
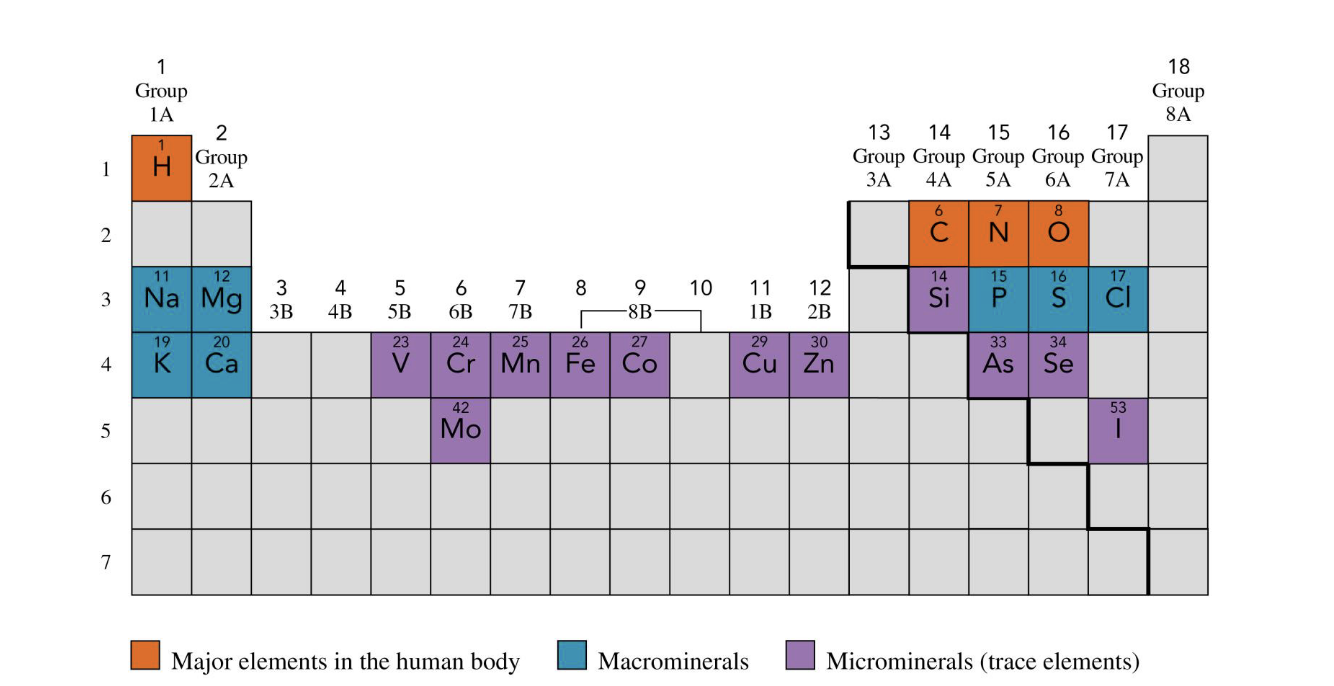
major elements in human body
H, C, N, O
orange
structure of an atom
nucleus containing protons (+) and neutrons(0)
the electrons (-) surround the nucelus
mass of the atom
mass of atom is due to proton and neutron in nucleus, electrons are much smaller
measured in amu
atpmic spectrum
when light from a heated element passes thorugh a prism, it seperates into distinct lines of color seperated by dark areas
lines in atomic spectrum are assoc w the changes in energies of electrons
electron energy levels
the lines in atomic spectrum are assoc w the changes in energies of electrons
each atom has a specific energy level —> assigned values called principle quantum numbers
highest priniipal quantum number =
lower prinicpal quantum number =
why?
higher energy
lower energy
the further away you get from nucleus, more energy
energy of electron is quantized.. what does that mean ?
electrons can only have certain energy values
energy level increases as the number gets higher
when electrons change from a lower level to a higher level…
they absorb the energy equal to the change in energy levels
when electrons change from high to lower level…
they emit energy equal to the change in energy levels
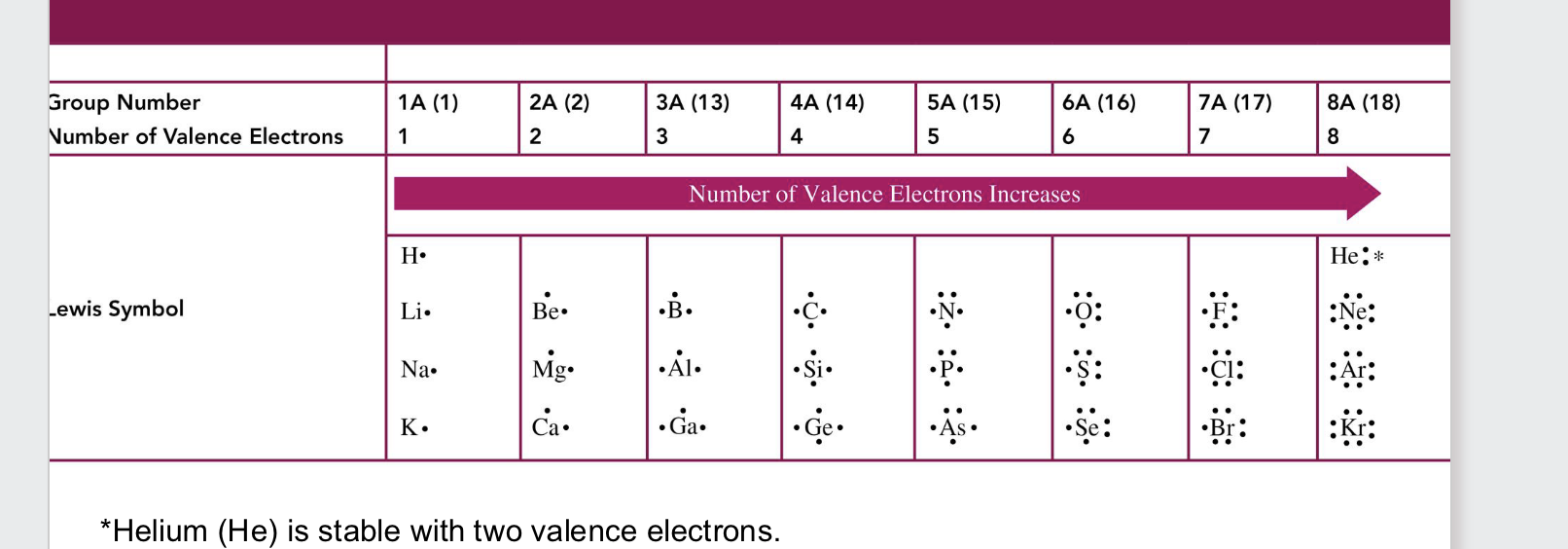
valance electrons
electrons in the outermost shell/energy level
lewis symbol
represents valence electrons in an element
SH of water (cals) (joules)
1.00 cal g/c
4.184 J/g c
Octet rule and stability
Chemical bonds are formed when atoms lose, gain, or share
valence electrons to acquire an octet of eight valence
electronswant to have an octet
Ionic bonds
transfer or shared?
melting point?
state?
occur when valance electrons of an atom of a metal are transferred to a nonmetal
strong attractive forces between negative and positive ion
high melting points
solid at room temp
metal+ nonmetal
How to name an ionic compound?
How to name an ionic compound?
name of metal= first
name of nonmetal is first syllable of nonmetal name + ide = second
ex: magnesium bromide
Covalent Bond
definiton
composition
properites
Definition: A bond formed when two nonmetals share valence electrons to reach the magic number 8 (Octet Rule).
Composition: Nonmetal + Nonmetal.
Properties:
Lower Melting Points: They turn into liquids or gases much easier than ionic salts.
naming a covalent compound
first= nonmetal , named by element name
second= nonmetal, followed by ide
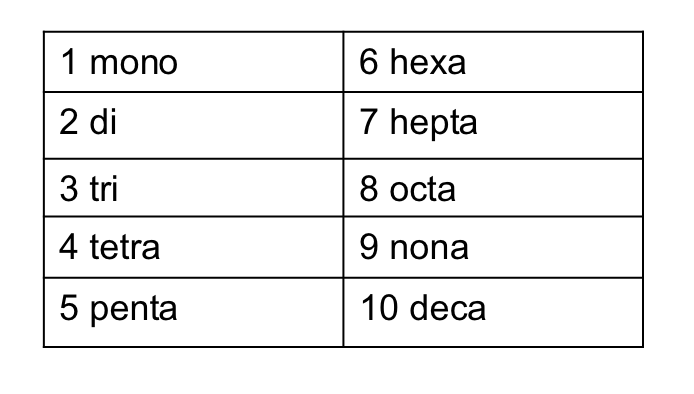
when subscript indicateds 2+ atoms= use a prefix
1: Mono-
2: Di-
3: Tri-
4: Tetra-
5: Penta-
6: Hexa-
What are the metals with variable charges?
transition elements except for Zn2+, Cd2+, Ag+
form two or more positive ions
Transition Metals & Roman Numerals
The Rule: Use Roman Numerals for Transition Metals (the middle of the table) because they can have more than one possible charge.
I - first
II - second
III - three
IV - four
V - five
polyatomic ions
covalently bonded groups of 2+ atoms w overall charge
“package deal”
usually nonmetal covalently bonded to oxygen atoms
negative charge except for ammonium (NH+4)
naming polyatomic ions
most common polyatomic atoms end in -ate
ex: sulfate , phosphate, nitrate
when a related ion has one less O atom= ends it -ite
ex: sulfite, phosphite, nitrite
naming compounds w polyatomic ions
first= positive ion, usually metal
second= name of polyatomic ion
no prefixes used
prefixes for convalent compounds 1-10
Which three Transition Metals never get a Roman Numeral?
Silver (Ag): Always +1
Zinc (Zn): Always +2
Cadmium (Cd): Always +2
Covalent compounds
form when atoms of nonmetals share valence electrons to achieve stability
nonmetal + nonmetal
covalent
double bond
triple bond
occurs when atoms share two pairs of electrons
occurs when atoms share three pairs of electrons
forms when there are not enough electrons to complete octets
what kind of bonds have polarity?
covalent bonds
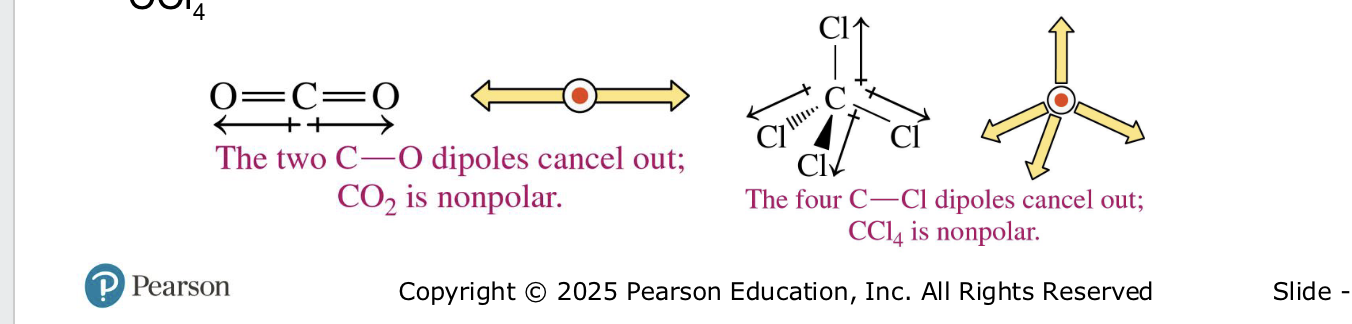
non polar molecules
H2, Cl2, O2 —> nonpolar
polarbonds can be nonpolar if they cancel each other out in symmetrical arrangement (CO2, CCl4)
electrons shared equal
polar molecules
one end of the molcule= more negatively charged
polar bonds do not cancel out
electrons shared unequally
ex: HCl

How does an Ionic Bond differ from a Polar Covalent bond in terms of electrons?
No sharing. One atom is so strong it completely steals the electron.
Composition: Usually a Metal + Nonmetal.
Predicting bond type by electronegativity differences
???
electronegativity
the measure of an atom's ability to attract shared electrons in a chemical bond towards itself
most electronegative element
least electronegative element
F
Cs
ionic bonds
strongest most attractive force
solids at room temp
high melting pt
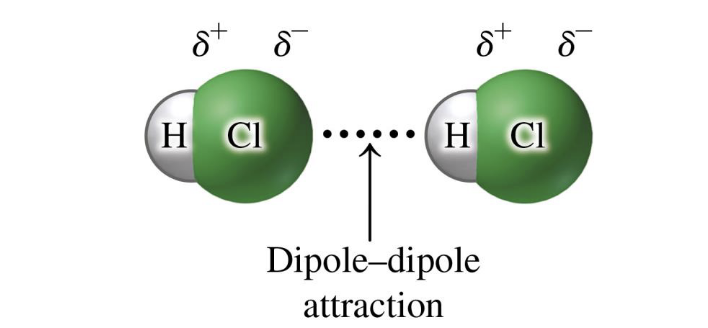
dipole dipole attractions
in covalent polar molecules
have dipoles where positively charged end of dipole in one molecule is attracted to negatively charged end of the dipole in another molecule
hydrogen bonds
hydrogen atoms bonded to F, O, N or a lone pair of F, O, N
dispersion forces
weak attractions between nonpolar molecules
caused by temporary dipoles
make it possiuble for nonpolar molecules to form liquids
what are diatomic molecules and which elements are they?
diatomic molecules exist in nature in twos
hydrogen, nitrogen, flourine, chlorine, bromine, iodine, oxygen
Have No Fear of Ice Cold BeeR
exceptions to the octet rule
hydrogen fills octet with 2 ve
central atoms w 2 electron groups
linear, 180 degrees
CO2
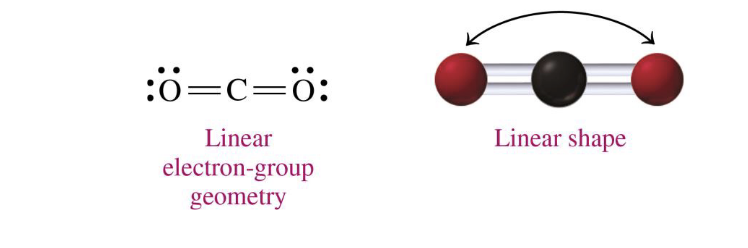
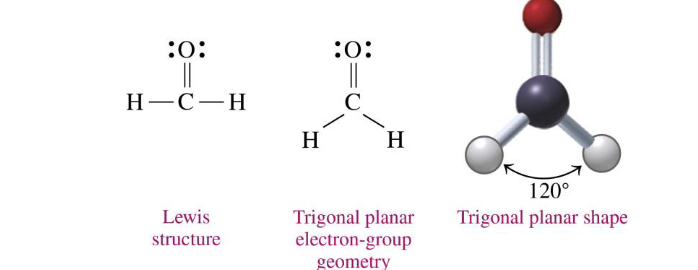
central atoms w three electron groups + 0 lone pairs on central atom
trigonal planar 120
H2CO
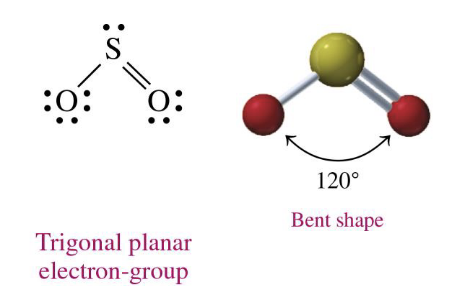
central atom w 3 electron groups (one of which is a lone pairs on the central atom)
bent 120
SO2
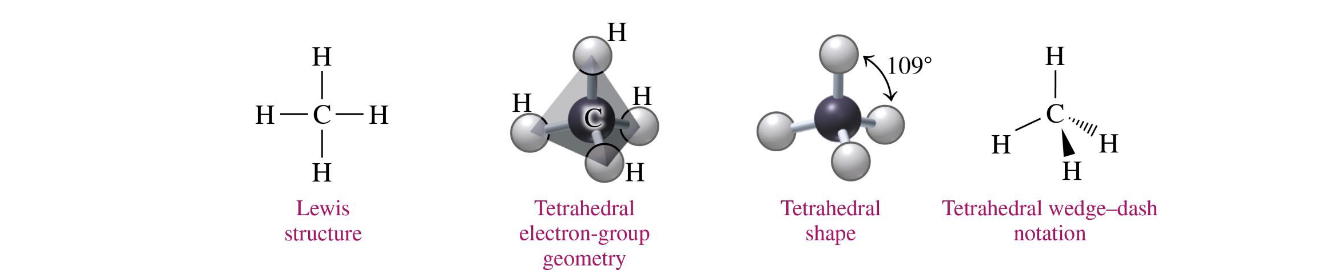
central atoms w 4 electron groups
tetrahedral 109
central atoms w four electron groups (1 lp)
trigonal pyramidal 109
NH3
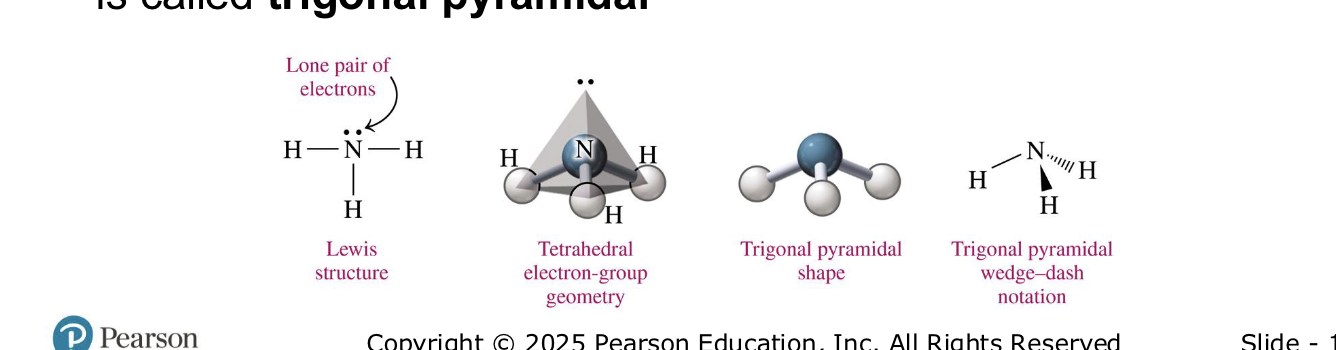
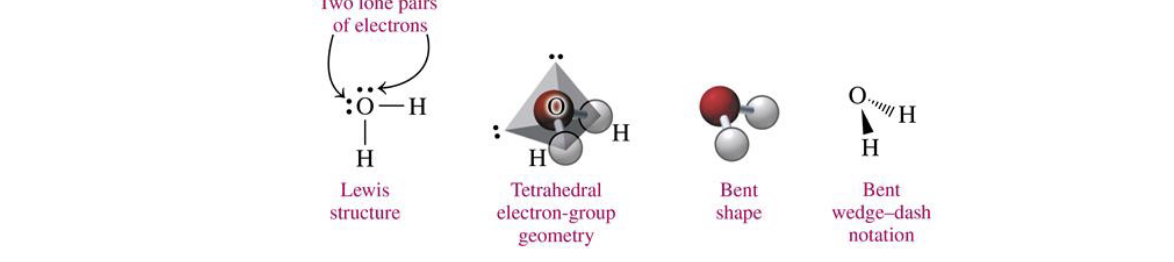
central atom w 4 electron groups (2 lp)
bent 109
H2O
electronegativty
the measure of an atom's ability to attract shared electrons in a chemical bond
electronegativity trends
Across a Period (Left to Right): Electronegativity increases. (The atoms get better at pulling electrons).
Down a Group (Top to Bottom): Electronegativity decreases. (The atoms get larger, and the nucleus is too far away to "pull" effectively).
most= F, least Fr
VESPR - valence shell electron-pair repulsion (V SEPR) theory
describe oreintation of electron groups around central atom
states that electron groups are arranged as far apart as possible around central atom
specific shape of molecule is determined by the number of atoms attached to central atom