Chapter 3 - Amino acids, Peptides, And Proteins
1/19
Earn XP
Description and Tags
This is based off lehnigers biochemistry
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
20 Terms
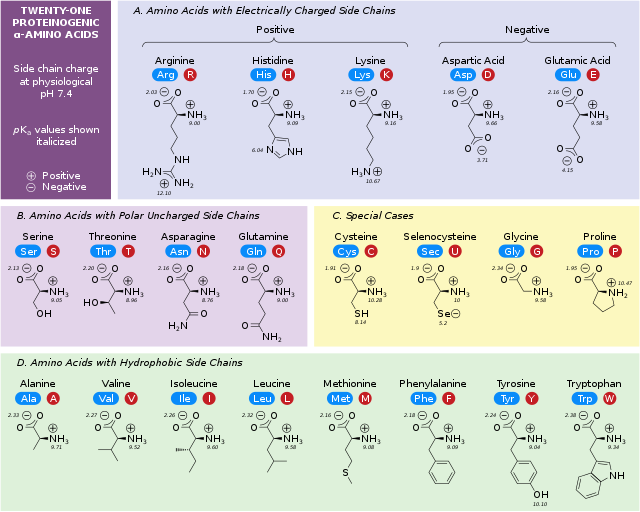
List all 20 Amino acids
- Glycine. - Serine
- Alanine - Threonine
- Proline - Cysteine
- Valine - Asparagine
- Leucine - Glutamine
- Isoleucine - Phenylalanine
- Methionine - Tyrosine
- Tryptophan - Lysine
- Arginine - Histidine
- Aspartate - Glutamate
What are the 4 principles of proteins and amino acids?
1) All living organisms are made from the 20 amino acids
2) Amino caids are joined together by a link called a “peptide bond'“
3) Proteins can be seperated from other proteins based on their chemical/functional properties coming from their amino acid sequences
4) Amino acid sequences are a key gate way into understanding functional and evolutionary relationships
Define Amino acid residue
An amino acid residue is the portion of an amino acid that remains in a polypeptide after losing H and OH during peptide bond formation.
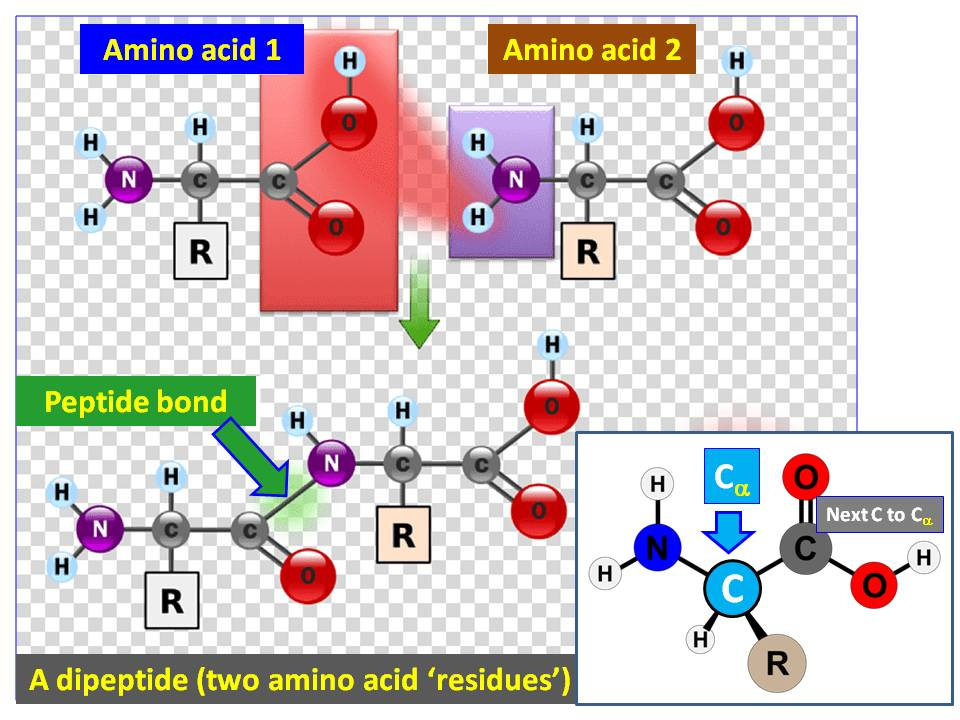
What do all 20 amino acids have
Carboxyl group(functional group in organic chemistry, denoted by the formula (-{COOH})
An Amino group bonded to the Alpha Carbon( Amino group is a group which consist of nitrogen bonded with 2 hydrogens -NH2)
An R group( this is how they differ)
how does the R group affect the amino acid formation
the structure of the amino acid
The size
The electric charge
The solubility of amino acids
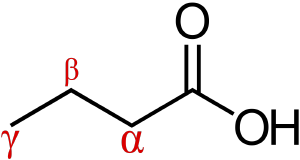
for all amino acids but Glycine, what is the aphla carbon bonded to
Carboxyl group
Amino group
R group
Hydrogen atom
it is different in Glycine since its R group is another Hydrogen atom
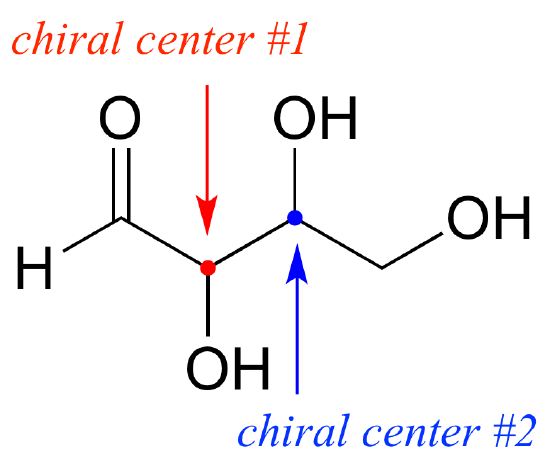
define “Chiral center”
it is a tetrahedral atom, which is bonded to 4 different atoms or groups of atoms
it acts a Stereocenter( which means its arrangement of the different atoms or groups make the molecule non-superimposable on its image mirrion
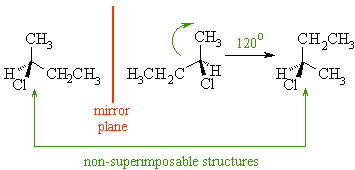
define “ non superimposable”
It means that although molecules/objects can be mirror images of each other they cannot be placed on top of each other to align all the way
define “stereoisomers”
molecules which have the same molecular formula but have a different 3d shape
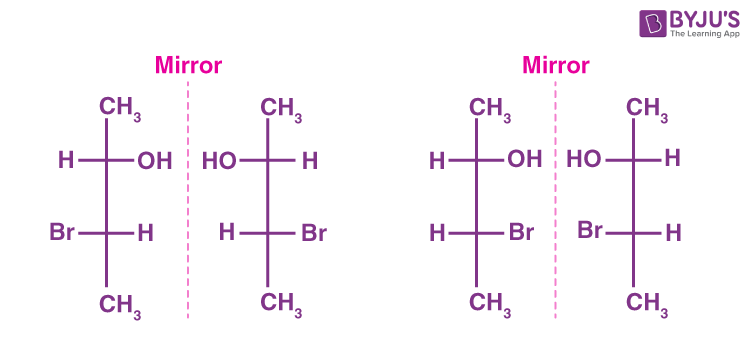
Define “enantiomers”
Pairs of chiral molcules which are non superimposable images of eachother
define “optically active”
these are compounds which have the ability to roate the plane of plane polarized light because of there molecule chirality(asymmertry
define “moleculechirality
it is proptery which a molucule is non superimpoable on the mirror image, which leads to a left and right handed form(these are called enantiomers)
define “absoule confirguration
this is the actual 3d shape of atoms around a chiral center in a molecule
D,L system
the absoule config for simple sugars and aminos are given by this system
this system is based on the 3 carbon sugar glyceralhehyde
for all chiral compounds, if they have a config related to L-glyceraldehyde then they have the desingation of L-
the stereoismoers which related to the R-glyceraldehyde config they are disgnated R
what does absoule config refer to
it refers to the four bonded atoms/compounds atound the chiral
define “ RS system’
a more pricec way to see the confiruration of moleules with more than 1 chiral group
what percent of amino acids are found with a d-config and why is this?
<1%
this is because they tend to have structural importance in some bioglical aspect and because they are introduced by a protein by enzymes catalyzed reaction which happen after ribosomes
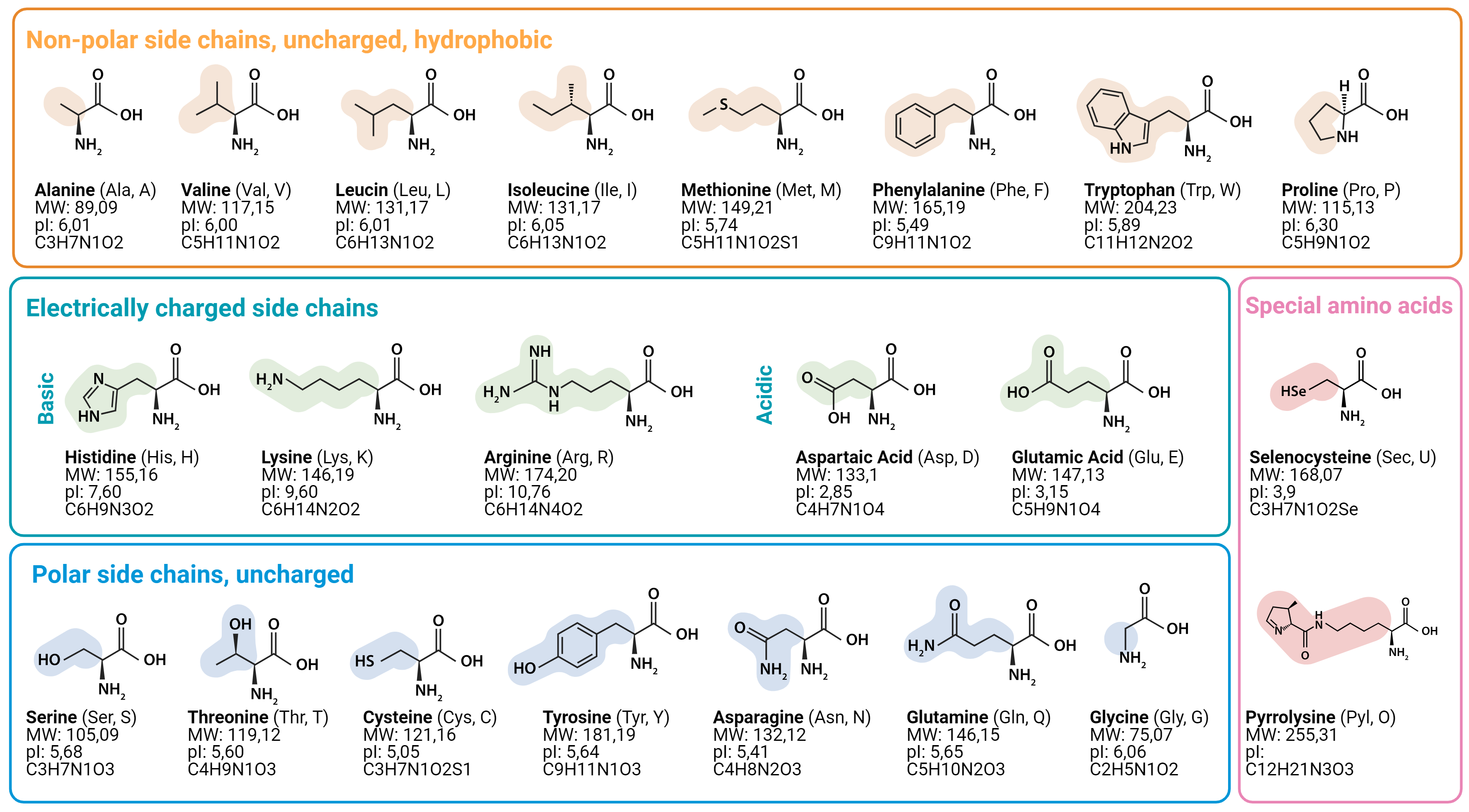
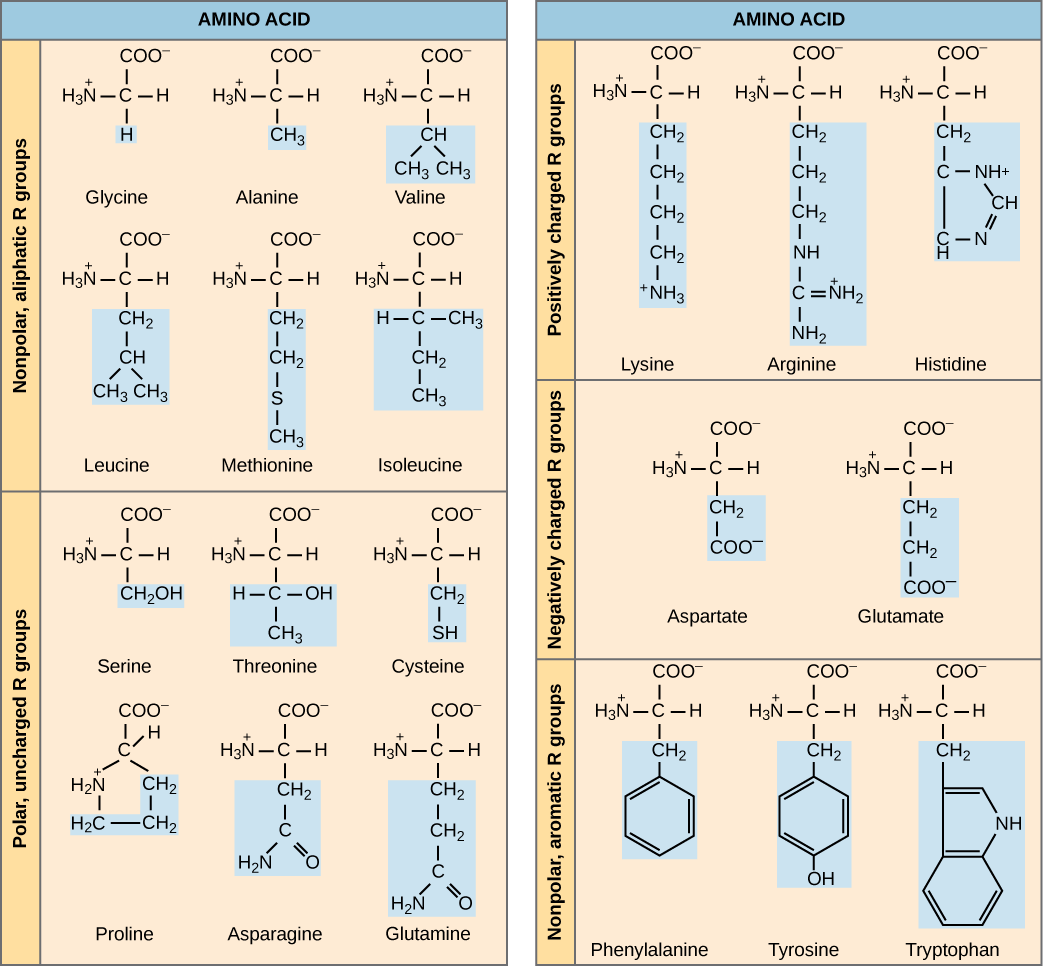
what are the 5 main classes of group of amino acids
non polar, aliphatic R groups
Aromatic R groups
Polar, uncharged R groups
Positively charged R groups
Negatively R groups
define “ polarity”
this is the tendency to interact with water at a biological ph(7)
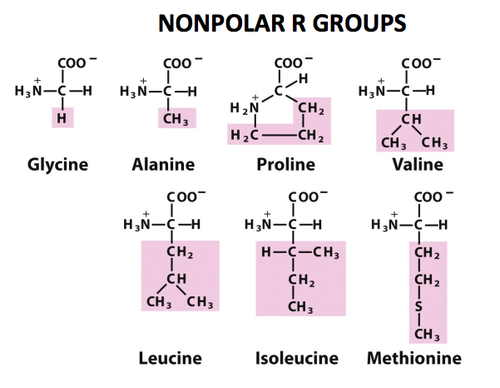
what are the amino acids in the group “ nonpolar, aliphatic R groups” and what is the charateristics of these
there R groups are hydrophobic and nonpolar
the side chains of alaine, valine, leucine, isoleucine tend to cluster all together in the formation of proteins
proline has a “aliphatic side chain”( it is a side chain which is non aromatic and is composed of carbon and hydrogen which is attached to the backbone of molecules), its 2nd amino group, imino, is proline rsodures. which is held in a rigid form which reduces the strucutres flexability
the amino acids present in the group are:
Glycine: Gly, G
Alanine: Ala, A
Proline: Pro, P
Valine: Val, V
Leucine: Leu, L
Isoleucine: Ile, I
Methionine: Met, M