C5 - Energy Changes
1/48
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
49 Terms
Where is energy stored?
Chemical bonds
What is an exothermic reaction?
A reaction where energy is transferred to its surroundings
If an exothermic reaction occurs in a closed container, what would happen to the temperature inside the container?
Temperature increases
What is an endothermic reaction?
A reaction that takes in energy from its surroundings
If an endothermic reaction occurs in a closed container, what would happen to the temperature inside the container?
Temperature decreases
What type of reaction does this image show?
Endothermic
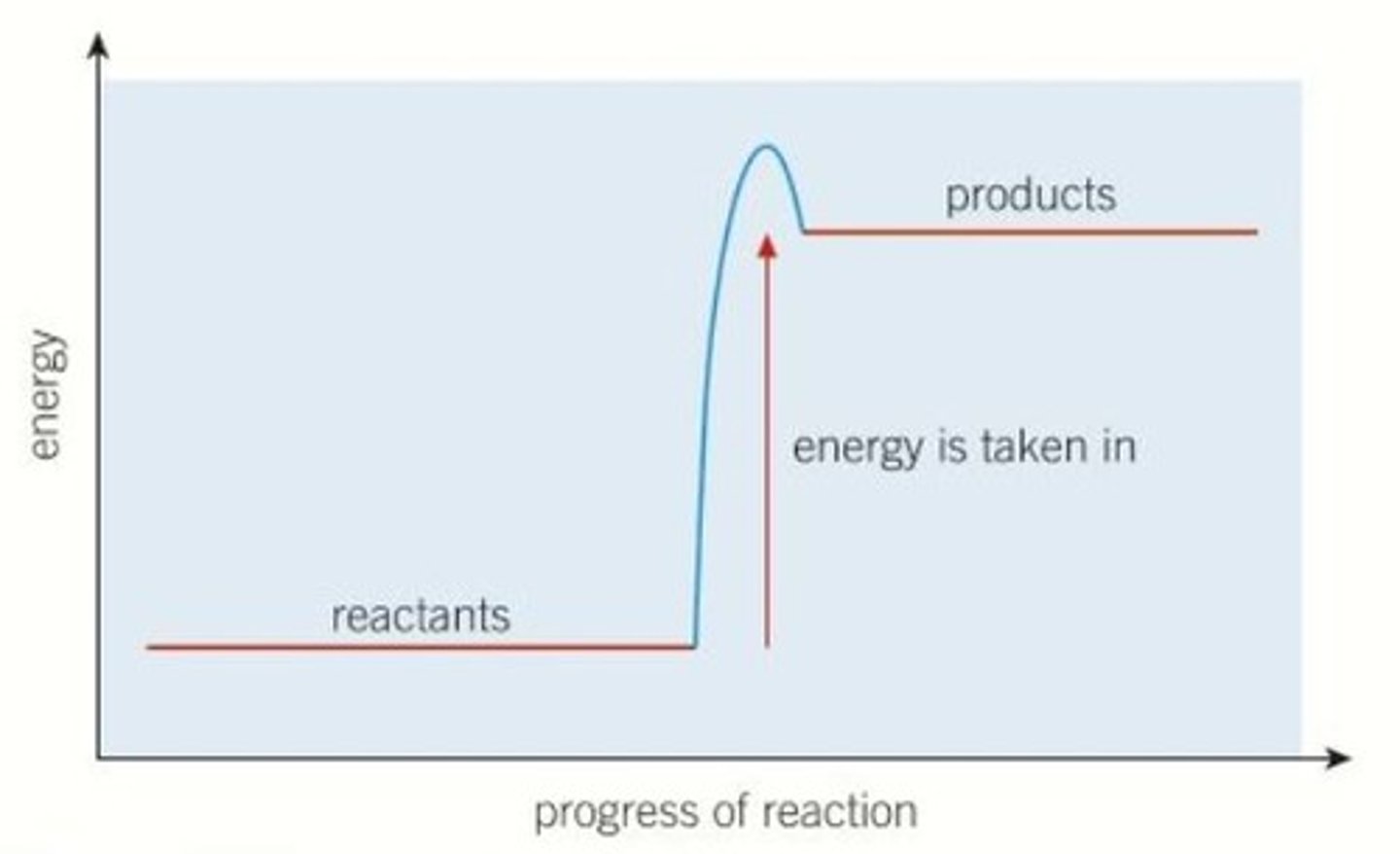
What type of reaction does this image show?
Exothermic
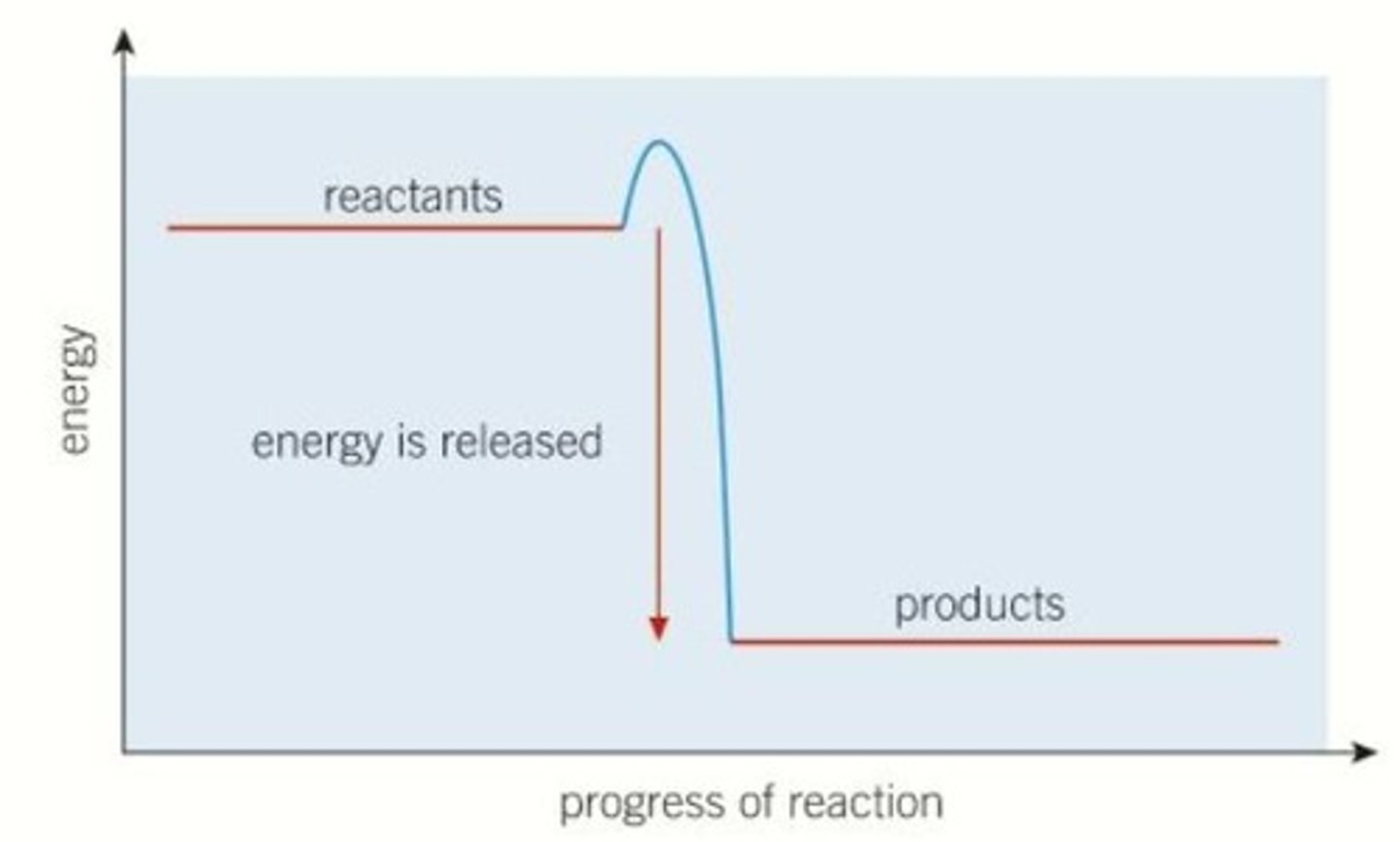
Define activation energy
The minimum amount of energy the reactant particles need in order to collide with each other and react
Does breaking bonds release energy or require energy?
Requires energy
Is breaking bonds endothermic or exothermic?
Endothermic
Define bond energy
The energy required to break one mole of a particular covalent bond
During a chemical reaction, the bonds of the products must be:
formed
Name three reactions that are exothermic
Combustion, neutralisation and oxidation
Name a common endothermic reaction
Thermal decomposition
Which part of a reaction profile shows the activation energy?
reactants to the peak of the curve
How do chemical cells transfer energy to their surroundings?
By electricty
Are alkaline batteries rechargeable or non-rechargeable?
Non rechargeable
Fuel cells convert the chemical energy in a fuel and oxygen into:
electricity
Which direction do the electrons flow in a hydrogen-oxygen fuel cell?
Anode to cathode
What is the overall reaction for a hydrogen oxygen fuel cell?
hydrogen + oxygen -> water
Which compartment does the oxygen enter in a hydrogen-oxygen fuel cell?
The cathode compartment
What enters the anode compartment in a hydrogen-oxygen fuel cell?
Hydrogen
What type of reaction would a negative overall energy be?
Exothermic
What would a positive value mean in a bond energy calculation final answer?
Endothermic
What type of reaction is breaking bonds?
Endothermic
What type of reaction is forming bonds?
Exothermic
What do bonds require to break?
Energy
What factors affect the voltage of a cell?
Difference in reactivity between metals for electrodes, concentration and type of electrolyte, temperature of the electrolyte
What are the usual external sources of fuel used in a fuel cell?
Hydrogen and oxygen
What is oxidised in a fuel cell?
Hydrogen
What is reduced in a fuel cell?
Oxygen
Name a disadvantage of hydrogen fuels cells
Hydrogen hard to be obtained,
Hydrogen highly flammable and explosive,
Hydrogen hard to store and transport (gas at room temp)
Name an advantage of hydrogen fuel cells
Reaction does not produce toxic pollution,
Stores more energy than a rechargeable battery
What do the electrons that have left the oxygen travel to the cathode through?
The wire
What equation happens at the anode (oxidation) in a fuel cell?
H2 -> 2H+ + 2e-
What equation happens at the cathode in a fuel cell?
O2 + 4H+ + 4e- -> 2H2O
Is breaking bonds exothermic or endothermic?
endothermic
Is forming a bond an exothermic or endothermic process?
exothermic
What is the overall equation for a hydrogen fuel cell?
2H2 + O2 -> 2H2O
What is formed at the cathode in a hydrogen fuel cell?
water
Why do alkaline batteries eventually stop working?
A reactant is used up
Which direction do the electrons flow in a hydrogen-oxygen fuel cell?
negative anode to positive cathode
What are the electrodes in fuel cells made from?
porous carbon
True or false: The oxygen enters the cathode compartment and the hydrogen enters the anode compartment
true
What are the main advantages of hydrogen-oxygen fuel cells?
Hydrogen is a gas at room temperature meaning it is difficult and dangerous to store
What is the half equation at the anode of a fuel cell?
H2 -> 2H+ + 2e-
What compartment does hydrogen enter in a fuel cell?
anode compartment
What compartment does oxygen enter in a fuel cell?
cathode compartment
What is the half equation at the cathode of a fuel cell?
O2 + 4H+ + 4e- -> 2H2O