AP Bio Unit 2: Chemistry of Life
1/53
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
54 Terms
Biological Macromolecules
Proteins, Carbohydrates, Lipids, & Nucleic Acids (all made up of CHONP)
Monomers
Single unit

Polymers
Many units (monomers) bound together

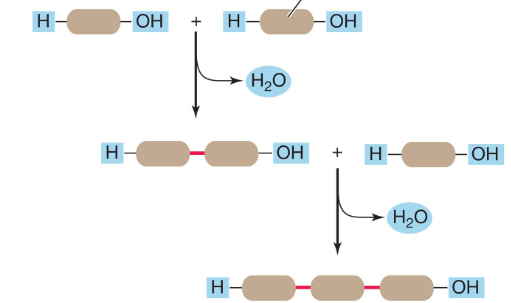
Dehydration Synthesis
How cells build large molecules (polymers) from smaller ones (monomers).
An enzyme removes a hydroxyl group from one molecules and a hydrogen atom from another. A covalent bond forms between the molecules, and water forms.
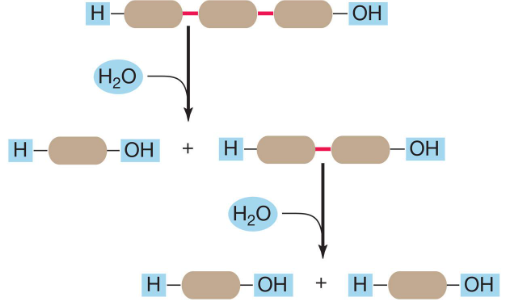
Hydrolysis
How cells breaks large molecules (polymers) into smaller ones (monomers).
An enzyme attaches a hydroxyl group and a hydrogen atom (both from the addition of water) at the cleavage site.
CHONP
Carbon, Hydrogen, Oxygen, Nitrogen, & Phosphorous
The building blocks of life as they make up all of the biological macromolecules.
Carbon’s Importance
Carbon is important to life because of its versatile bonding behavior. Each carbon atom can form up to four covalent bonds, can be polar or nonpolar, can form carbon rings/chains, and can be assembled and remodeled into a variety of organic compounds.
Functional Groups
Clusters of atoms on a molecule that gives the molecular polarity or acidity. They help determine how that molecule will behave.
Hydroxyl Group
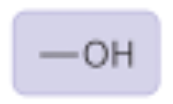
Properties: Polar, forms hydrogen bonds, soluble
Found in: sugars and alcohols (ex: ethanol)
Methyl Group
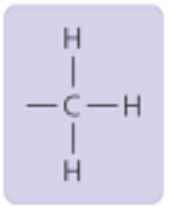
Properties: Non-polar, insoluble
Found in: fatty acid chains (ex: 5-methanyl cytosine)
Carbonyl Group
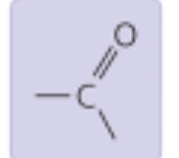
Properties: Polar, can form hydrogen bonds
Found in: Carbohydrates, some amino acids, fatty acids (ex: acetone)
Carboxyl Group
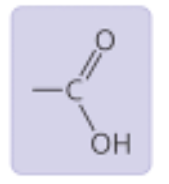
Properties: Acidic, can form hydrogen bonds
Found in: Amino acids, fatty acids (ex: acetic acid)
Amino Group
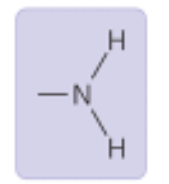
Properties: basic, can form hydrogen bonds
Found in: Amino acids, proteins (ex: glycine)
Sulfhydryl Group
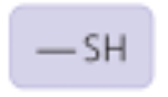
Properties: Can form disulfide bonds
Found in: Some amino acids (ex: cysteine)
Phosphate Group
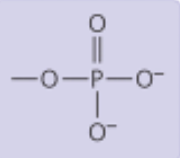
Properties: Negatively charged, polar
Found in: Nucleic acids (ex: glycerol phosphate)
Monosaccharide
Simple ring sugars (monomers) that are mostly used as an energy source or structure. They are very soluble in water and may be in a linear or ring form
ex: glucose, fructose, galactose
Disaccharide
Two monosaccharides combined (oligosaccharides/short polymers)
ex: sucrose, lactose, maltose
Polysaccharide
Polymers of monosaccharides (complex carbs)
ex: cellulose, chitin, starch, glycogen
Carbohydrates
Consists of carbon, hydrogen, and oxygen in a 1:2:1 ratio (CHO). They can be used for energy, structure, and for storing/transporting energy.
ex: glucose, starch, etc
Lipids
Function as long term energy storage and is the structural foundation of cell membranes. They can be fatty, oily, or waxy and are insoluble in water (non-polar, hydrophobic). They are composed of CHO.
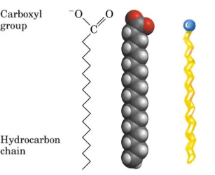
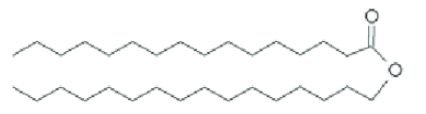
Fatty Acids
Simple organic compounds with a carboxyl group joined to a backbone of 4-36 carbon atoms. They have hydrocarbon, hydrophobic tails, and carboxyl group, hydrophilic heads.
Identifying them: they have long zig-zag chains or fused rings with few oxygens
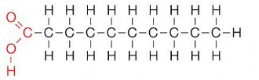
Saturated Fatty Acids
Has single bonds within their hydrocarbon tails. They are “fully saturated” with hydrogen atoms, have flexible tails, and can “stack up” at room temperature, forming a soft solid.
ex: butter
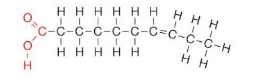
Unsaturated Fatty Acids
They contain 1 or more double bonds in the carbon chain. This makes their hydrocarbon tail slightly bent, making them less flexible and unable to stack together (liquid at room temperature)
ex: vegetable oils
Fats
Lipids with 1-3 fatty acids bonded to glycerol (an alcohol). A fatty acid can easily bond with other molecules, like glycerol, via the carboxyl group (which loses its hydrophilic properties)
ex: triglycerides, phospholipids
Phospholipids
A part of the phospholipid bilayer and is composed of a glycerol backbone, 2 fatty acid tails, and a phosphate group. They also are amphipathic, meaning they have a hydrophilic head and hydrophobic tails.
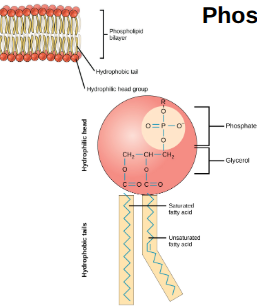
Phospholipid Bilayer Function
Provides selective permeability (controls what enters/exits cell), fluidity (allows molecules to move within membrane), & structural integrity (maintains cell’s shape and protects it)
Wax
Complex molecules with a varied mixture of lipids with long fatty acid tails bonded to long-chain alcohols or carbon rings. Wax is firm and water-resistant, allowing it to serve many purposes (ex: wax helps protect leaves, human hair, bird feathers, etc)
Steroids
Lipids with a rigid backbone of four carbon rings and no fatty acid tails
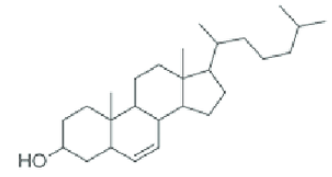
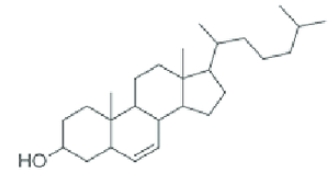
Cholesterol
The most common type of steroid in animal cells membranes (helps maintain cell structure). They don’t need a cell wall, they can change shape, and they can move freely.
Proteins
Used in all cellular processes such as structure, nutrition, enzymes, transport, communication, & cellular defense
Amino Acids
The monomer of proteins and are joined by peptide bonds. Thousands of different proteins can be made by arranging amino acids in different ways.
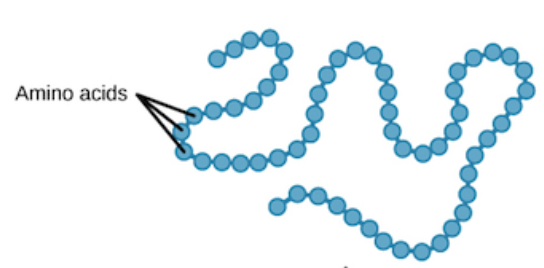
Polypeptide
A linear chain of amino acids
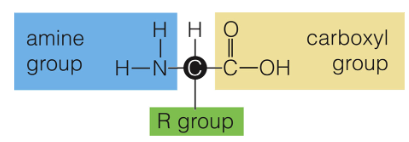
Amino Acid Structure
All amino acids have an amine group, carboxyl group, and an R-group. They are made up of CHON elements.
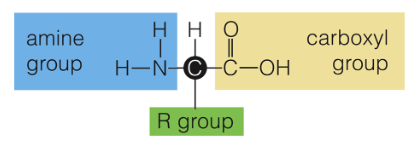
R-Group
a.k.a the Variable Group, and gives the amino acids its properties (hydrophobic, acidic, etc)
The 4 Protein Structures
Primary, Secondary, Tertiary, & Quaternary
Primary Protein Structure
Sequence of amino acids held by peptide bonds
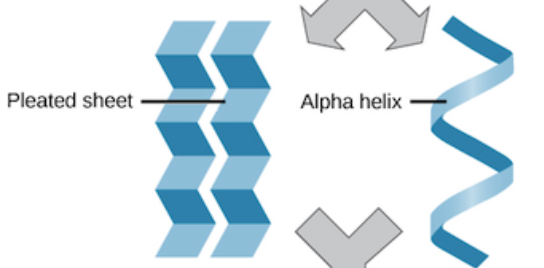
Secondary Protein Structure
Local folding which forms hydrogen bonds between the amino acids, which makes two kinds of shapes → α-helix & β-sheet (hydrogen bonds)
Tertiary Protein Structure
3D shape of one chain formed, which depends on interactions between R-groups
Quaternary Protein Structure
Functional protein formed by two or more folded polypeptide chains in close association or covalently bonded together

Denature
When a protein loses its shape and no longer functions correctly
Nucleic Acids
Repeating units of nucleotides (monomers) make up DNA (polymer, nucleic acid)
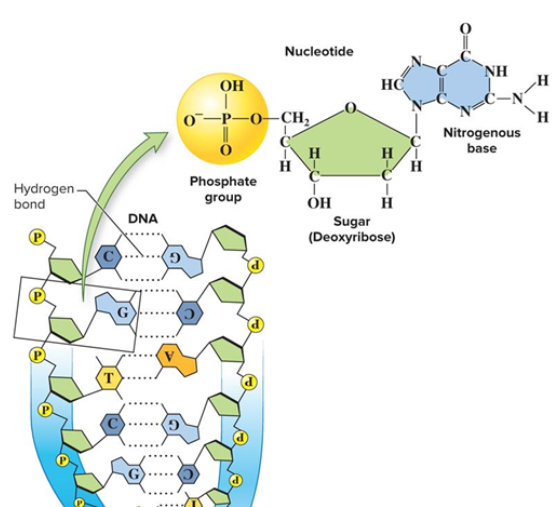
Nucleotide Structure
Have three parts: negatively charged phosphate groups, 5-carbon deoxyribose sugars, and nitrogen bases (A, T, C, G).
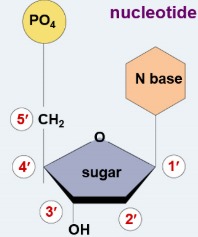
5-carbon Deoxyribose
Deoxyribose has 5 Carbon atoms, which are numbered clockwise
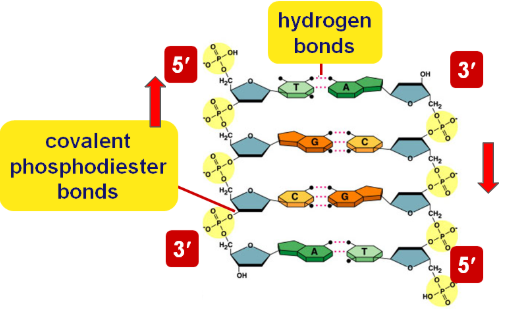
DNA Directionality
DNA is antiparallel because the complementary strands run in opposite directions
(5’ → 3’ & 3’ → 5’)
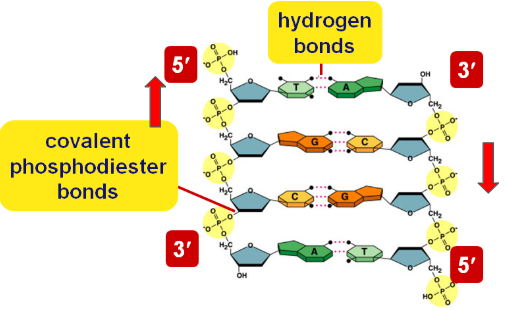
Bonds in DNA
Hydrogen bonds hold bases together (pyrimidines with purines) and covalent phosphodiester bonds hold nucleotides together at the backbone.
Pyrimidines
T, C, with single rings
Remember: CUT the Py
Purines
A, G, with double rings
Remember: PUR As Gold
CHONP Elements in Carbohydrates
CHO (1:2:1)
CHONP Elements in Nucleic Acids
CHONP
CHONP Elements in Lipids
CHO
CHONP Elements in Proteins
CHON
Intramolecular Forces
Hold atoms together within a molecule
Intermolecular Forces
Forces between separate molecules
Organic Compounds
Molecules that contain carbon atoms covalently bonded to other elements (H, O, N). They form the basis of all living organisms