Biochemistry Introduction
1/105
Earn XP
Description and Tags
Flashcards created for key vocabulary and concepts in biochemistry based on lecture notes.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
106 Terms
Biochemistry
The study of chemical processes within and relating to living organisms.
Endocytosis
Transport of substances into a cell.
Exocytosis
Transport of substances out of a cell.
Phototrophs
Organisms that obtain energy from light.
Chemotrophs
Organisms that obtain energy from chemical compounds.
Aerobic
Processes that require oxygen.
Anaerobic
Processes that do not require oxygen.
Cellular Foundations
Basic building blocks of life, including structures like plasma membrane, cytoplasm, and organelles.
Primary Alcohol
An alcohol in which the hydroxyl group is attached to a primary carbon.
Secondary Alcohol
An alcohol in which the hydroxyl group is attached to a secondary carbon.
Tertiary Alcohol
An alcohol in which the hydroxyl group is attached to a tertiary carbon.
Hydrocarbons
Compounds consisting only of hydrogen and carbon.
Functional Groups
Specific groups of atoms within molecules that are responsible for the characteristic chemical reactions of those molecules.
Structural Isomers
Compounds with the same chemical formula, but different order of attachment
Stereoisomers
Compounds with the same formula and same order of attachment, but different arrangements of atoms in space.
Geometric Isomers
Stereoisomers that differ in the spatial arrangement of groups around a double bond.
Cis Isomers
Isomers where similar groups are on the same side of a double bond.
Trans Isomers
Isomers where similar groups are on opposite sides of a double bond.
Epimers
A type of diastereomer that differs at only one specific stereogenic center.
Redox Reactions
Reactions involving the transfer of electrons between two species.
Dehydration
The removal of water. H2O is a product
Hydration
Addition of water across a double bond. H2O is a reactant
Hydrolysis
A chemical reaction in which water breaks down another compound.
Methylation
Addition of a Methyl (CH3) group
Phosphorylation
The addition of a phosphate group to a molecule.
Endergonic Reactions
Reactions that require energy to proceed.
Exergonic Reactions
Reactions that release energy.
Metabolic Pathways
Series of chemical reactions occurring within a cell.
Catabolic Pathways
Pathways that break down molecules to produce energy.
Anabolic Pathways
Pathways that construct molecules from smaller units.
In Vitro
Experiments performed in glass
In Vivo
Experiments done in whole, living organisms
Chemical foundations of life
99% of mass of cell is made of hydrogen, oxygen, nitrogen, and carbon
Alkanes
Carbon-Carbon single bond
Alkenes
Carbon-Carbon double bond
Alkynes
Carbon-Carbon triple bonds
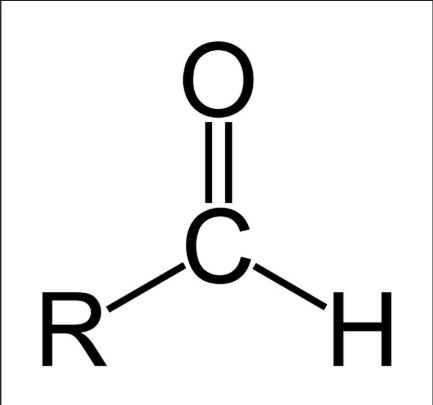
Aldehyde
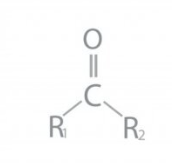
Ketone
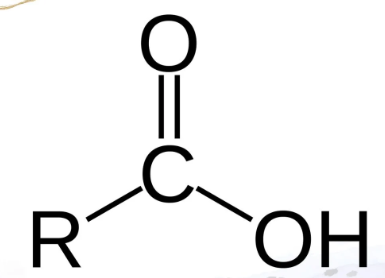
Carboxylic Acid
Ester
Acid + Alcohol
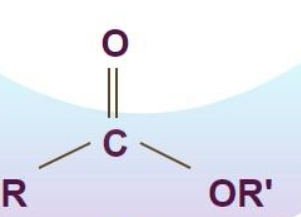
Ether
Two alcohols
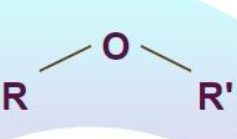
Amine
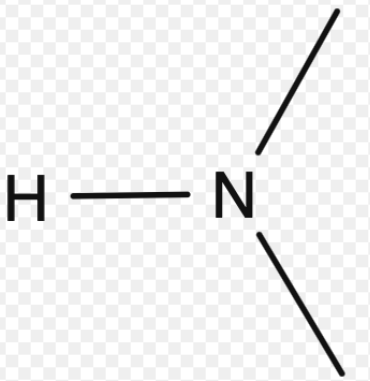
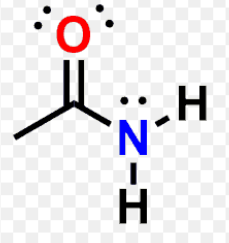
Amide
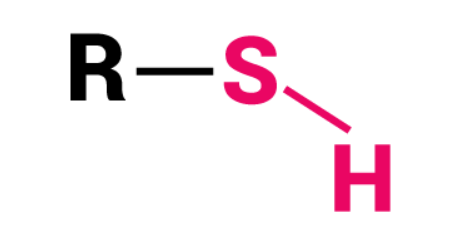
Thiol
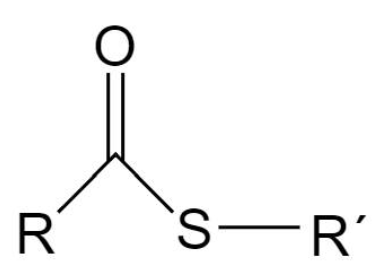
Thioester
Homeostasis
Maintenance of condition in the body necessary to maintain life within the body’s internal environment
Dynamic steady state
Rate of intake or synthesis occurs at the same rate that it is being used up
Equilibrium
Reaction where the forward and reverse reaction are occurring at the same rate resulting in no net change in amounts of reactants and products
Water
the most abundant substance in living systems
Nonpolar covalent bonds
Between 2 nonmetals, electronegativity of 0-0.4
Polar covalent bonds
Between metals and nonmetals, electronegativity of 0.41-1.8
Ionic bonds
electrons transferred from metal to nonmetal, electronegativity of 1.8 or above
easily
Polar molecules dissolve blank in water
Cannot readily
Polar molecules blank dissolve in water
Polar covalent bonds
Bond found in water
Polar bonds
can dissolve in water, like dissolves like
Nonpolar bonds
cannot dissolve in water
Hydrogen donor and hydrogen acceptor
what is required for hydrogen bonds to occur
hydrogen donor
These can be a hydroxyl or amine
hydrogen acceptor
these are usually O or N
hydrogen bonds
weak attraction between hydrogen atom and a highly electronegative atom in another
Electrostatic interactions
forces between cation and anions or dipoles
London dispersion forces
The weakest of all attractive forces; caused by short lived dipole moments
Dipole-Dipole interaction
an attraction between two polar molecules, positive and negative dipoles
Ion-dipole
ions attract or repel dipoles
hydrophobic interactions
molecules that cluster together in a polar solution
passive diffusion
movement of a molecule from high to low concentration gradient
facilitated diffusion
movement of a molecule from high to low concentration with the help of a transport protein
osmosis
diffusion of water molecules from high to low water concentration
hypotonic
solutions with high water and low solute concentration
lysis
water flows into cells and cause them to burst
hypertonic
solutions with low water and high solute concentration
cremation
water flows out of cell and causes it to shrink
isotonic
equal solute concentration, water flows in and out at the same rate
lower ph
an increase in H+ causes
higher ph
decrease in H+ causes
H+ is still attached
The ph is below the PK
H+ is lost
The ph is above the PK
Biochemical acids
substances that can donate H+ to lower the ph
Biochemical Bases
Substances that can accept H+ to increase ph
Acidosis
Accumulation of acid or loss of base
Alkalosis
Accumulation of base or loss of acid
intracellular buffers
protein buffer system and phosphate buffers are what
extracellular buffers
Bicarbonate-carbonic acid buffer system are what
Respiratory buffers
control concentration of carbon dioxide
Metabolic buffers
Controls concentration of metabolic process concentration
Respiratory Acidosis
Causes increase in breathing rate to control pH
Respiratory Alkalosis
Causes decrease in breathing rate to control pH
Metabolic Acidosis
Caused by any other mechanism other than increased co2; usually due to loss of bicarbonate or excessive nonvolatile acids
Metabolic alkalosis
Excessive bicarbonate production; can be due to fluid imbalance or other mechanisms
Cohort Studies, Randomized control studies, Systematic Reviews, Meta-analysis
Types of Study designs that are better than opinions and case studies
Absolute risk
true incidence of disease; also called risk difference
Relative risk
measure of probability of risk, only used in cohort studies, (risk ratio)
Odds ratio
measure of odds of risk, can be used to calculate risk in cohort, case-
control and meta-analyses
OR and RR is not significant
If the confidence interval crosses the referent number (1)
OR and RR is significant
If the confidence interval stays on 1 side of the referent number (1)
Absolute Risk: Interpretation
risk in control minus risk in treatment, divide by 100 to get %
OR=1
exposure is not related to disease; no association
OR>1
exposure is positively related to disease; positive association (increased
risk)
OR<1
exposure is negatively related to disease; negative association
(decreased risk)