Chemistry- Chapter 19 - Amino Acids and Proteins
1/9
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
10 Terms
Amino Acids
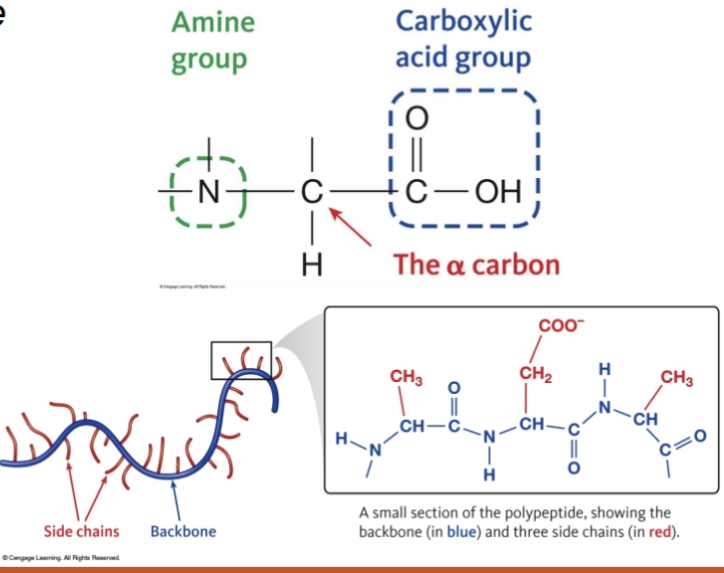
Proteins are composed of building blocks called
Primary structure
the sequence of amino acids in a polypeptide, polymers of amino acids held together by peptide bonds
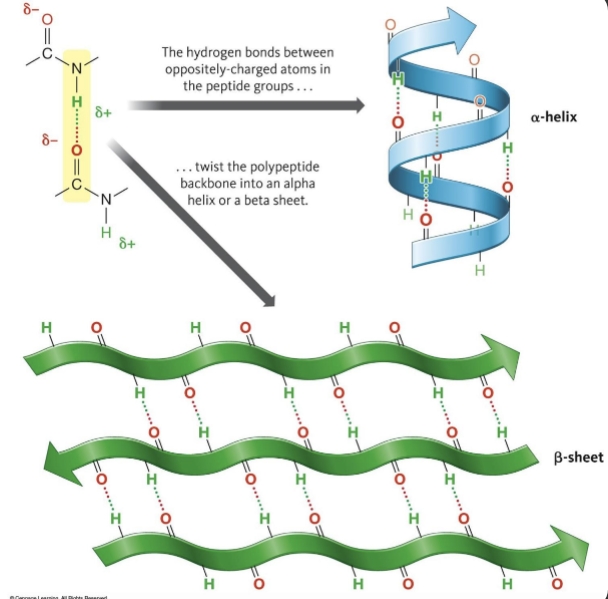
secondary structure
peptide bonds can H- bond with each other
a-helices: backbone forms a tight coil with side chains projecting outward
B-sheets: sections of backbone lined up in parallel rows
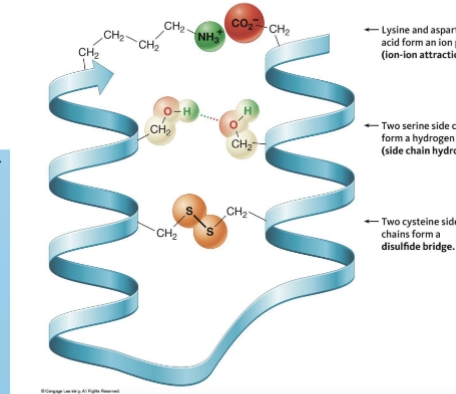
tertiary structure
shape of protein that results from all interactions involving side chains
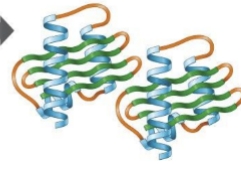
quaternary structure
some proteins contain two of more polypeptide chains that are attracted to one another, the way in which two or more polypeptides join to form active proteins
protein denaturation
any disruption of interactions in the protein that renders it inactive is known as denaturation, may unfold completely or shift into a different, non-active fold
changing the solvents
organic solvents will shift the positions of hydrophobic and hydrophilic side chains
hydrophobic: side chains will move to the exterior
hydrophilic: side chains will cluster in interior
changing the PH
the ionization of the acidic and basic side chains will change, and ion-ion attraction is disrupted
lowering the PH will protonate carboxylic acid side, increasing the pH will deprotonate the amine side chain
raising temp and violent agitation
As temperature increases, molecules have increasingly rapid
molecular motion
This motion will denature most proteins between 50°C and
70°C.
adding ionic substances
addition of high concentrations of ionic compounds will interrupt ion-ion attractions, soaps will interrupt ion-ion attractions and hydrophilic and hydrophobic interactions, heavy metal ions such as ions of lead, mercury are strongly attracted to sulfur and will disrupt disulfide bridges