Bio 201 - Building Blocks & Macromolecules
1/40
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
41 Terms
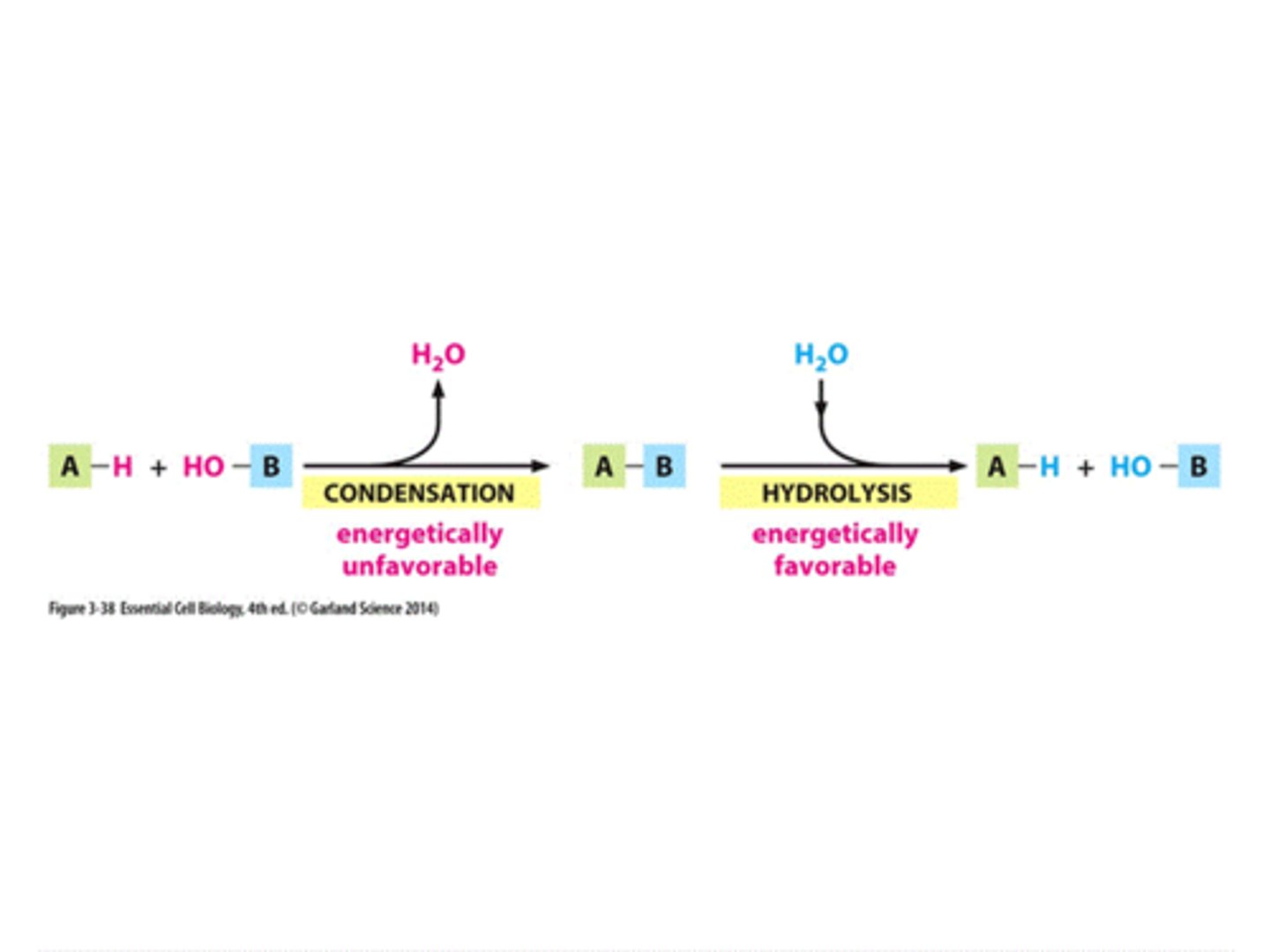
Subunit - Macromolecule: Carbohydrates
sugars - monosaccharides (glucose, fructose)
Intermediate molecule - Macromolecule: Carbohydrates
oligosaccharides
Polymer - Macromolecule: Carbohydrates
polysaccharides - glycogen (animals), starch (plants)
Covalent bond - Macromolecule: Carbohydrates
glycosidic bond
Polarity (distinct ends) - Macromolecule: Carbohydrates
polar (but not directional polarity)
Importance/Function - Macromolecule: Carbohydrates
-sources of stored energy
-can transport chemical energy (energy in covalent bonds)
-carbon skeletons for many other molecules (some amino acids are synthesized from carbon skeletons derived from glucose)
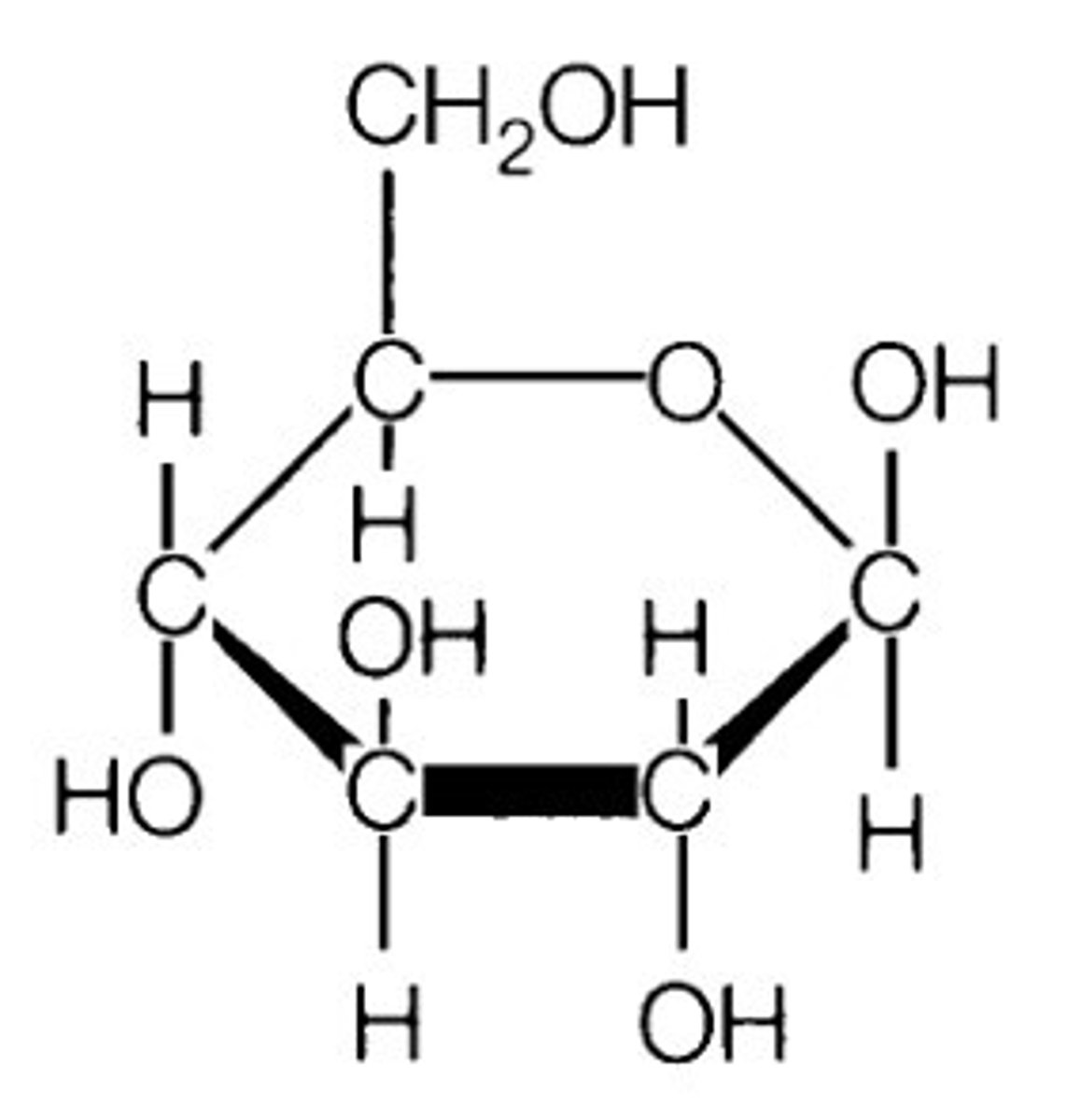
Structure - Macromolecule: Carbohydrates
-contains, C, H, and O
-formula (of monomers): (C₁H₂O₁)ₙ *where n can be 3, 4, 5, or 6
-take on a ring structure in aqueous solution
-glucose: C₆H₁₂O₆ (a subunit of both glycogen and starch)
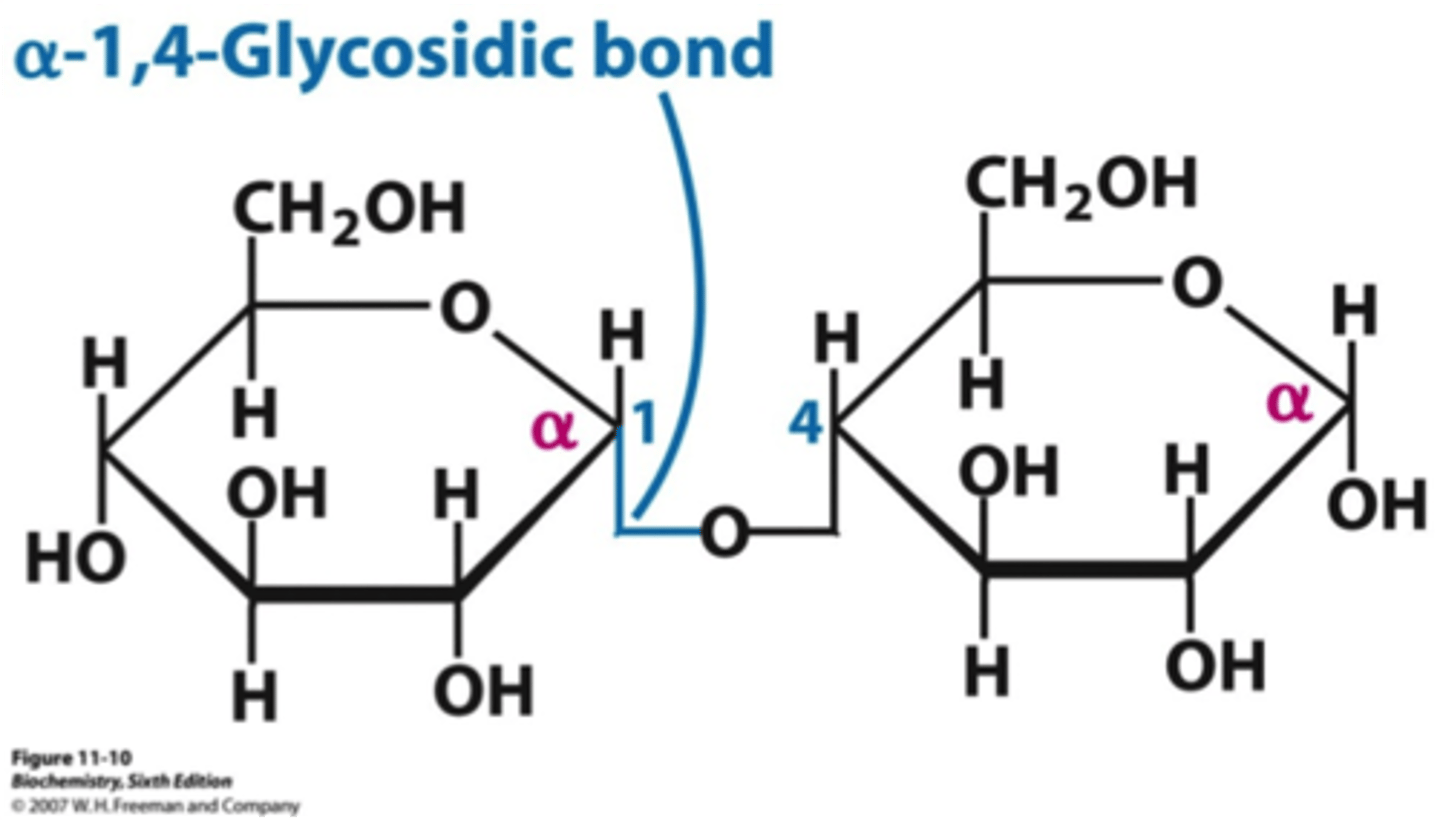
Polymerization of Sugars (Monosaccharides) - Macromolecule: Carbohydrates
-monosaccharides can combine (glycosidic bond) to form disaccharides through condensation reactions -> water is lost
-polymerization can continue to form oligosaccharides and polysaccharides
-reaction is reversible -> hydrolysis reactions break glycosidic bonds and use water
Subunit - Macromolecule: Proteins
amino acids
Intermediate molecule - Macromolecule: Proteins
peptides
Polymer - Macromolecule: Proteins
proteins
Covalent Bond - Macromolecule: Proteins
peptide linkages/bonds
Polarity (distinct ends) - Macromolecule: Proteins
directional polarity (N vs C terminus)
Importance/Function - Macromolecule: Proteins
muscle contraction
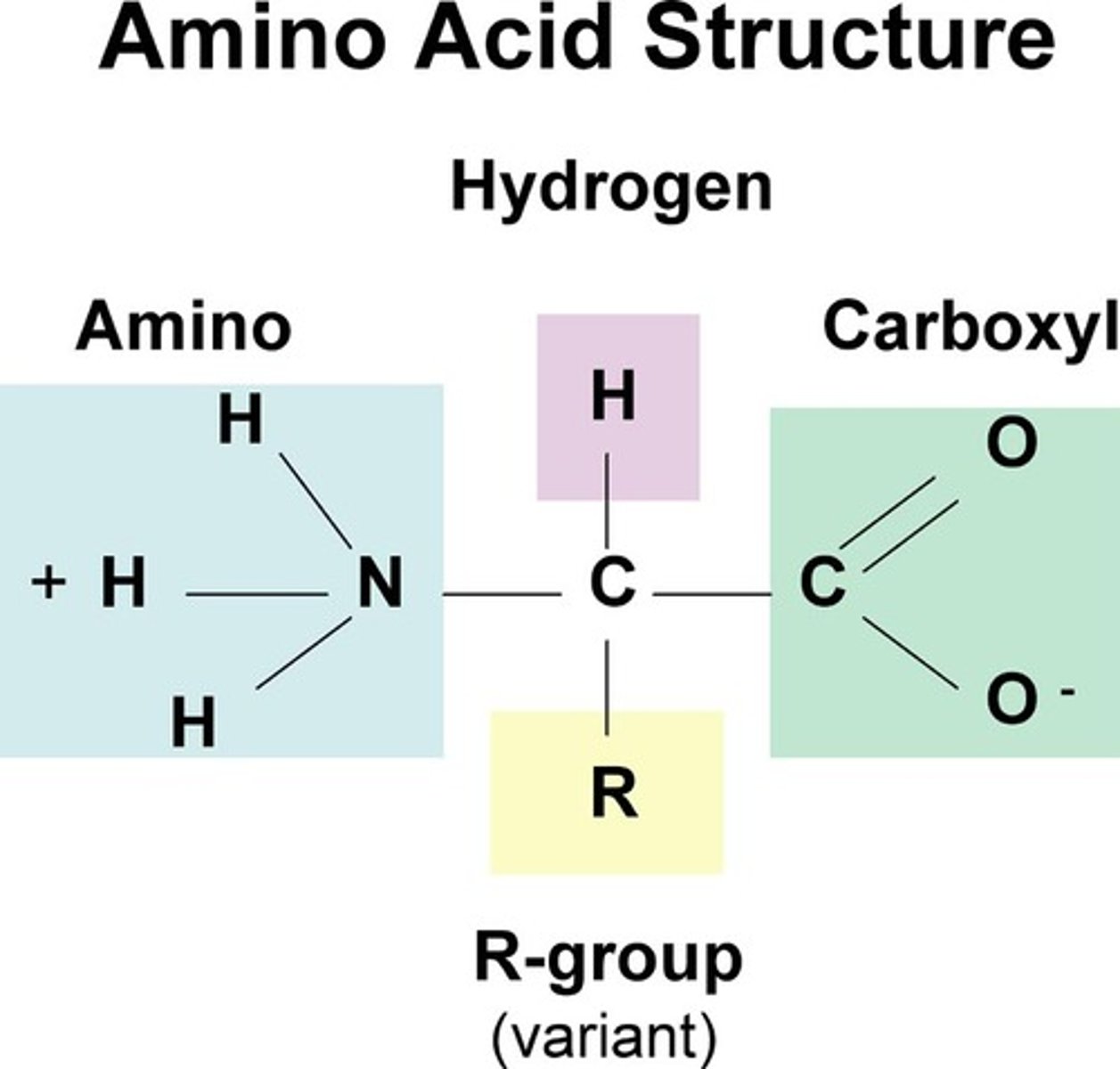
Structure - Macromolecule: Proteins
-5 components:
1) Central Carbon
2) Amino group (H₃N⁺)
3) Carboxyl group (COO⁻)
4) Single Hydrogen
5) "R" group (makes the amino acids differ, 20 "R" groups -> 20 different amino acids, attached to the Central Carbon)
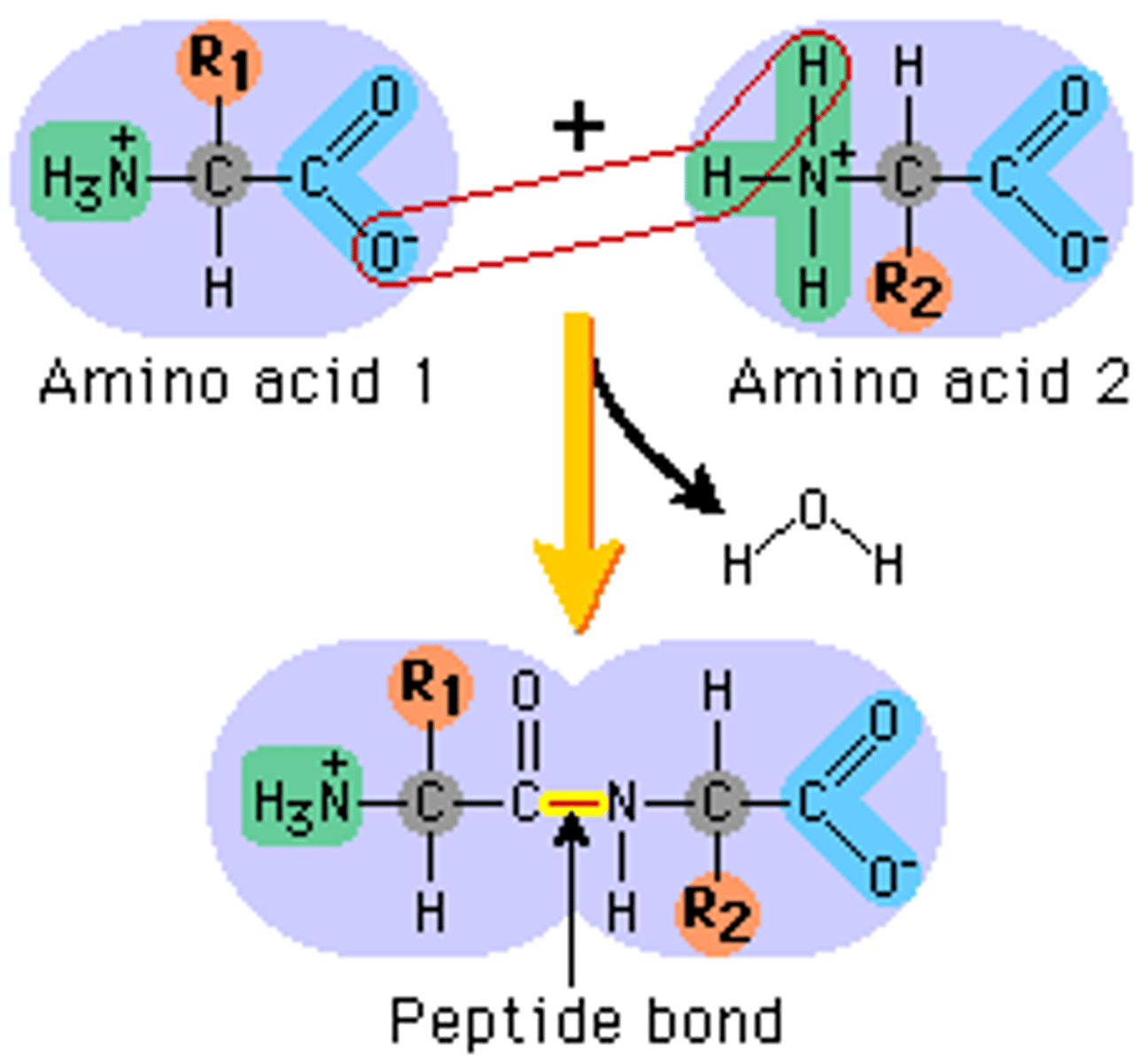
Polymerization of Amino Acids - Macromolecule: Proteins
↳ amino acids join together through the formation of peptide linkages/bonds that links the C (from the carboxyl group) and N (from the amino group)
↳ resulting polymer is read from N terminus -> C terminus
↳ when a peptide bond is broken, the monomers to the right of the bond are released (dimers, etc.)
Polypeptides (proteins) extra notes
↳ the # of "R" groups determines the # of
amino acids
↳covalent bonds surrounding the Central Carbon are flexible, allowing the chain to twist
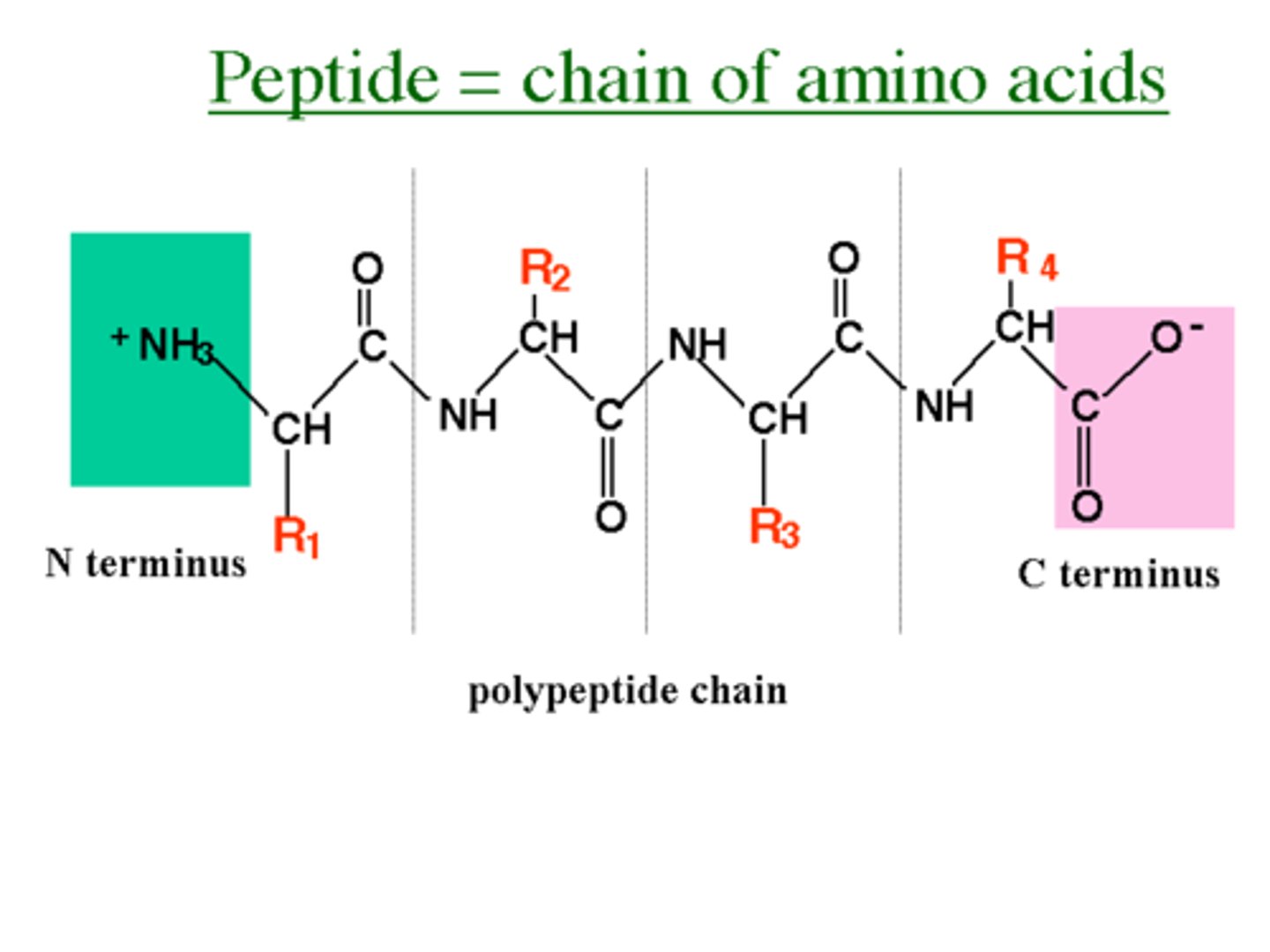
Subunit - Macromolecule: Nucleic Acids
nucleotides
Intermediate Molecule - Macromolecule: Nucleic Acids
oligonucleotides
Polymer - Macromolecule: Nucleic Acids
DNA & RNA (nucleic acids)
Covalent Bond - Macromolecule: Nucleic Acids
phosphodiester bonds
Polarity (distinct ends) - Macromolecule: Nucleic Acids
directional polarity (5' end and 3' end)
Importance/Function - Macromolecule: Nucleic Acids
carry genetic information in the sequence of its nucleotides
Structure - Macromolecule: Nucleic Acids
-3 components:
↳ nitrogen base (differs, names the nucleotide (ex: ATP))
↳ phosphate group
↳ Sugar (5C)
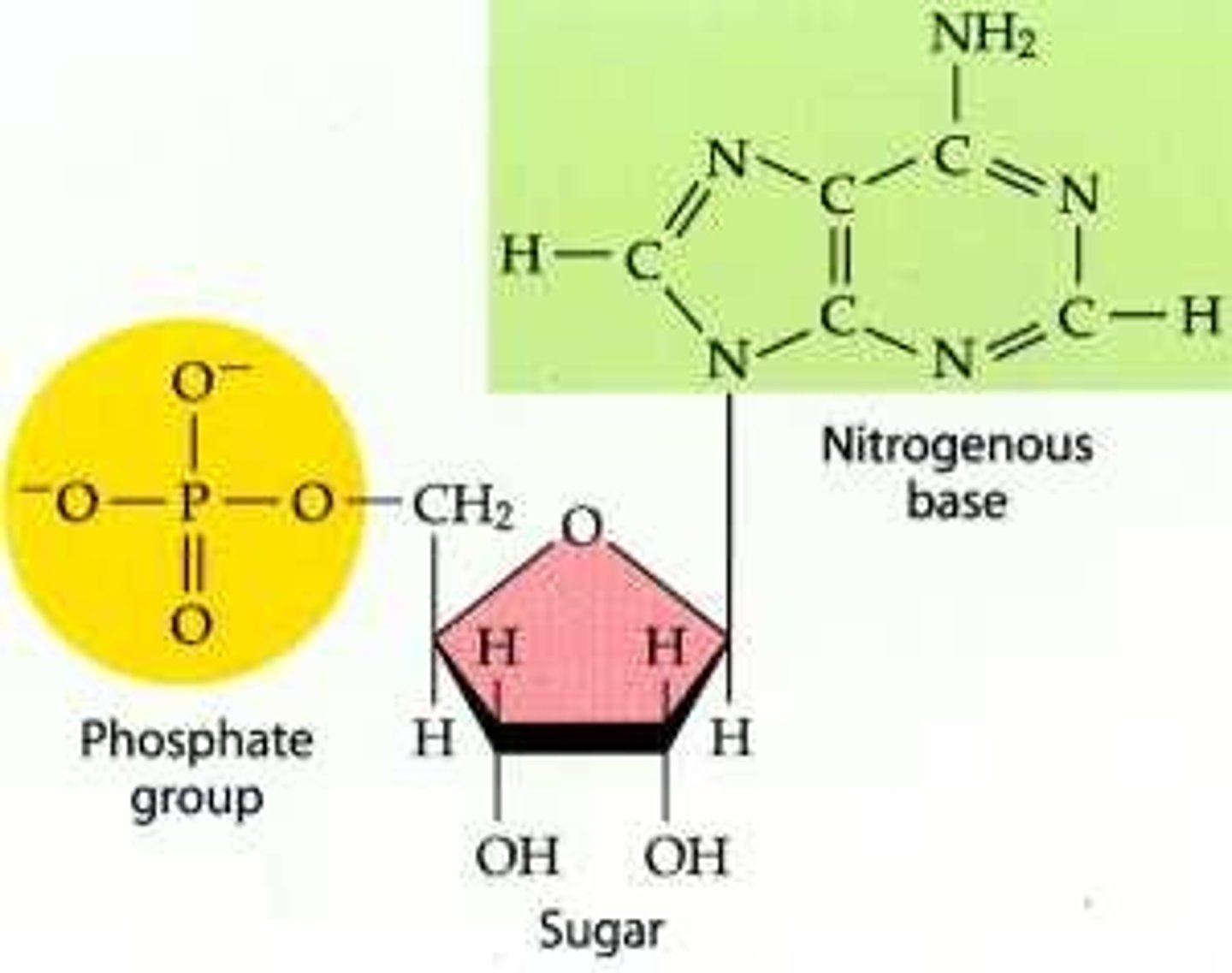
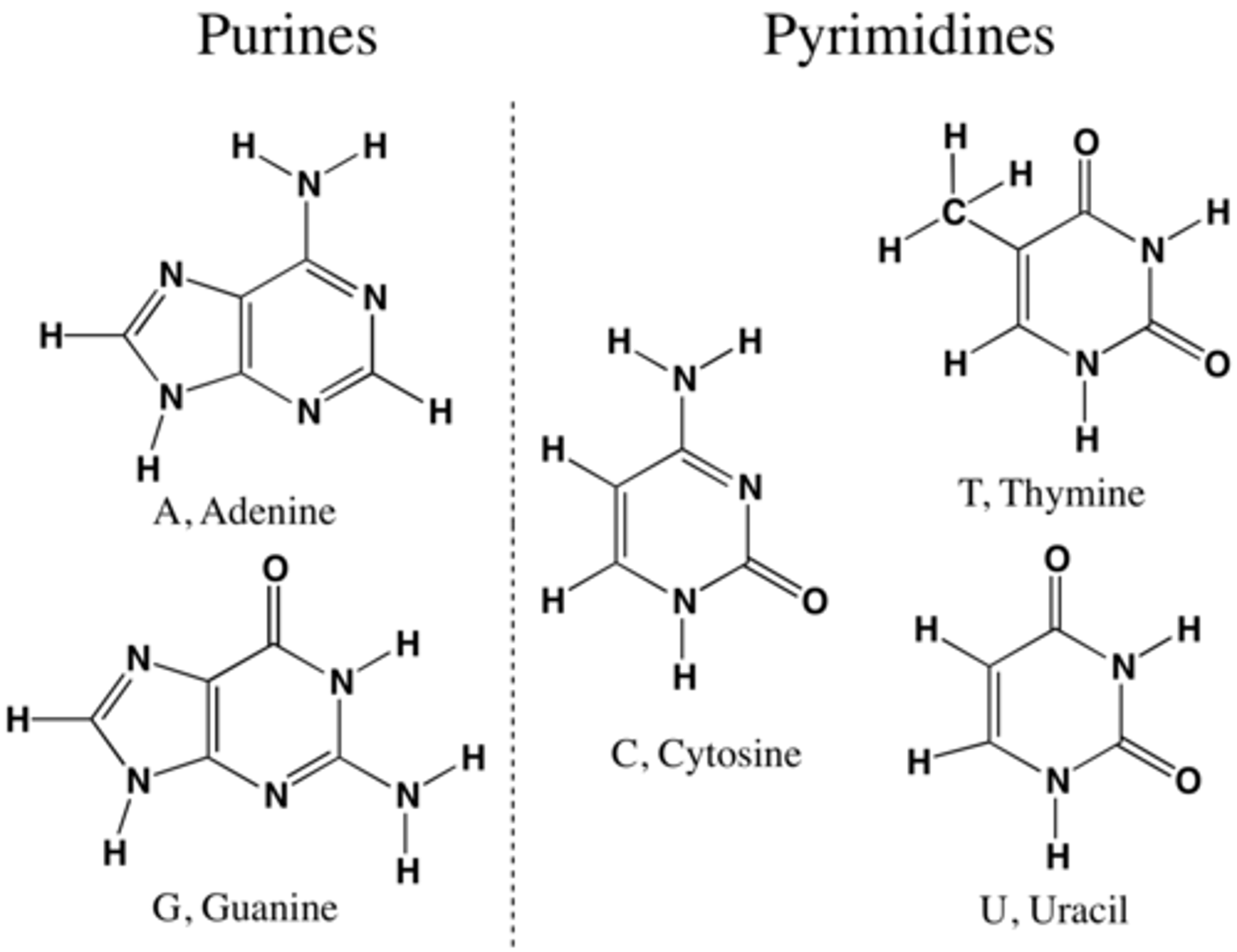
Nitrogenous Bases Differ in DNA and RNA
↳ thymine - DNA
↳ uracil - RNA
↳ purines (double ring): adenine and guanine
↳ pyrimidines (single ring): cytosine, thymine (DNA), uracil (RNA)
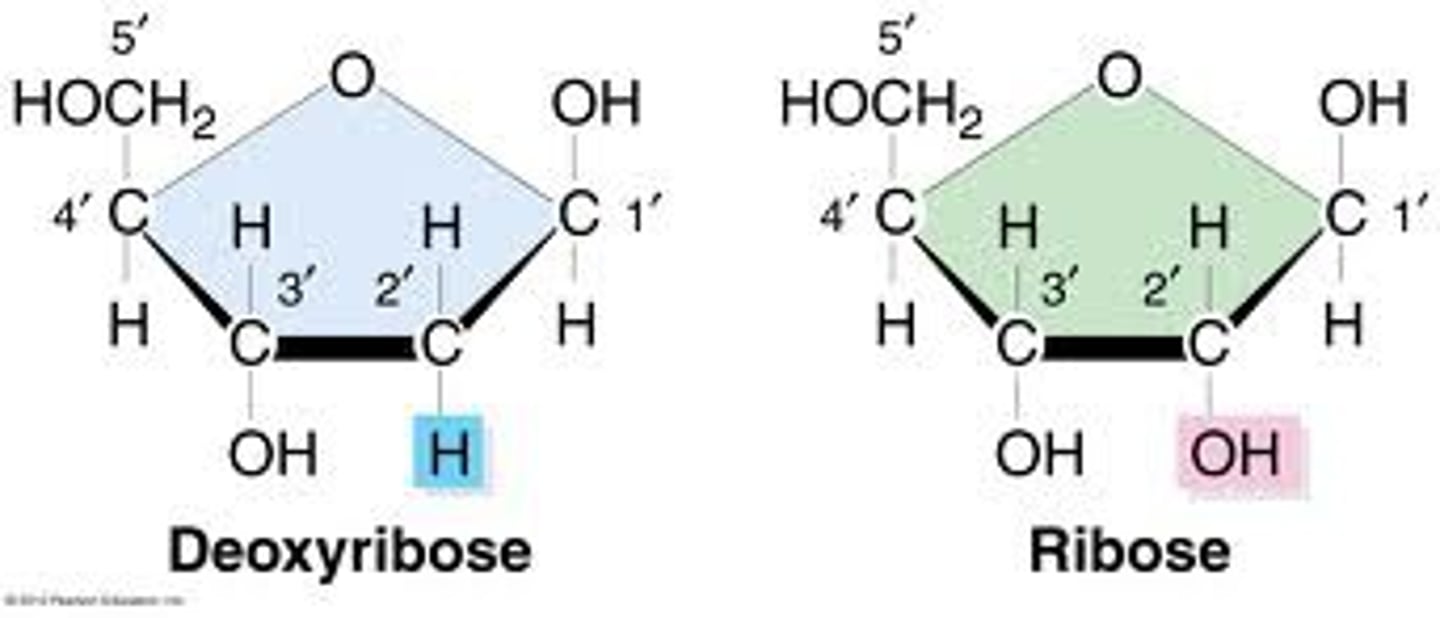
Sugar Group Differs in DNA and RNA
-> ribose - RNA -> C2 bonded with an OH
↳ ribonucleotides
->deoxyribose - DNA -> C2 bonded with H
↳ deoxyribonucleotides
- (if there is no H or OH shown bonded to C2, the default is H (DNA))
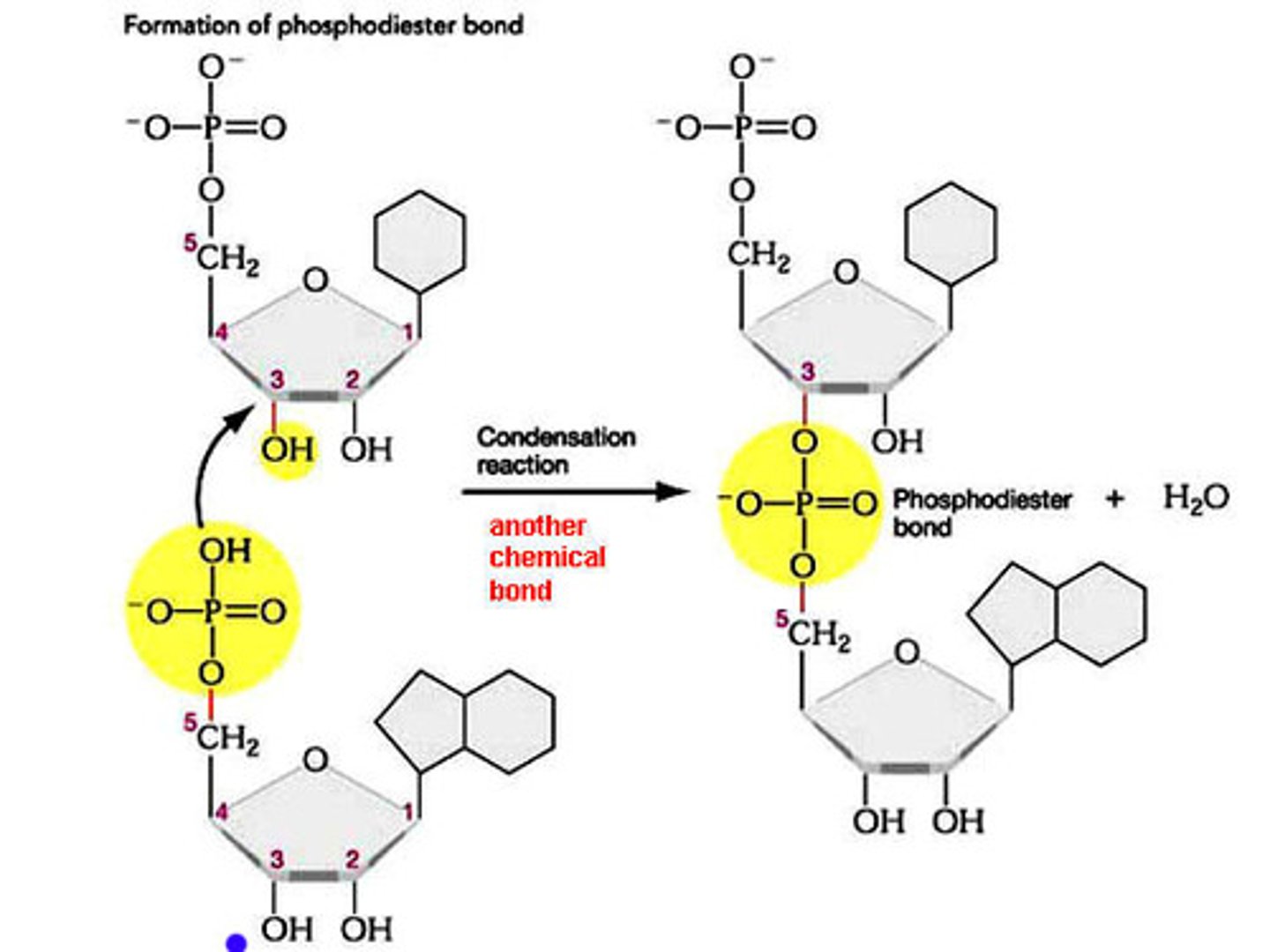
Polymerization of Nucleotides - Macromolecule: Nucleic Acids
↳ nucleotides join together through phosphodiester bonds between the OH group of one nucleotide (C3) and a phosphate group on another (C5)
↳ the C3 loses a hydrogen in a condensation reaction when a nucleotide is added
Subunit - Macromolecule: Fats & Lipids
fatty acids
Intermediate Molecule - Macromolecule: Fats & Lipids
none
Polymer - Macromolecule: Fats & Lipids
fats & membrane lipids
Covalent Bond - Macromolecule: Fats & Lipids
none
Polarity - Macromolecule: Fats & Lipids
non-polar
Importance/Function - Macromolecule: Fats & Lipids
↳make up membranes of the cell (phospholipids) and are stored in the cytosol in droplets (triacylglycerols) as an energy reserve
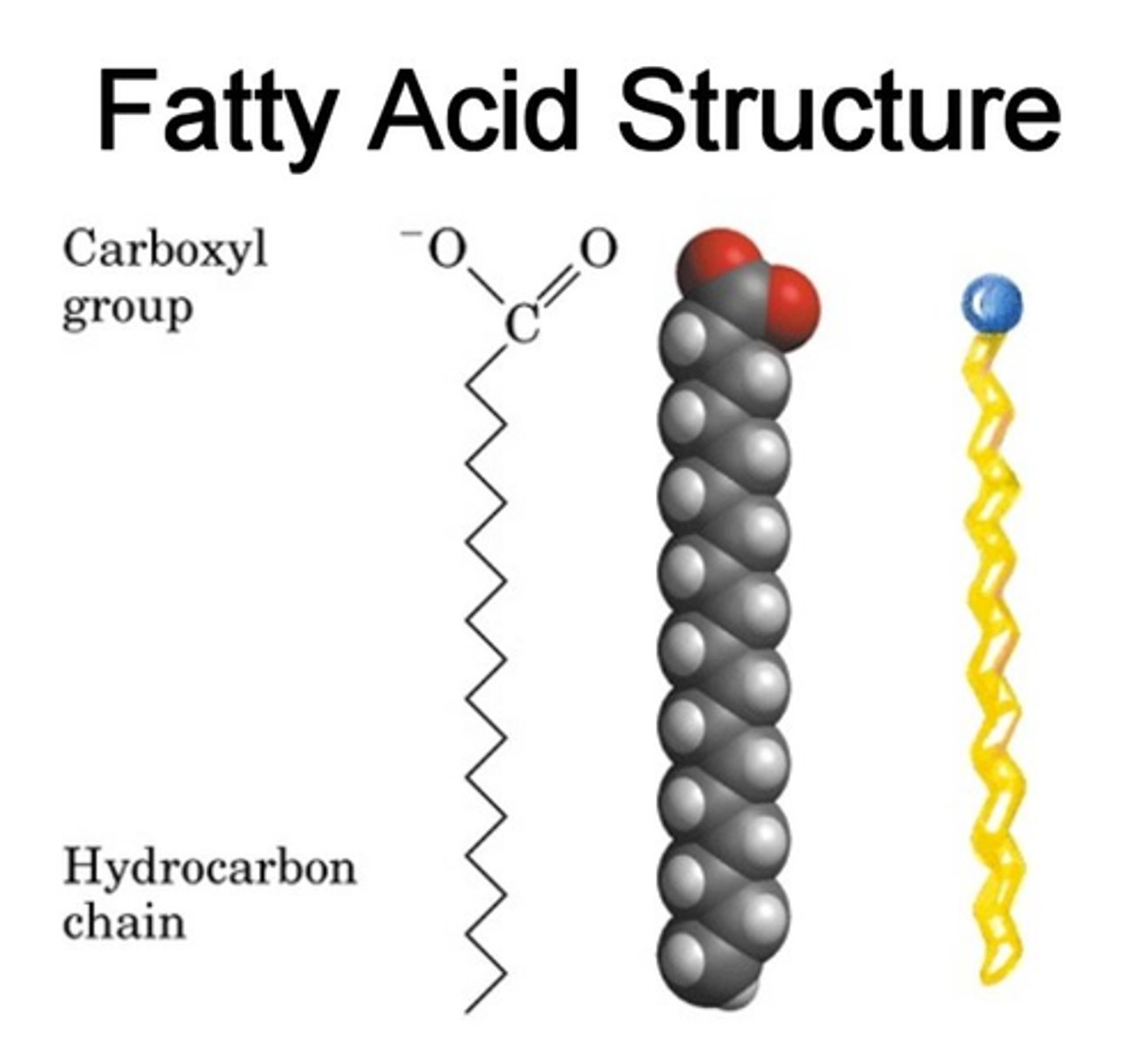
Structure - Macromolecule: Fats & Lipids
↳ a fatty acid has 2 regions:
1) hydrophobic, hydrocarbon tail
2) hydrophilic, carboxylic acid head (usually covalent attached to head groups)
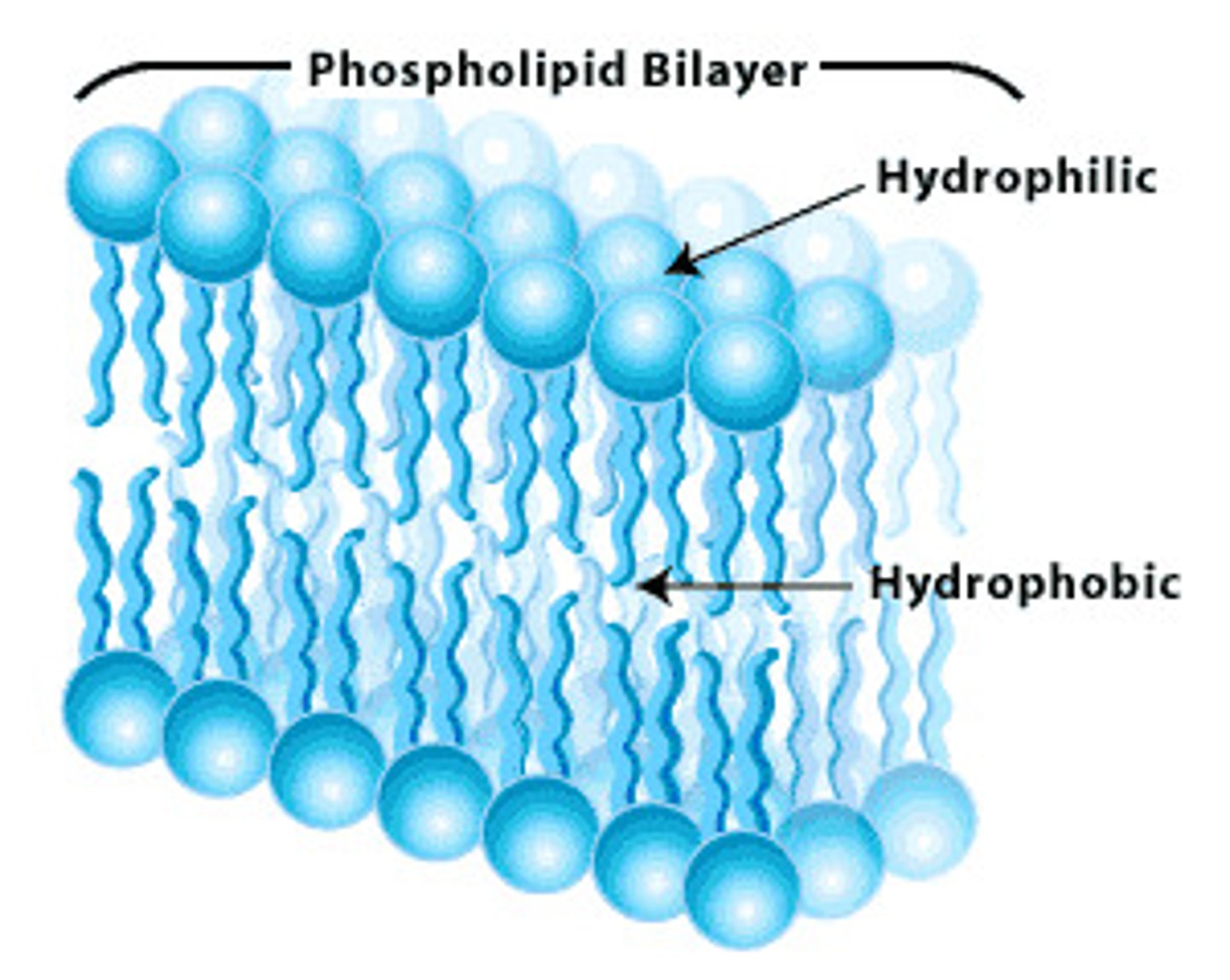
Phospholipids Make Up Biological Membranes
↳ 2 fatty acid tails are attached to a glycerol and hydrophilic head group
↳ amphipathic nature (hydrophilic and hydrophobic ends) promotes formation of a membrane in aqueous solution
↳ lipids do not form "traditional polymers" - no covalent bonds between adjacent lipid molecules
A Cell is Formed from Carbon Compounds
↳ all molecules are based on carbon which forms highly stable covalent bands
↳ C-C bonds can form the backbone of complex molecules
↳ other chemical groups include: methyl (-CH3), hydroxyl (-OH), carboxyl (-COOH), carbonyl(-C=O), phosphoryl (-P0₃), and amino (-NH₂) groups
↳ cells contain 4 major families of molecules (macromolecules) made of carbon backbones and different chemical groups
4 families of macromolecules
1) carbohydrates (polysaccharides - glycogen and starch)
2) fats and lipids
3) proteins
4) nucleic acids
Macromolecules and Their Subunits
1) carbohydrates (polysaccharides - glycogen and starch) - sugars (monosaccharides)
2) fats and lipids - fatty acids
3) proteins - amino acids
4) nucleic acids - nucleotides
↳ the relationship is reversible - macromolecules can be broken down into their subunits
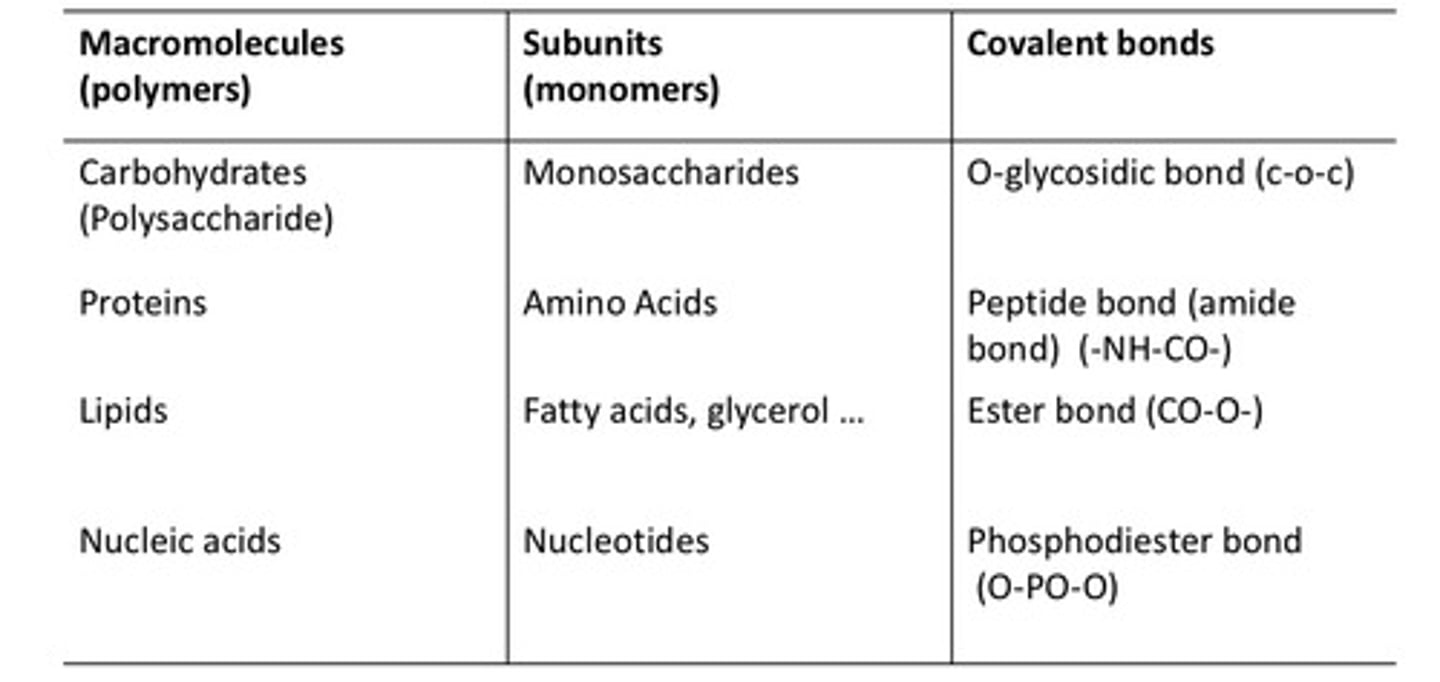
Condensation vs. Hydrolysis
condensation: the process that forms the macromolecules
↳ requires energy, energetically unfavorable
↳ loses/produces H₂O
hydrolysis: the process that breaks down the macromolecules
↳ loses energy, energetically favorable
↳ gains H₂O
-> applies to carbohydrates, proteins, and nucleic acids
How to remember the elements in Macromolecules (mnemonic):
- Carb - CHO
- Lipid - CHO
- Protein - CHON
- Nucleic Acid - CHONP
Still learning (23)
You've started learning these terms. Keep it up!