pmcol hallucinogens
1/69
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
70 Terms
besides hallucinations, what else is associated with hallucinogens
substantial changes in thoughts, emotions, and conciousness
what do most hallucinogen drugs alter
the function of monoaminergic (esp. serotonin) or glutamatergic systems
4 classes of hallucinogens
classical psychedelics (serotonin systems)
dissociative agents
deliriants
oneirogens
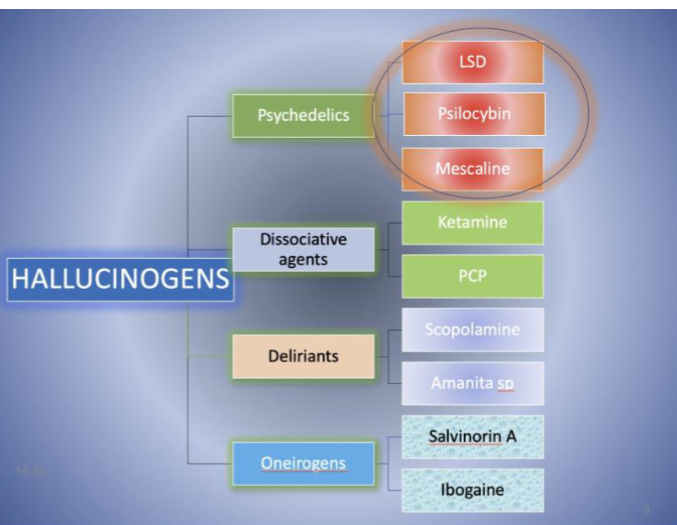
drugs included in classical psychedelics (3)
lysergic acid diethyl amide (LSD)
psilocybin
dimethyltryptamine (DMT)
common mechanism of action of classical psychedelics
serotonin 5HT2a receptor (Gq coupled protein)
discovery of LSD
fungus growing on rye produced ergot alkaloids → unstable = break down to LSD
ergot alkaloids
good vasoconstrictors from fungus that grows on rye
not stable in solution
mechanism of LSD
has over 50 targets (many 5HT + monoamine)
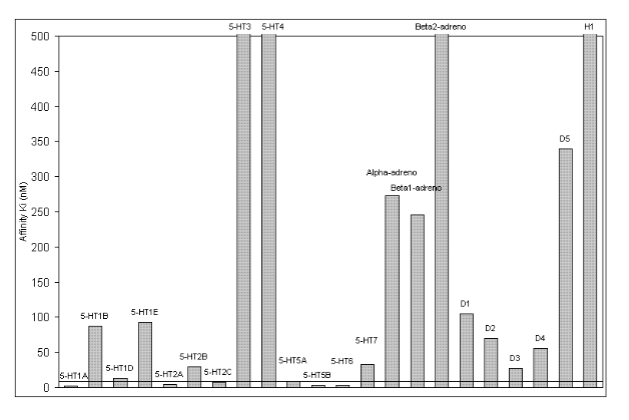
LSD at 5HT2a receptor
high affinity partial agonist
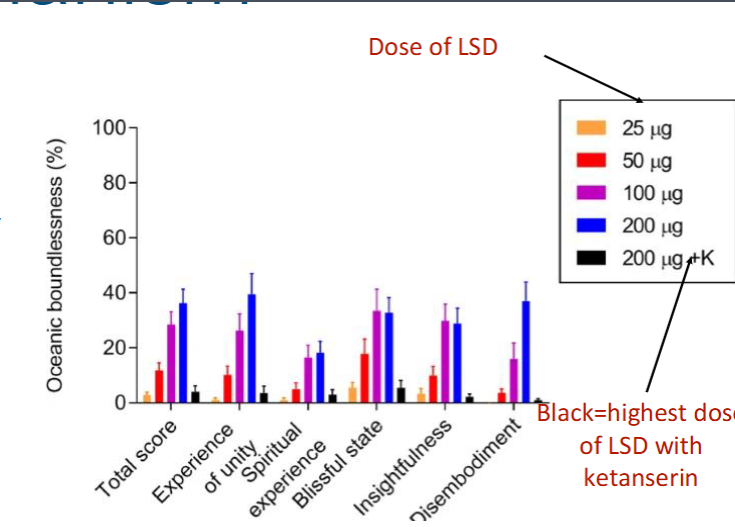
proof that 5HT2a activation causes hallucinogens (2)
modified mouse models lacking these receptors do not show hallucinogenic like behavior to LSD
humans with 5HT2a antagonist (ketanserin) pretreatment block LSD hallucinogen affects
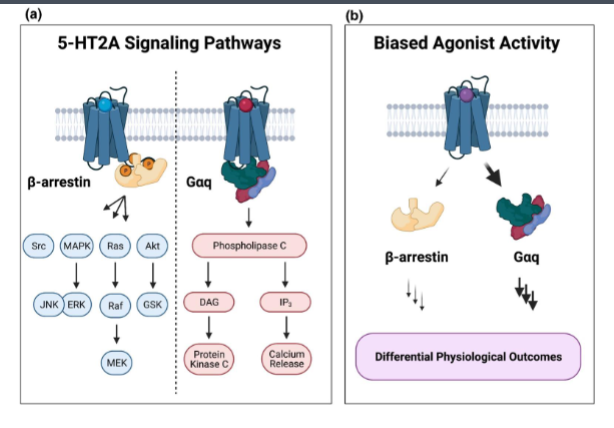
T of F: all 5HT2a agonists are hallucinogenic and why
false due to biased agonism
LSD (+other hallucinogenic serotonin agonists) activates phospholipase C while non-hallucinogenic agonists activate beta-arrestin signaling over PLC signalling
effects of 5HT1 receptor activation (3)
dilated pupils
increased heart rate
increased blood pressure
5HT1 are Gi coupled
effects of 5HT2b receptor activation
valvulopathies over long time use (changes in heart valves) → lethal
hallucinogenic persisting perception disorder
distressing visual hallucinations/ disturbances that appear following drug use
LSD tolerance
1 dose of LSD can lead to tolerance that lasts for several days by downregulation of only 5HT receptors
cross tolerance with other serotonin hallucinogens
psilocybin
indole + amine group → homologous to serotonin (bind with high affinity to serotonin receptors)
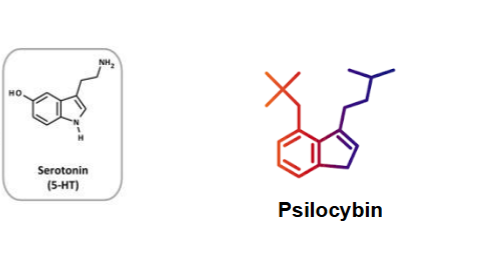
origin of psilocybin
naturally occurring in psilocybin mushrooms used for years
what happens pharmocokinetically after ingestion of psilcybin
psilocybin (prodrug) rapidly dephosphorylated to psilocin in body
psilocybin at 5HT receptors
partial agonist at 5HT receptors
high affinity at: 5HT2b + 5HT2c
lower affinity at: 5HT2a → hallucinogenic effects (higher doses needed)
dissociative hallucinogens
distort perception of sight and sound and produces feelings of detachment from environment or self → can lead to depersonalization
analgesia
phencyclidine
(aka angel dust)
NMDA receptor antagonist (like ketamine) + dopamine transporter inhibitor (abuse liability)
datura
deliriant hallucinogen containing tropane alkaloids (scopolamine + atropine)
that is lethal @ higher doses
atropine + scopolamine
competitive antagonists at muscarinic cholinergic receptors (loss of cognition + conciousness (brain) + ANS effects)
deliriant hallucinogens
characterized by extreme confusion and inability to control ones actions
highly susceptible to anterograde amnesia
low lucidity
low lucidity
part of deliriant hallucinations where hallucinations are perceived as real and users may not be aware they are in a state of drug-induced consciousness
oneirogen
substance that produces dreamlike state of consciousness similar to REM state +low lucidity
salvia divinorum
sage plant that includes active ingredient salvinorin A
salvinorin A
active ingredient in salvia that is an agonist at the kappa opioid receptor
produces short acting hallucinogens
4 key interventions to treatment of addiction emergnecies
reversal agents
downstream effects on body
alternative pathways
chronic changes
what are the more serious presentations of alcohol consumption in ER
trauma, suicide ideation, alcholoic ketoacidosis, panreatitis
what drugs to give to patient who is agitated and swinging widlly in ER
sedating agents (minimal effect on respiratory drive): benzodiazepines + first gen antipsychotic drugs (haloperidol)
what drugs to give patient who has chronic alcohol use
chronic use = depletion of:
thiamine (vitamin B1) → reduction thiamine related enzymes
folic acid (Vitamin B12) → increased urinary excretion
magnesium → increased urinary + bowel excretion
what happens during alcohol withdrawl
down regulation of GABA and increases glutamatergic signaling, removal will lead to increase in glutamate
seizures
what to give patient who comes into ER for withdrawl symptoms
GABA enhancing drugs to resotre GABA:glutamate imbalance
IV benzodiazepines + phenobarbital to open GABA + ketamine
diazepam vs lorazepam
diazepam most commonly use benzodiazepine but lorazepam used for history of liver disease
propofol
hyperpolarize GABA → inhibit NMDA receptors → stop respiratory drive → intubation
symptoms from opioid overdose
hypoxia, pneumonia, arrythmias
symptoms of opioids withdraw
dehydration, pain, electrolyte imbalance
Narcan
used in ER to treat opioids overdose
3 opioids agonists in Alberta
methadone: full agonist at mu opioids
suboxone
sublocade: injection buprenorphine
methamphetamine pharmacology
causes large dopamine surges particulary in reward systems
inhibits dopamine reuptake + triggers dopamine vesicle release
aura
preceeds migraines
visual disturbances consisting of flashing lights or zigzag lines moving acroos field of vision
what are auras thought to be driven by
cortical spreading depression: wave of neuronal depolariztion followed by desensitization (depression) that propagates across cortex
what are migraines causes by
mix of genetic and environmental factors
increases in women after puberty (hormones?)
familial hemiplegic migraine
genetic autosomal dominant inheritance that infludes weakness in half of the body
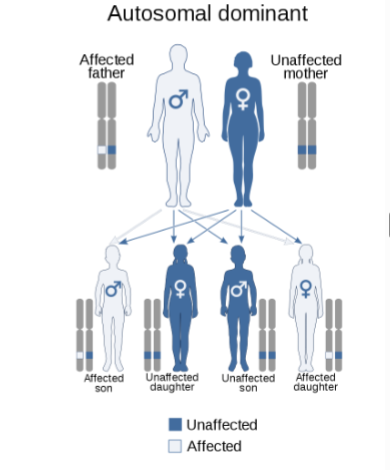
what are the 3 known genetic mutations associated with familial hemiplegic migraines
P/Q-type calcium channel
Na+/K+ ATPase
Na+ channel subunit
what do the mutations that cause familial hemiplegic migraines do
they lower the threshold for cortical spreading depression
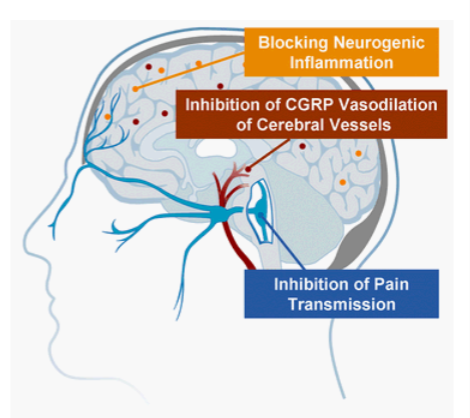
trigeminal nerve
largest cranial nerve that:
sense pain + temp in head region
innervate dura mater
control cerebral blood vessels (trigminovascular system)
what are the three branches of peripheral processes of the trigeminal nerve
ophthalmic, maxillary and mandibular
how is the pain of a migraine detected
by the ophthalmic branch of trigeminal nerve innervating dura mater and blood vessuls
steps of migraine
extracerebral vessels dilate during migraine attack
cranial blood vessel stimulation provokes headaches
vasoconstrictor drugs alleviate pain
what is the vasoconstricotr of migraines
5HT (particularily 1B), migraineurs have low levels of this between attacks and is released during attacks
calcitonin gene-related peptide
located in and released by trigeminal peripheral afferents in response to pain
leads to vasodilation
(elevated elvels in those with migraine)
treatment strategies of migraine
prophylactic: taken daily to prevent
abortive: taken when attack occurs
both used
what are the prophylactic non-pharmacological interventions of migraines
identifying triggers (diet, excercise, sleep, etc.)
what are the prophylactic pharmacological interventions of migraines (3)
beta blockers (propanolol): decrease bp
anticonvulsatns (gabapentins): block pain transmission
antidepressants (amitriptyline): serotonin reuptake inhibitor
rare treatments
what are the abortive treatments of migraine + risk
non-specific analgesics
risk: medication overuse headahce (chronic use makes ur headaches worse over time)
caffeine
adenosine receptor antagonist (on vessles of trigenimal system) → vasoconstriction in migraines
increases abosorption of some analgesics
may trigger headaches of rebound vasodilation headache (withdrawal)
ergot alkaloid
like LSD, binds 5HT-1b.d receptors → inhibit neurogenic inflammation
downsides of ergot alkaloid
low degree of receptor selectivity (bind to other 5HT receptors and adrenergic receptors) → increases drug induced side effect risk
ex. is coronary vasoconstriction
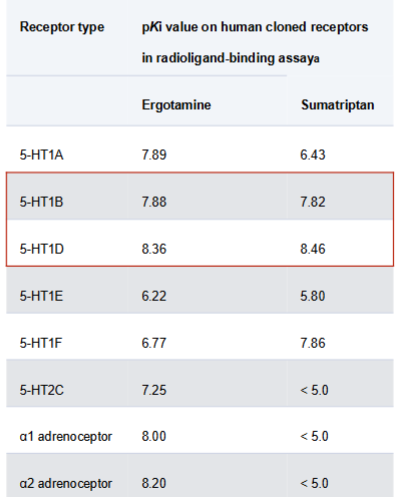
pharmacokinetics (absorption, distribution, metabolism and excretion) of ergotamines
abs + dist: large first pass metabolism = low bioavailibility (caffeine helps improve rate and extent of absorption)
metabolism: poorly metabolized by liver (2 hr half life)
excretion: in bile
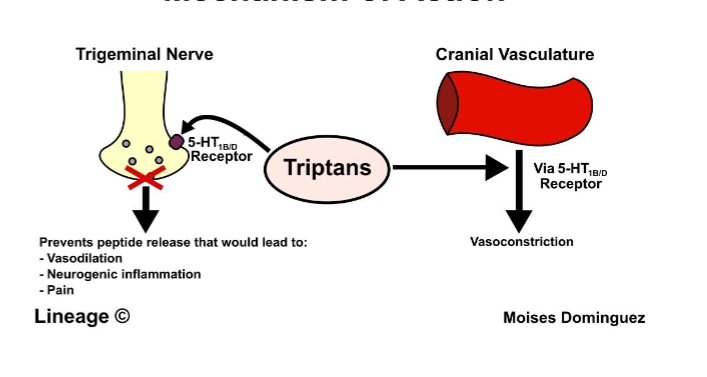
triptans
Sumatriptan
first line migraine therapy
selective 5HT 1b/d agonist → vasoconstriction + inhibition of trigeminal nerve = less pain
no ergotamine like side effects
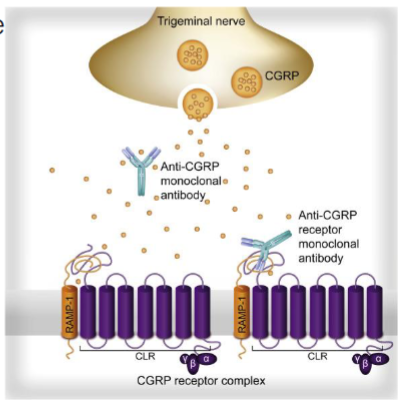
CGRP inhibitors
small molecule of CGRP antagonists or antibodies
monoclonal antibodies
sent to CGRP receptor of CGRP itself to inhibit CGRP signaling → vasoconstriction
BIBN4096
Olcegepant, a CGRP inhibitor that has good efficacy in treating migraines
poor bioavailibitliy (abandoned at phase II)
MK-0974
Telcagepant (CGRP inhibitor)
had serveral phase III trials showing anti-migraine efficacy + safety
problems: elevation of liver aminotransferease = abandoned
Rimegepant (Nurtek)
small molecule CPRP receptor antagonist that is effective in migraine treatment and less effect on liver aminotransferease levels
gepants
small moleculr antagonists at CGPR receptor
biologics
antibodies made naturally in lab
easier to make but expensive + less stable