Chem 105a Lab Exam: Experiment 7 (Gas Volumetric Analysis, the Concentration of a Hydrogen Peroxide Solution)
1/22
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
23 Terms
volumetric analysis
method of analysis based on volume
what was the purpose of this experiment?
determining the concentration of a solution of hydrogen peroxide volumetrically through application of the ideal gas law to the volume of gas that is evolved when we decompose hydrogen peroxide
H2O2(l) → H2O(l) + 1⁄2O2(g)
why does the FDA not require printed expiration dates on containers of low-concentration hydrogen peroxide?
because hydrogen peroxide is extremely stable at low concentrations and only decomposes at a rate of about 1% per year
is the decomposition of pure liquid hydrogen peroxide an endothermic or exothermic process?
it is a highly exothermic process
activation energy
the minimum amount of energy required to start a chemical reaction
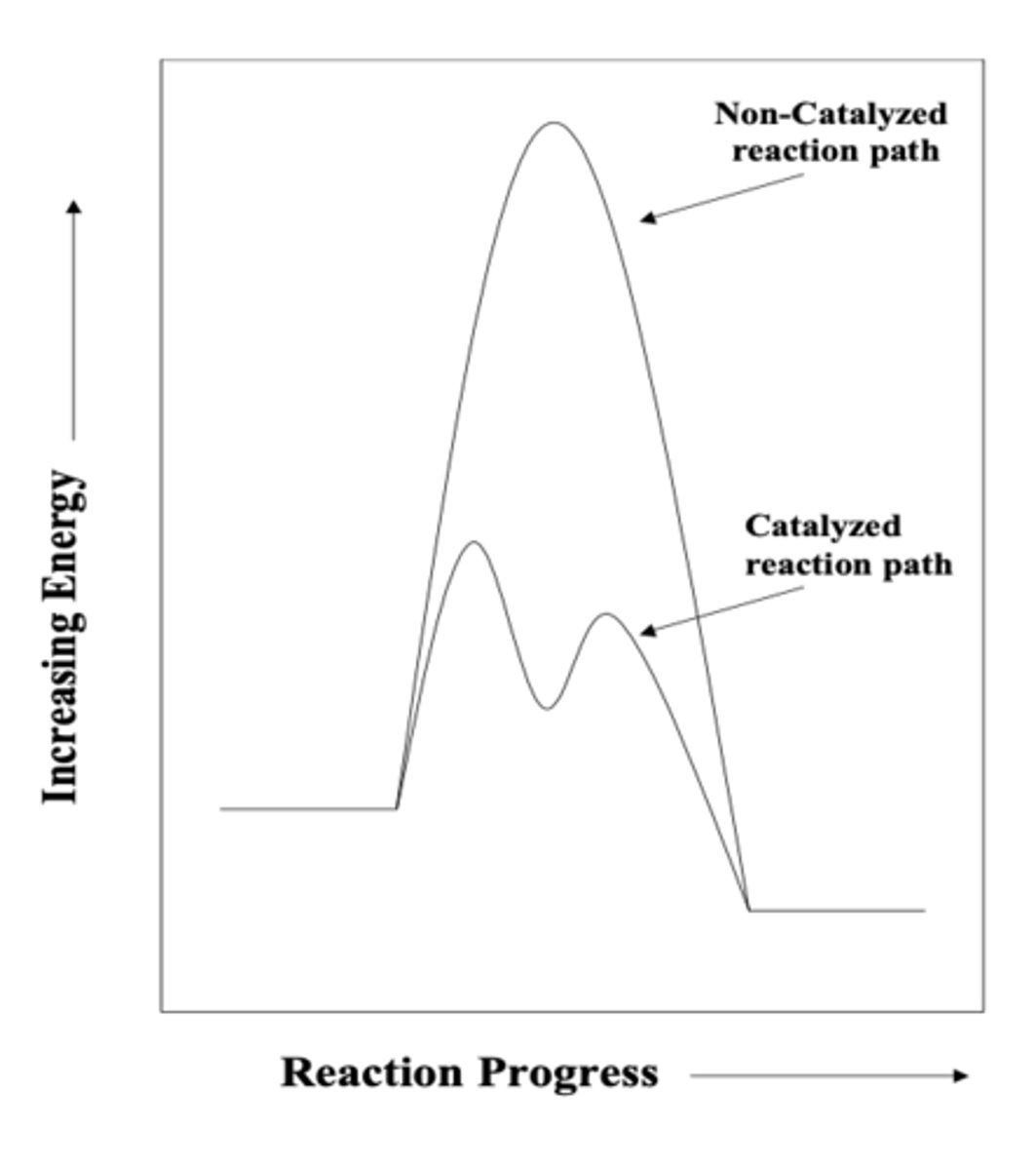
catalyst
substance that speeds up the rate of a chemical reaction by offering an alternative reaction pathway that has a lower energetic barrier
do catalysts get consumed in the overall reaction?
no; they are consumed an then reproduced
what metals acts as a catalyst between fuel compounds and oxygen in a catalytic converters in cars?
platinum, palladium or rhodium
what was the catalyst in this experiment?
iodide in the form of potassium iodide
what is the first step in the iodide-catalyzed decomposition of hydrogen peroxide?
hydrogen peroxide reacts with the iodide ion to produce water and the hypoiodite intermediate
H2O2(aq) + I-(aq) → OI-(aq) + H2O(l)
what is the second step in the iodide-catalyzed decomposition of hydrogen perxide?
the hypoiodite reacts with a second molecule of hydrogen peroxide to produce water and oxygen and convert the hypoiodite back to iodide
H2O2(aq) + OI-(aq) → I-(aq) + O2(g) + H2O(l)
what were the learning objectives for this experiment?
- understand the concept of a reaction catalyst and why it is necessary for this experiment
- understand the concept of partial pressure as it applies to collecting a gaseous sample over water
what were the experimental objectives for this experiment?
- perform a volumetric analysis of a gas evolution reaction
what was the purpose of the second pipet, pipet B, in this experiment's gas-collection appartus?
it was used to equalize the pressure of pipet A (the gass collection pipet) with the atmospheric pressure

when the water levels in pipet A and B are at the same height, what is the pressure in pipet A equal to?
the atmospheric pressure
does the exact mass of KI used in this experiment matter?
no, the exact mass does not matter, as the catalyst is not consumed
how are the pressures in pipet A and B equalized?
by lowering pipet B
how is the volume of O2(g) produced related to the volume of H2O2(aq) used in the trial?
for every two moles of H2O2 used, one mole of oxygen gas is produced
2H2O2(aq) —> O2(g) +2H2O
if the volume of O2(g) produced exceeds 50 mL, how should you adjust the volume of H2O2 used?
the volume of H2O2 used should be decreased since it has a directly proportional relationship with the amount of O2 gas produced
Avogadro's Law
equal volumes of gases at the same temperature and pressure contain equal numbers of moles (V1/n1=V2/n2)
how is avogadro's law used to find the moles of oxygen gas produced in pipet A?
if the pressure in pipet A is equalized to atmospheric pressure for both the initial and final volume readings, the moles of oxygen gas produced are proportional to the change in volume reading in pipet A.
Ideal Gas Law
the relationship PV=nRT, which describes the behavior of an ideal gas
P= pressure in atm
V = volume in liters
n = moles
R = ideal gas constant (.08206 Latm/molK)
T = temperature in Kelvin
what were common sources of error in this experiment?
- systematic error: not making sure the water levels were equal would affect the recorded amount of oxygen produced; not letting the reaction complete would affect the amount of oxygen gas produced