Enzymes as Drug Targets
1/33
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
34 Terms
What is Ki?
Inhibition constant
What does Ki measure?
Measures the binding affinity (potency) of an inhibitor for an enzyme. Lower Ki = stronger binding
A low Ki value means?
Binds strongly (more potent)
A high Ki value means?
Binds less strongly (less potent).
Weaker inhibitors have higher Ki values
How is Ki related to thermodynamics?
Ki is related to binding free energy (ΔG).
Lower Ki corresponds to more negative ΔG and stronger binding.
How do inhibitors act on enzymes?
Inhibitors can resemble the substrate in structure and alter Km, Vmax or both
What are the key features of competitive inhibition?
Km increases
Vmax is unchanged
Inhibition decreases with increasing substrate concentration
What is the equation for Km in competitive inhibition?
Km(I) = Km (1 + [I]/Ki)
Km(I) = apparent Km (with inhibitor)
[I] = inhibitor concentration
Ki = inhibition constant (strength of inhibitor)
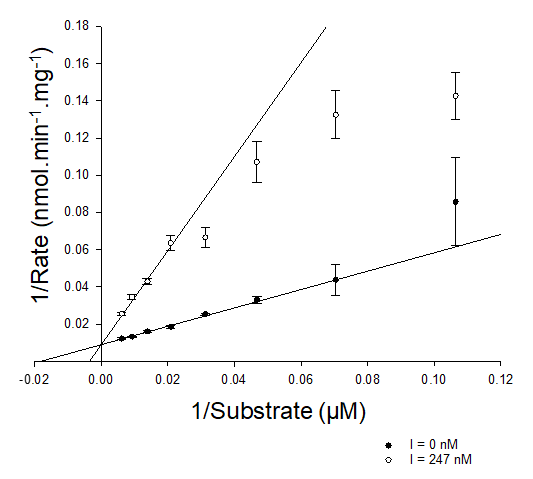
How do you identify competitive inhibition on a Lineweaver–Burk plot (1/v vs 1/[S])?
Km increases, so X-intercept shifts towards 0
Vmax stays the same, so y-intercept stays the same
Steeper plot
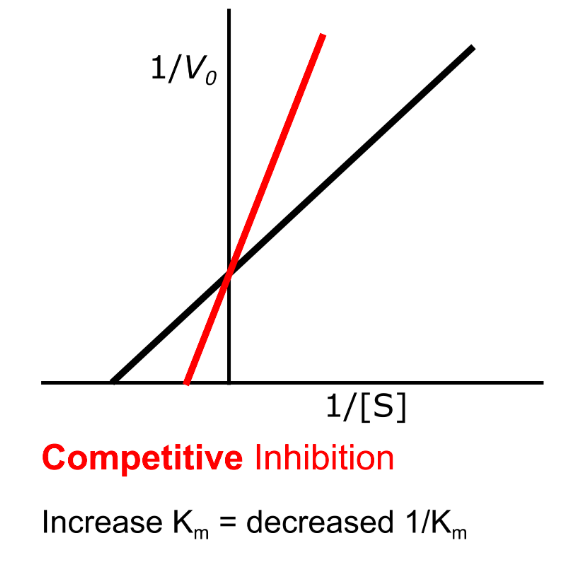
What are the key features of non-competitive inhibition?
Km is unchanged
Vmax decreases
Inhibition is not reduced by increasing substrate concentration

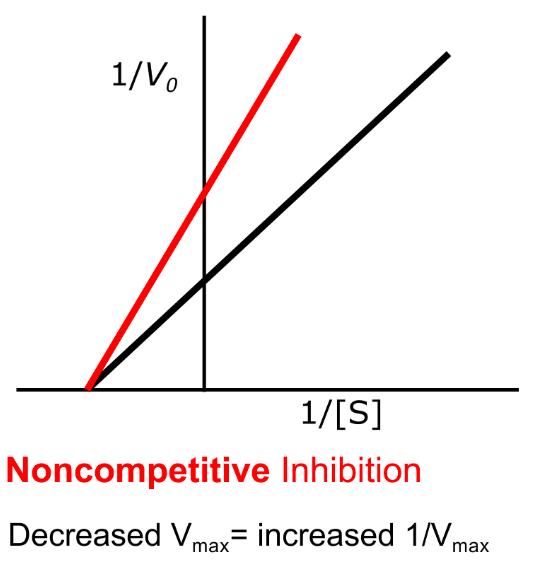
What is the kinetic relationship in non-competitive inhibition?
Vmax/Km (I) = Vmax/Km(1 + [I]/Ki)
Km (I) = apparent Km (with inhibitor)
[I] = inhibitor concentration
Ki = inhibition constant (strength of inhibitor)
How does non-competitive inhibition appear on a Lineweaver–Burk plot?
Vmax is reduced, so Y-intercept increases
Km is unchanged, so X-intercept is unchanged
Slope increases
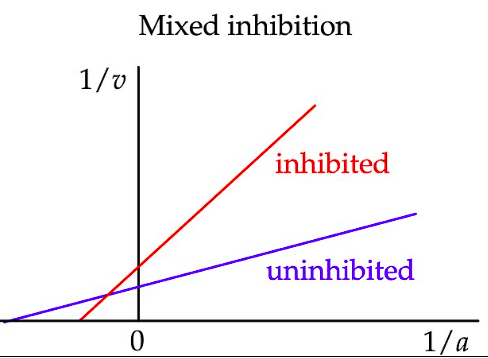
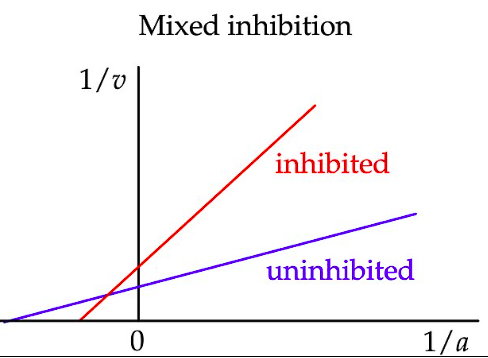
What are the key features of mixed inhibition?
Km increases
Vmax decreases
How does mixed inhibition appear on a Lineweaver–Burk plot?
Km is increased (as for competitive inhibition). X-intercept shifts left or right.
Vmax is decreased (as for non-competitive inhibition). Y-intercept increases.
The lines intersect left of the y-axis
What does the α factor describe in mixed inhibition?
The relative contribution of competitive and non-competitive inhibition
What is the mechanism of uncompetitive inhibition?
The inhibitor binds to the ES complex, not to the free enzyme, forming an inactive ESI complex
Parallel lines in Lineweaver-Burk plot
Both Km and Vmax reduced by same amount, (1 + [I]/Ki)
Increasing substrate concentration increases inhibition.
![<p style="text-align: justify;">The inhibitor binds to the ES complex, not to the free enzyme, forming an inactive ESI complex</p><ul><li><p style="text-align: justify;">Parallel lines in Lineweaver-Burk plot</p></li><li><p style="text-align: justify;">Both <em>K</em><sub>m</sub> and <em>V</em><sub>max</sub> reduced by same amount, (1 + [I]/<em>K</em><sub>i</sub>)</p></li><li><p style="text-align: justify;"><strong>Increasing</strong> substrate concentration <strong>increases </strong>inhibition.</p></li></ul><p></p>](https://knowt-user-attachments.s3.amazonaws.com/6ca8a47c-efc6-45ef-91e2-08ae874c4f1b.png)
How does uncompetitive inhibition affect Km and Vmax?
Both Km and Vmax are decreased by the same proportion.
They are reduced by a factor of 1 / (1+ [I]/Ki)
What are the key graphical and functional features of uncompetitive inhibition?
The Lineweaver-Burk plot gives parallel lines
Increasing [S] increases inhibition because more ES complexes (and so more binding sites for the inhibitor)
![<ul><li><p>The Lineweaver-Burk plot gives parallel lines</p></li><li><p>Increasing [S] increases inhibition because more ES complexes (and so more binding sites for the inhibitor)</p></li></ul><p></p>](https://assets.knowt.com/user-attachments/bd5e9902-1fef-4a3d-b25c-fb2adbd591fd.png)
What do dose response curves measure?
They measure the drug potency at a fixed substrate concentration

Example of a dose response curve:
IC50 curves
What is IC50?
The concentration of inhibitor that reduces enzyme activity by 50%.
Why is IC50 not equal to Ki?
Ki = how tightly the inhibitor binds
IC50 = inhibitor concentration that gives 50% inhibition
At first glance, it seems logical that the concentration needed for 50% inhibition should directly reflect binding strength.
However, IC50 depends on experimental conditions (e.g. substrate concentration), whereas Ki is the inhibitor’s intrinsic binding constant.
What does IC50 represent?
The inhibitor concentration required to reduce enzyme activity by 50%; used as an experimental measure of drug potency under specific assay conditions.
What is tight-binding inhibition?
Tight-binding inhibition means the inhibitor binds so strongly to the enzyme that it’s almost irreversible — the inhibitor and enzyme form a very stable complex.
When does tight binding inhibition occur?
When the concentrations of active enzyme and inhibitor are similar.
In tight binding inhibition, what will happen to the Ki value if you increase the enzyme concentration?
The apparent Ki value will increase with increasing enzyme concentration
Because free inhibitor ≠ total inhibitor.
What features of tight-binding inhibitors make them desirable drugs?
Tight-binding inhibition often has very slow onset, and the enzyme inhibitor complex can have a very long half-life (Ki = koff/kon);
This means that infrequent dosing regimens can be used (which is easier for patient, reduces side-effects etc)
What are irreversible inhibitors?
Inhibitors that permanently inactivates the enzyme, often by covalent bonding.
Give a drug example of irreversible inhibition.
Penicillins (β-lactams)
What enzymes do penicillins inhibit?
Penicillin-binding proteins (PBPs).
If Km increases but Vmax stays the same, what type of inhibition is present?
Competitive inhibition.
If Vmax decreases but Km is unchanged, what type of inhibition is present?
Non-competitive inhibition.
If both Km and Vmax decrease proportionally, what type of inhibition is present?
Uncompetitive inhibition.
What do parallel lines on a Lineweaver-Burk plot indicate?
Uncompetitive inhibition.