CHEM250 Final Review
1/61
Earn XP
Description and Tags
Inorganic chemistry
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
62 Terms
Nitrogen oxide reactions
NO2 / N2O / N2O3 / N2O5 + H2O —> HNO3 / HNO2
Natural Nitrogen vs Phosphorus
N2: gas molecule with 1 sigma and 2 pi bonds
P4: solid molecule with 3 sigma bonds
Natural Oxygen vs Sulfur
O2: gas molecule with 1 sigma and 1 pi bond
S8: solid molecule with 2 sigma bonds
Natural Carbon vs Silicon
C2: sigma and pi bond combo (single, double, triple bonds)
Si5: tetrahedral network of sigma bonds
Key characteristics of period 2 and 3 bonds (4)
P2: pi bonds common as 2p orbital overlap is easy
P3: pi bonds not common as 3p orbital overlap is not easy
P3: expanded octet from d-orbitals so easy sigma bonds
Compounds with pi bonds are less likely to be solid
Phosphorous allotropes in order of stability
White (P4, tetrahedral), Red (P4 loses a bond and connects to make a chain), Black (more bonds break and forms sheet)
Phosphorous oxide reactions
P2O3 / P2O5 / P4O6 / P4O10 + H2O —> PO4 3-
P4 and limited oxygen
P4 + O2 —> P4O6 + H2O —> H3PO3 (-ous)
P4 and excess oxygen
P4 + xs O2 —> P4O10 + H2O —> H3PO4 (-ic)
Synthesis of P4 from rocks calcium phosphate
2Ca3(PO4)2 (s) + 6SiO2 (s) + 10C(s) —> 6CaSiO3 (l) + 10CO(g) + P4 (g)
Environmental impacts of phosphorus
Banned from detergents bc PO4 3- is not biodegradable and cannot be removed easily via water treatment
Industrial importance of phosphorus
Water softening / binding agent
Fertilizers
Pyrophosphate reaction
PO4Na2H molecules combine in condensation reaction to produce TPP 3- ligands
Industry phosphoric acid reaction
H3PO4 <-> H2PO4 - <-> HPO4 2- <-> PO4 3-
loss of H+ each time
Phosphoric acid and Phosphate ions are most common
Phosphorous acid
Diprotic, weak
Phosphite salts
ox state = 3
From P4O6
Phosphoric acid
triprotic, weak
phosphate salts
ox state = 5
From P4O10
Phosphorous to oxyacid salts
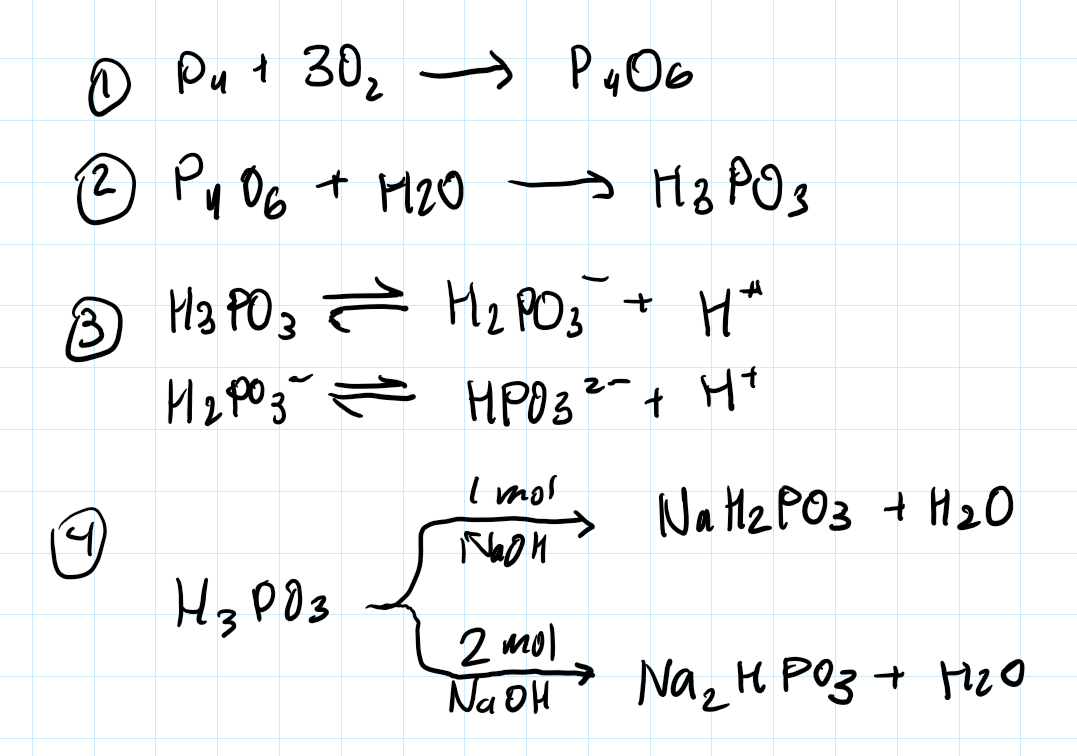
Natural Sulfur
Found as ores / in elemental form + has no smell (change of ox# = smell)
H2O vs H2S
H2O has hydrogen bonds, making it ordourless and liquid
H2S doesn’t, and creates a toxic smell
Claus Process Purpose
Converts H2S (g) —> S (s)
Claus Process Reactions
H2S (g) + 3/2O2 (g) —> SO2 (g) + H2O (l) | burning
SO2 (g) + 2H2S (g) —> 3S (s) + 2H2O (l) | catalyst, 200-300C
*Note ox states, disproportionate rxn
Acid Rain Reaction
S + O2 —> SO2 + H2O —> H2SO3
Acid Rain (cause, consequence, sol)
burning exhausts from industry creates oxides that react with water to form acid
Harms aquatic life and corrodes heritage buildings
Use scrubbers (eg. limestone) to reduce emissions
Sulfur oxides and acid production
S + O2 + heat —> SO2
2SO2 —> 2SO3 | V2O5 catalyst, O2
SO3 + H2SO4 —> H2S2O7
H2S2O7 + H2O —> 2H2SO4
Natural Sources of group 17
Found in combination with less electronegative elements, primarily metal halides (seawater and minerals)
Halogen inter reactions
½ I2 + Cl- —> no rxn
½ Cl2 + I- —> Cl- + ½ I2
2Br- + ½ Cl —> Br2 + Cl-
2HF —> F2 + H2 (electrolyte = molten KHF2)
Ox and Acid strength trend
Increasing oxidation state = Stronger acid = Salts are stronger oxidizing agent
Halogens with higher ox # will have more pull on the O, making H easy to remove
Chlorine compound usage
Bleach, Pool disinfectant, PVC, Rocket fuel
Chlorine Oxyacid production
Cl2 + H2O —> HOCl + HCl
Chlorine oxyacid table (compound, name, ion)

Oxyacid comparison of P,S,Cl
P
2 oxyacids (state: +3, +5)
Triprotic
S
2 oxyacids (state: 4,+6)
Diprotic
Cl
4 oxyacids (states: +1, +3, +5, +7)
Monoprotic
Metallic bonding
Atoms slide over each other because of e-, alloys swap metals and stay neutral
Ionic bonding