L8 - Ethical Challenges in SCI (spinal cord injury) Clinical Trials
1/13
Earn XP
Description and Tags
Spinal Cord Injuries
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
14 Terms
Review of ethics - discuss the following terms :
The Declaration of Helsinki
The Belmont Report
Tri-Council Policy Statement
FOUR principles of ethics
Main focus of the following document
The Declaration of Helsinki = global document for ethical principles regarding human experimentation & research ethics oversight
The Belmont Report = important biomedical ethics principles
Tri-Council Policy Statement = canadian policy for research involving human participants
FOUR principles of ethics
Autonomy = informed consent, respect for persons
Beneficence = maximize benefits
Nonmaleficence = minimize risks
Justice = fairness and equity
What are the ethics in clinical research (8) + give examples for each
Voluntariness
particpant’s choice to join a study
Informed Consent
participant is given details about the study, such as data collection, potential risks, privacy of data
Right to withdraw
participant’s ability to change their mind and leave the study without any consequences
Confidentiality
privacy of information / who has access to the information of the participant
Accessibility
participation is open to large population by accounting for different cultural backgrounds
Values and Priorities
focus on the outcome that matters to both the researcher and the participant
Follow up
check in with the participant after a study has ended to monitor effects
Safety
ensuring that side effects of a study are monitored and terminating the research if any serious risks are detected
What is the damage of spinal cord injury depending on the location of injury (3)
Cervical
difficulty breathing
shoulder movement
Thoracic
trunk stability
temperature regulation
Lumbar & Sacral
bladder and bowel control
leg function
What are some consequences of spinal cord injury - INTERNAL (5)
Paralysis → loss of motor and sensory function
Loss of control over bowel & bladder function
Pain
Susceptibility to infection
Affect other physiological functions → EX: cardiovascular / respiratory
What are some consequences of spinal cord injury - EXTERNAL (5)
Loss of independence
Limited employment opportunities
Altered purpose / meaning of life → EX: dancer loses their ability to move and so lose their identity
Personal and financial burden on the individual & caregiver
Societal cost (economic burden on healthcare system)
Explain the statement “Time is Spine” (pathophysiology of SCI)
Damage due to SCI evolves over time and early intervention is necessary to prevent irreversible destruction
Acute = seconds to days after incident
perfect timing where early intervention (stabilize blood flow) can prevent further damage
internal hemorrhage (bleeding)
inflammation
swelling due to extra fluid buildup
Intermediate = days to week after incident
still have time to intervene by focusing on supporting recovery
axons retracting SO leading to gaps between nerves
molecules released by astrocytes start to accumulate around injury site & prevent axon growth
Chronic = weekS to months after incident
low chance of recovery from damage SO shift focus to rehabilitation
physical scar is formed at injury site prevent aoxnal growth
loss of neuronal connections
Discuss the clinical trial phases of SCI (4) + some limitations ?
LIMITATIONS: financial reasons (too expensive to continue), enrollment issues (small population), low power (poor study design)
Phase I = safety
safest dose that is tolerable
closely monitoring side effects
testing on smaller population
Phase II = efficacy
aiming for most effective dose
measuring outcomes
testing on larger population
Phase III = confirmation
compare with other treatments (confirming effectiveness & safety)
monitor adverse events
involving control group
Phase IV = follow-up
detect long term side effects
put on the market
→ BUT lots of interventions do not make it to the market due to financial reasons
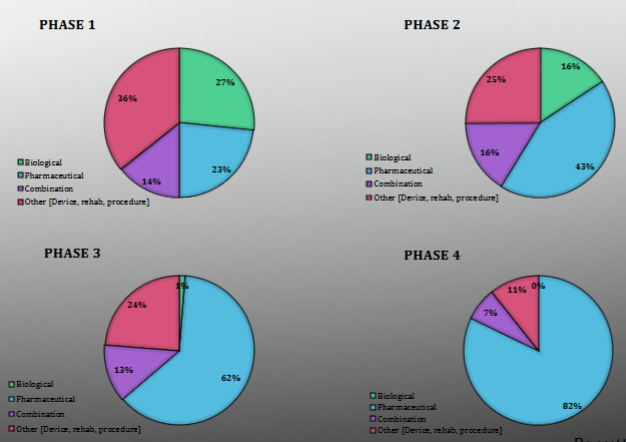
Discuss the distribution of SCI clinical trial intervention (depending on the stages)
Later phases of clinical trial intervention involve pharmaceutical companies more heavily as the focus has shifted from basic safety testing to larger scale production and commercialization of a product
Why might a SCI clinical trial be stopped? (4)
Safety → adverse events occur
Poor study design → low enrollment
Efficacy → results are insignificant
Commercial reasons → money is not enough
What are some commercial reasons for stopping SCI clinical trial + is it ethical to do so?
research budgets reduce
competitive products emerge
supply failures
pressure to terminate unproductive programs
What are some types of intervention for SCI? (5) + What is a combination intervention?
Drugs => reducing secondary damage after injury such as inflammation, pain, axon health
Stem cells => repair and replace damaged cells (regeneration & cell replacement)
Devices => exoskeletons used to support or restore physical abilities (rehabilitation)
Biomaterial => physically guiding axonal regrowth
Surgical => prevent further damage and relieve pressure
Effective intervention for SCI involve combination of different approaches since SCI is not caused by one problem
Discuss the Mend the Gap intervention + what type of intervention is it?
It is a proposed approach to spinal cord repair promoting axon regeneration => bridge the gap in spinal cord tissue
combination of
biomaterial injected into spinal cord injury site
drug delivered by biomaterials to eliminate scar tissue
surgical robot visualize tissue structure & guide the injection of biomaterials
Discuss the difference in values & priorities depending on severity of the SCI
Differ in their technology adoption behaviours
MAJORITY of people with chronic cervical SCI = late adopters
less willing to take risks
afraid to lose the current functions that they have
MAJORITY of people with chronic thoracic SCI = early adopters
higher tolerance to risk
see more gain from risky interventions

What is medical tourism - why are people seeking for it - what are the ethical implications
People traveling to other countries to receive medical interventions / healthcare services
Reasons:
cost savings
faster access / shorter wait times
access unapproved services in their home country
better expertise & quality
combine with tourism
Ethical implications
resource allocation → wealthy foreign patients are prioritized over local population
risk of complications when returning home
consent issues due to cultural differences
vulnerable to false → clinics offering treatments with limited scientific evidence
legal concerns across countries