Exam 3 NEED TO KNOW CSF BIOL 3040
1/89
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
90 Terms
CDK
cyclin dependent kinase - gets enzymatically activated when binds to cyclin
Cyclin
activates CDK
M-cyclin
Regulatory protein that binds to mitotic Cdk to form M-Cdk, the protein complex that triggers the M phase of the cell cycle.
the cyclin that acts in G2 to trigger entry into M phase
S cyclin
Regulatory protein that helps to launch the S phase of the cell cycle.
CDK helps initiate DNA synthesis
G1/S Cdk
Protein complex whose activity triggers entry into S phase of the cell cycle; consists of a G1/S cyclin plus a cyclin-dependent protein kinase (Cdk).
S-Cdk
Protein complex whose activity initiates DNA replication; consists of an S cyclin plus a cyclin-dependent protein kinase (Cdk).
CAK
CDK- activating kinase phosphorylates an activating site in Cdks
Wee1 kinase
phosphorylates inhibitory sites in Cdks; primarily involved in suppressing Cdk1 activity before mitosis
Cdc25 phosphatase
activates CDKs
removes inhibitory phosphates from Cdk1 so cyclin B can bind to activate mitosis
opposes Wee1
PP2A-B55
PP2A-B56
PP2A
Heterotrimer phosphatase: catalytic + 2 regulatory subunits. Regulated by phosphorylation, methylation. Substrate serine-threonine
Protein phosphatase 2A
regulatory subunit is SPECIFICITY DEPENDENT
PP1
protein phosphatase 1
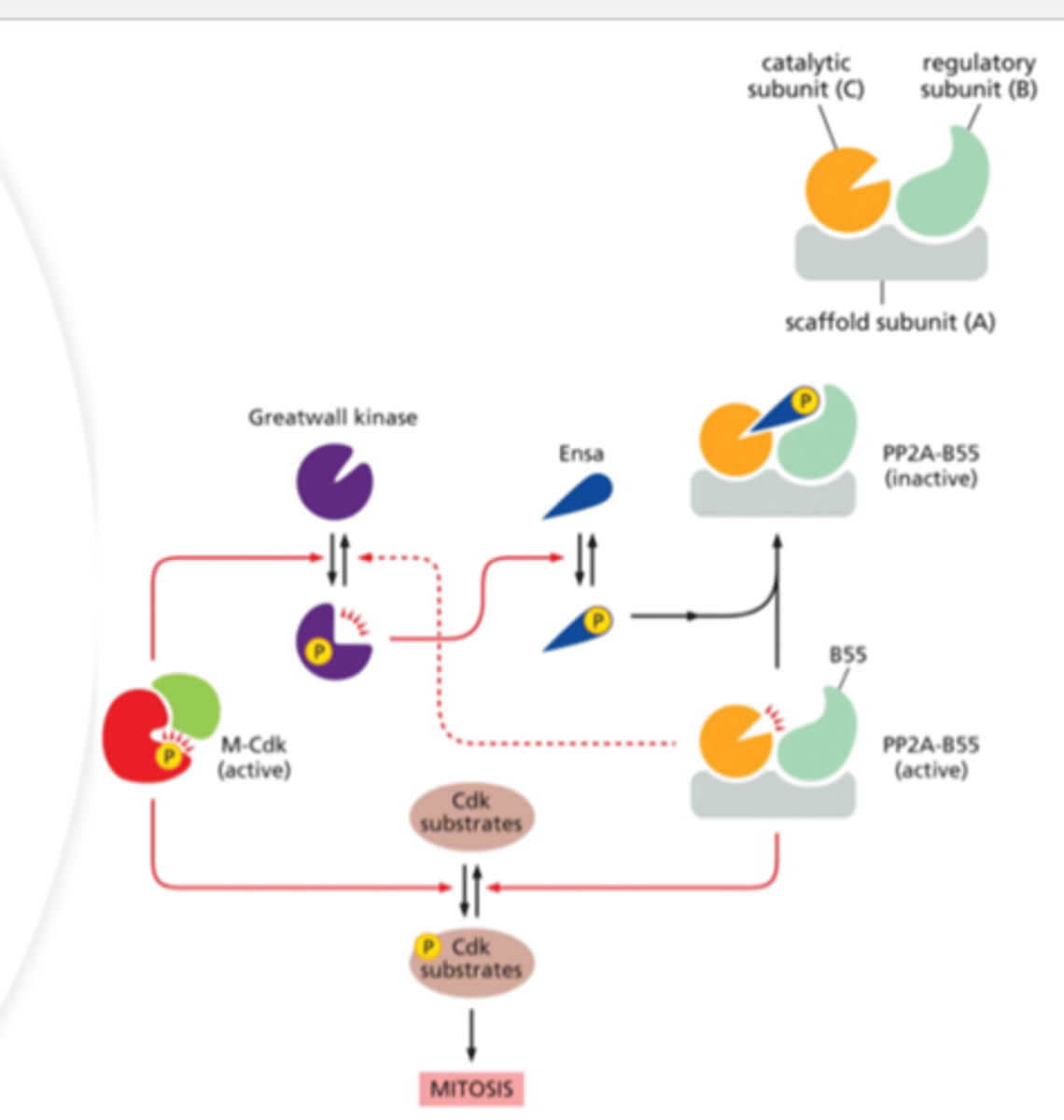
Greatwall Kinase
phosphorylates Ensa
phosphorylates
Ensa
inactivates PP2A-B55
phosphorylates
Protein Phosphate CDK Regulatory Pathway
M-Cdk
Protein complex that triggers the M phase of the cell cycle; consists of an M cyclin plus a mitotic cyclin-dependent protein kinase (Cdk).
p27
binds to cyclin (cdk) to block entrance of cell to the S phase of cell cycle
protein that binds to cyclin and cdk blocking entry into S phase
inhibitor
p21
Cdk inhibitor protein
cell cycle regulatory protein that inhibits the cell cycle; its levels are controlled by p53
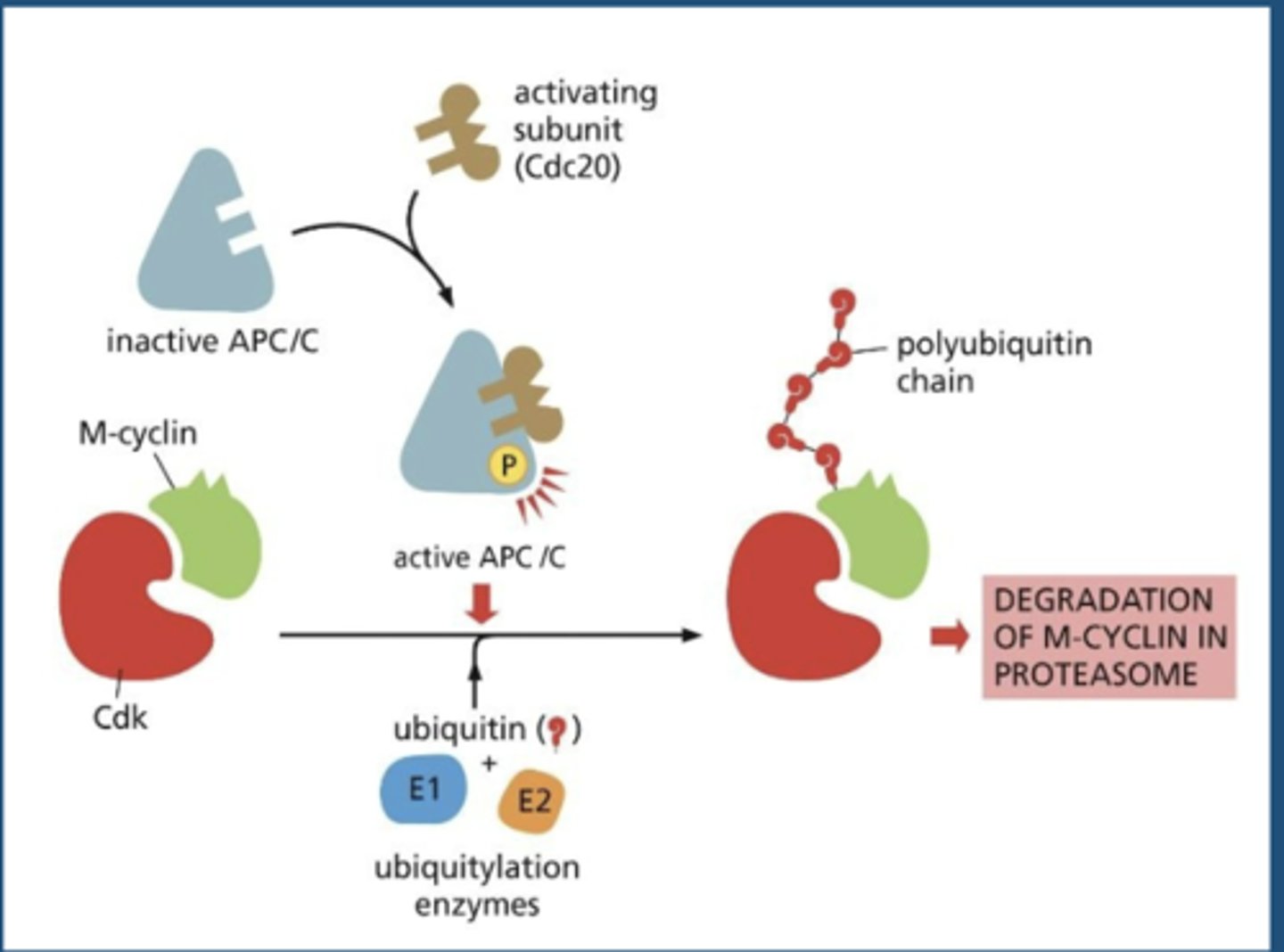
APC/C
the ubiquitin ligase that promotes the destruction of a specific set of proteins, thereby promoting the separation of sister chromatids and the completion of M-phase
anaphase-promoting complex/cyclosome
Cdc20
activates APC
triggers initial activation of APC/C at metaphase to anaphase transition - stimulated by M-CDK
Cdh1
Activating subunit which binds and activates APC/C in late mitosis through early G1
APC/C-activating subunit that maintains APC/C activity after anaphase and throughout G1; inhibited by Cdk activity
KNOW THIS GRAPH!
GEF
guanine nucleotide exchange factor - ON = GTP
GAP
GTPase activating protein - OFF = GDP
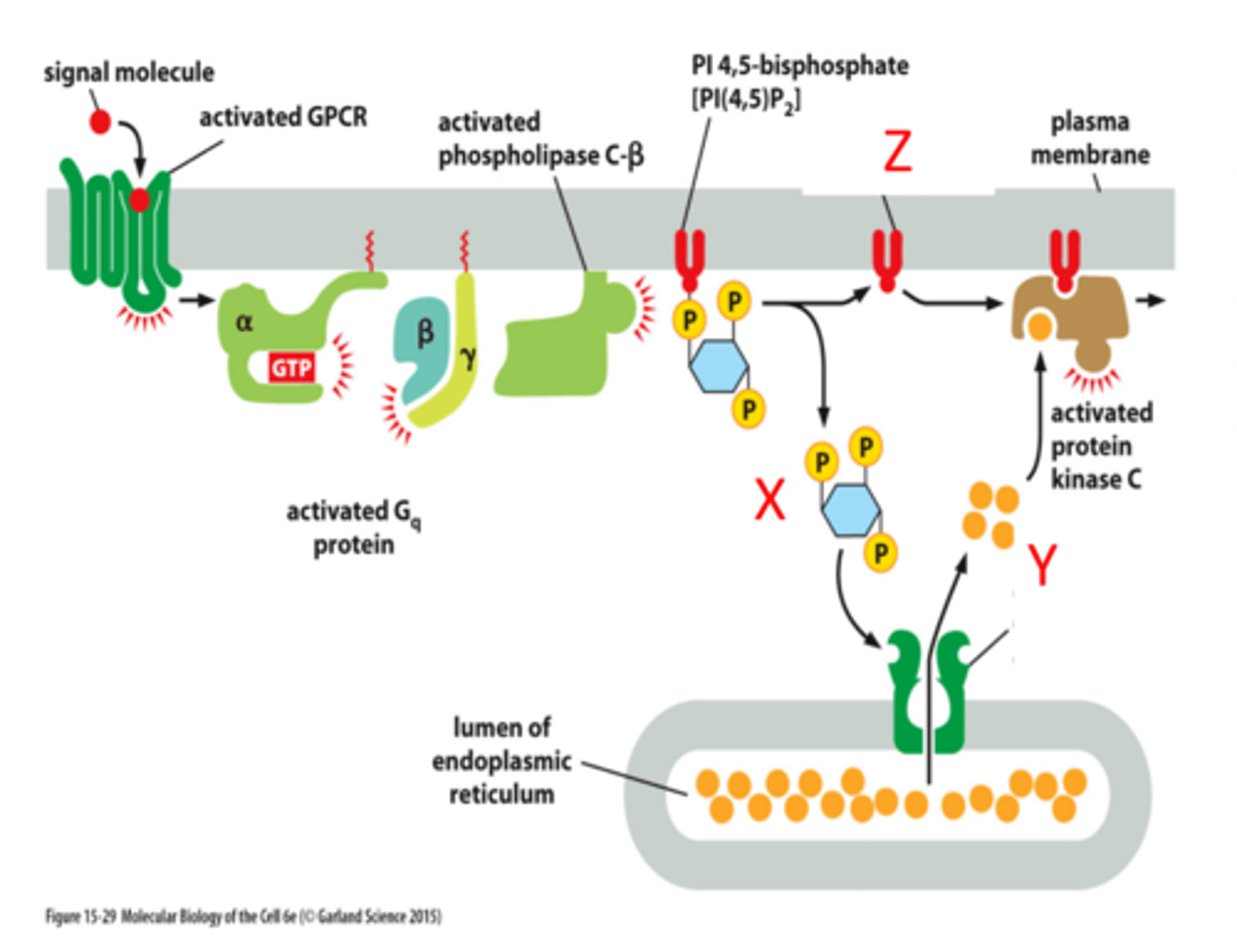
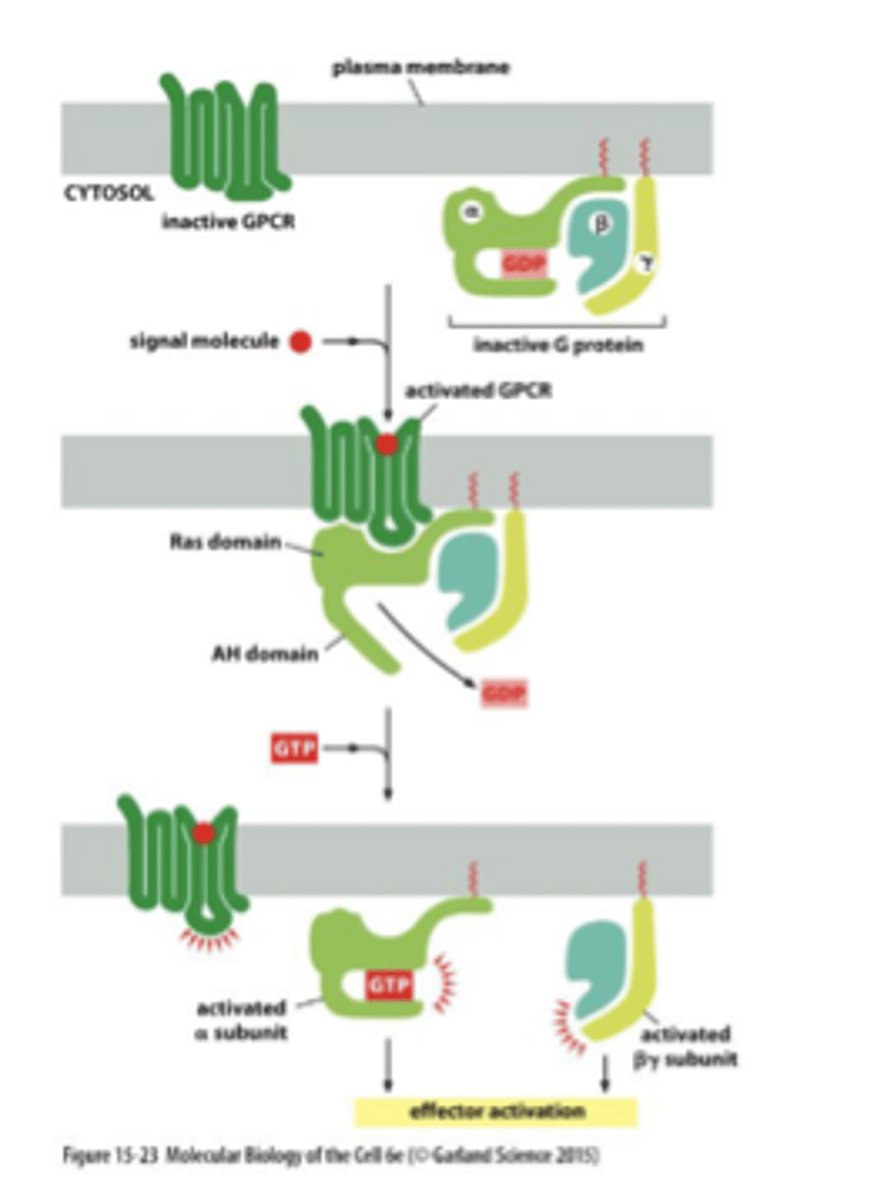
GPCRs
-G Protein Coupled Receptors: membrane proteins involved in signal transduction
-binding of an intracellular G protein turns on an active site to affect an intracellular pathway
3 kinds: Gs, Gi, Gq
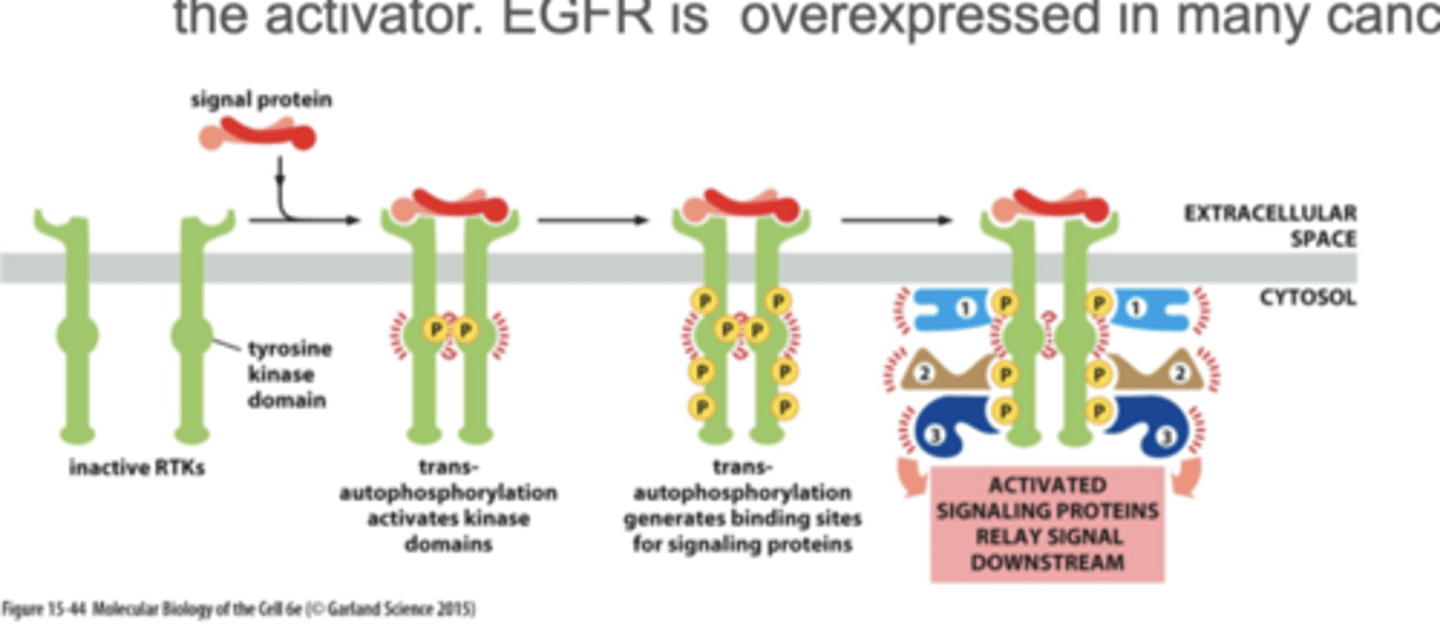
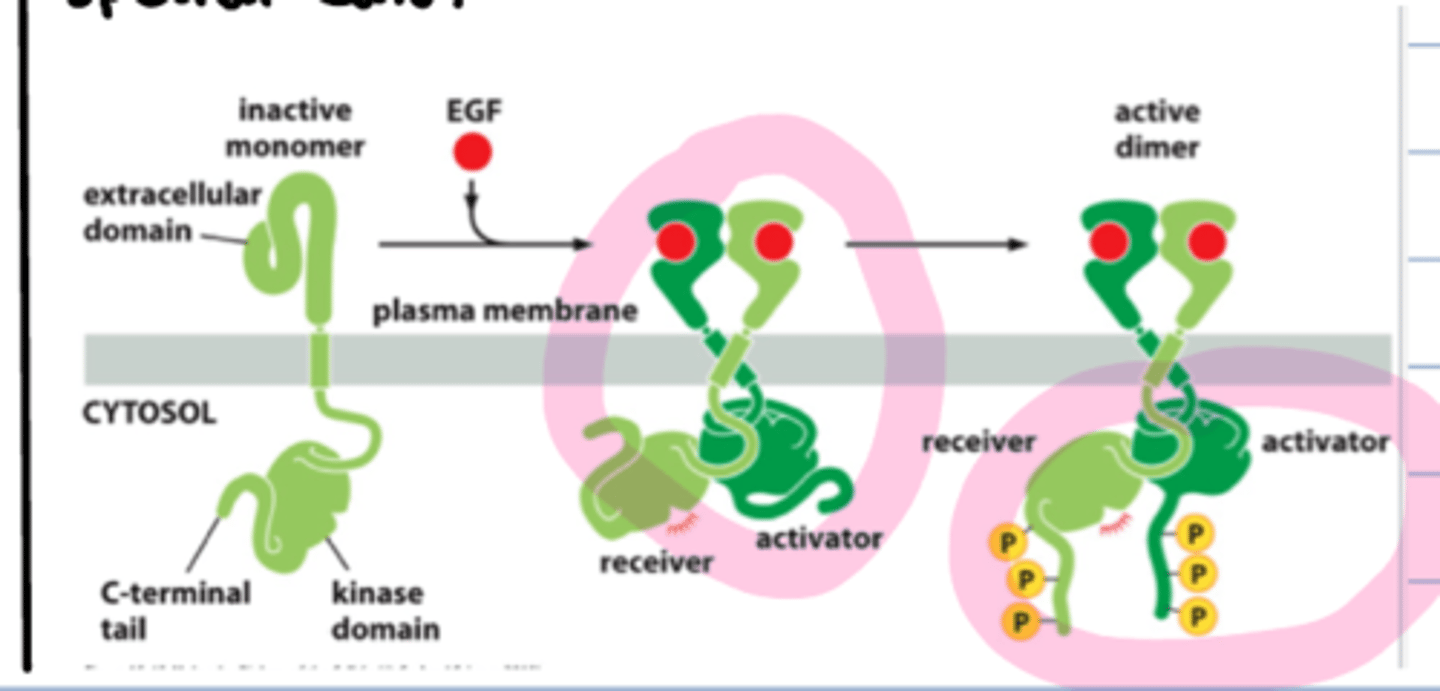
activated RTK - receptor tyrosine kinase
upon binding to RTKS, monomers can form dimers and phosphorylated each other
PLC-beta pathway
X= IP3
Y= Ca2+
Z= Dag
EGF binding
different than receptor tyrosine kinase
GPCR binding
Three G-proteins (alpha, beta, and gamma) associate with the GPCR but only the G-alpha binds to the GTP after receptor activation.
binds on the cytosolic side
adenylyl cyclase
an enzyme that converts ATP to cAMP in response to an extracellular signal
activates PKA
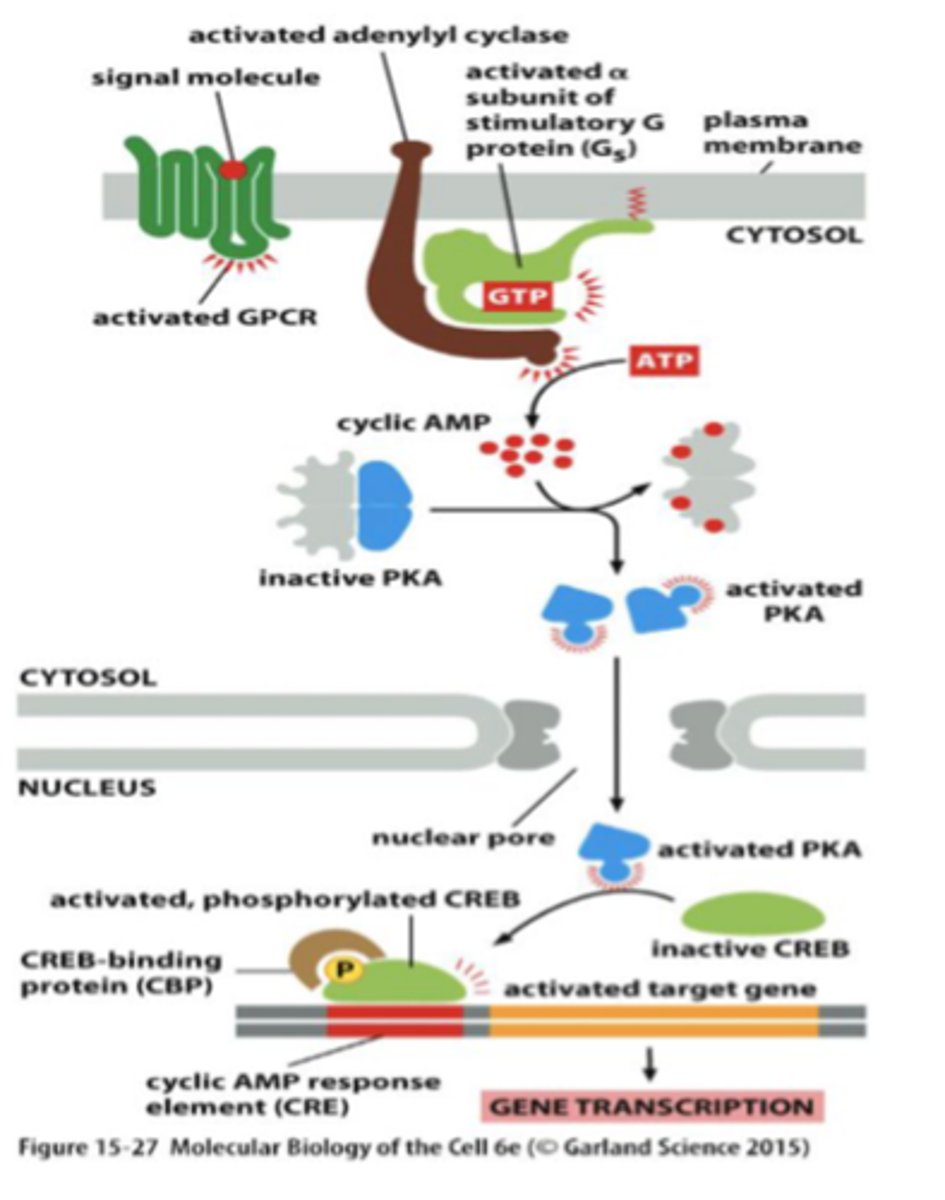
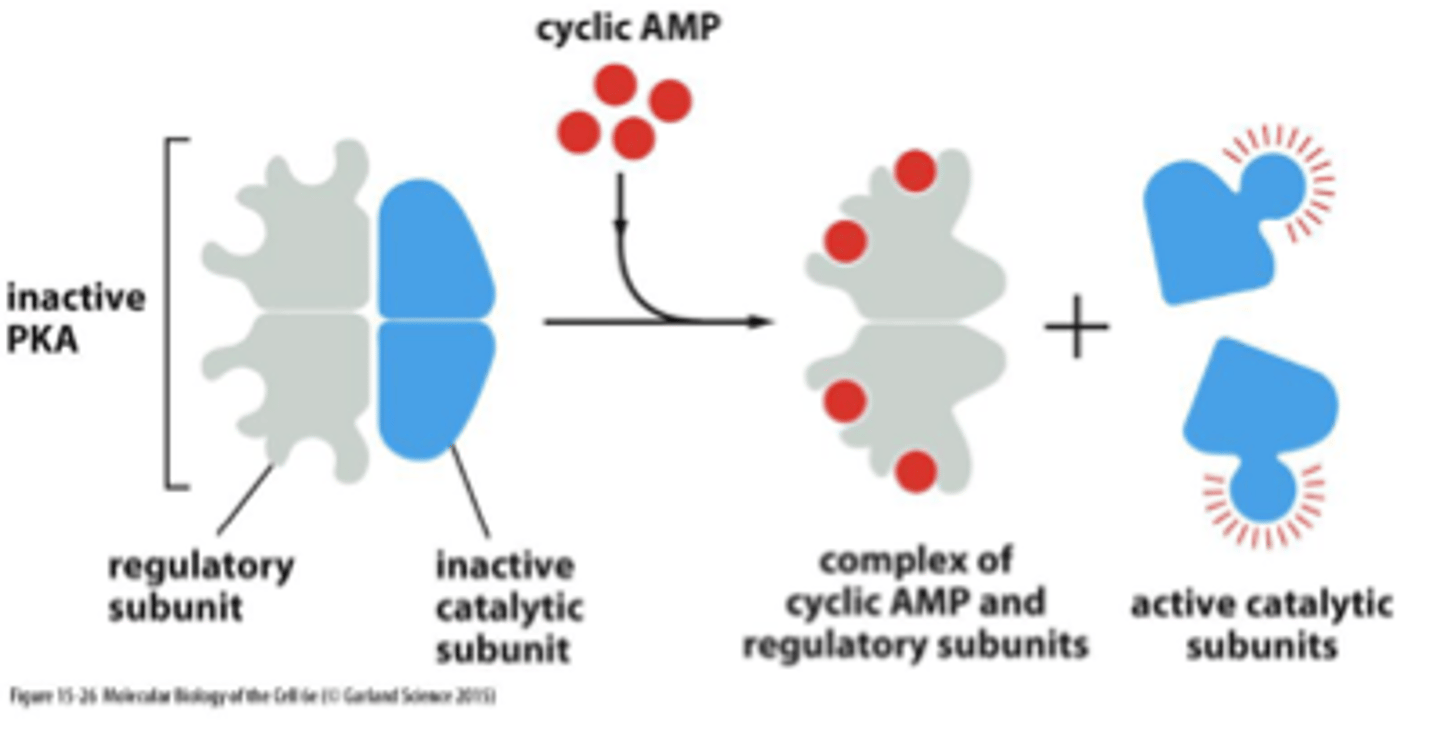
cAMP
cyclic adenosine monophosphate
binds to PKA and activates it - binds to the regulatory subunit of PKA and that releases the active catalytic subunits
activates protein kinase A
Molecular interaction domains
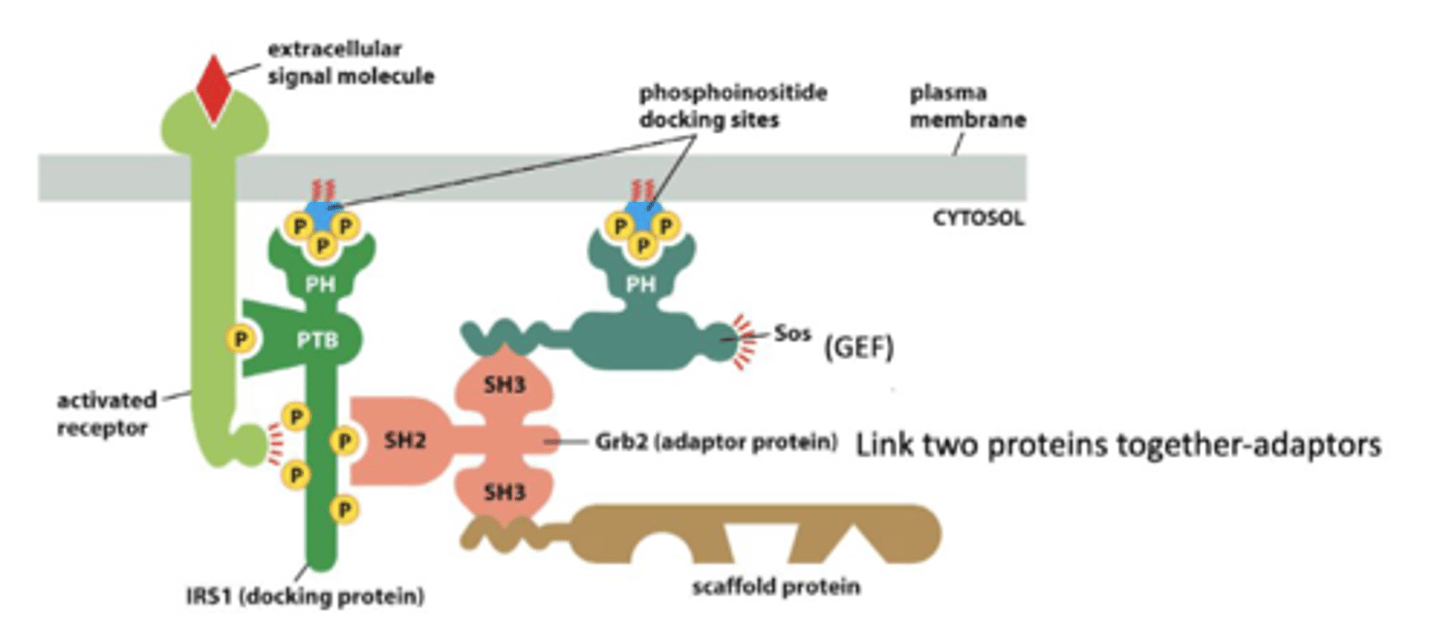
PTB domain
phosphotyrosine binding domain
SH2 domain
A protein domain that binds phosphorylated tyrosine residues.
SH3 domain
binds proline rich sequences
Pleckstrin homology (PH) domain
binds to inositol phospholipids
phospholipase C
G-protein linked receptor signals phospholipase C
splits PIP into IP3 and DAG:
1. IP3 increases intracellular Ca2+
2. DAG activates kinases (PKC) to activate enzymes
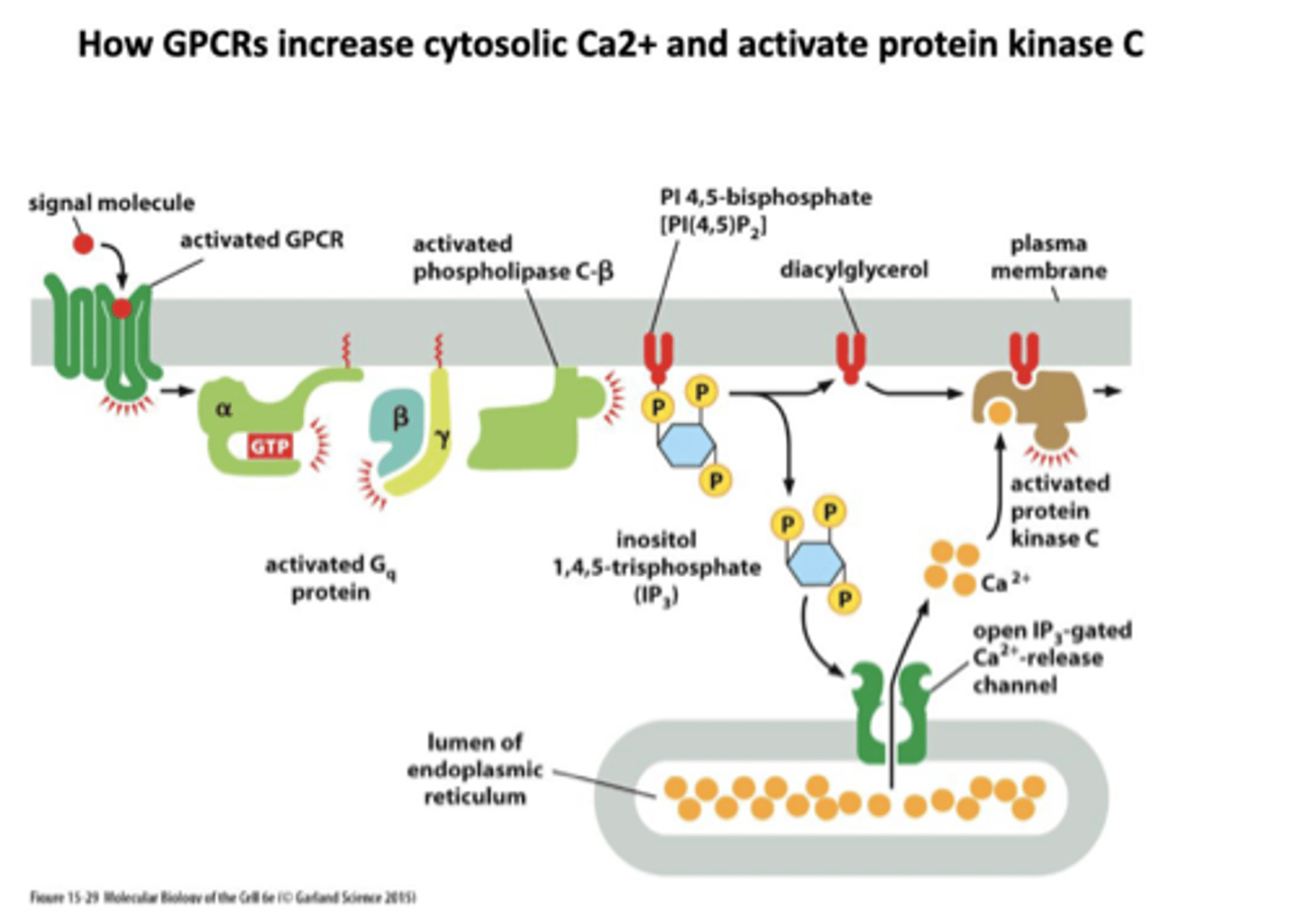
IP3
One of the products of PIP2 cleavage in the Ca++/phosphoinositide signal pathway.
Moves to cytoplasm to trigger Ca++ release from intracellular stores leading to Ca++/calmodulin cascade.
DAG
diacylglycerol
Another of the products of PIP2 cleavage
activates protein kinase C - PKC
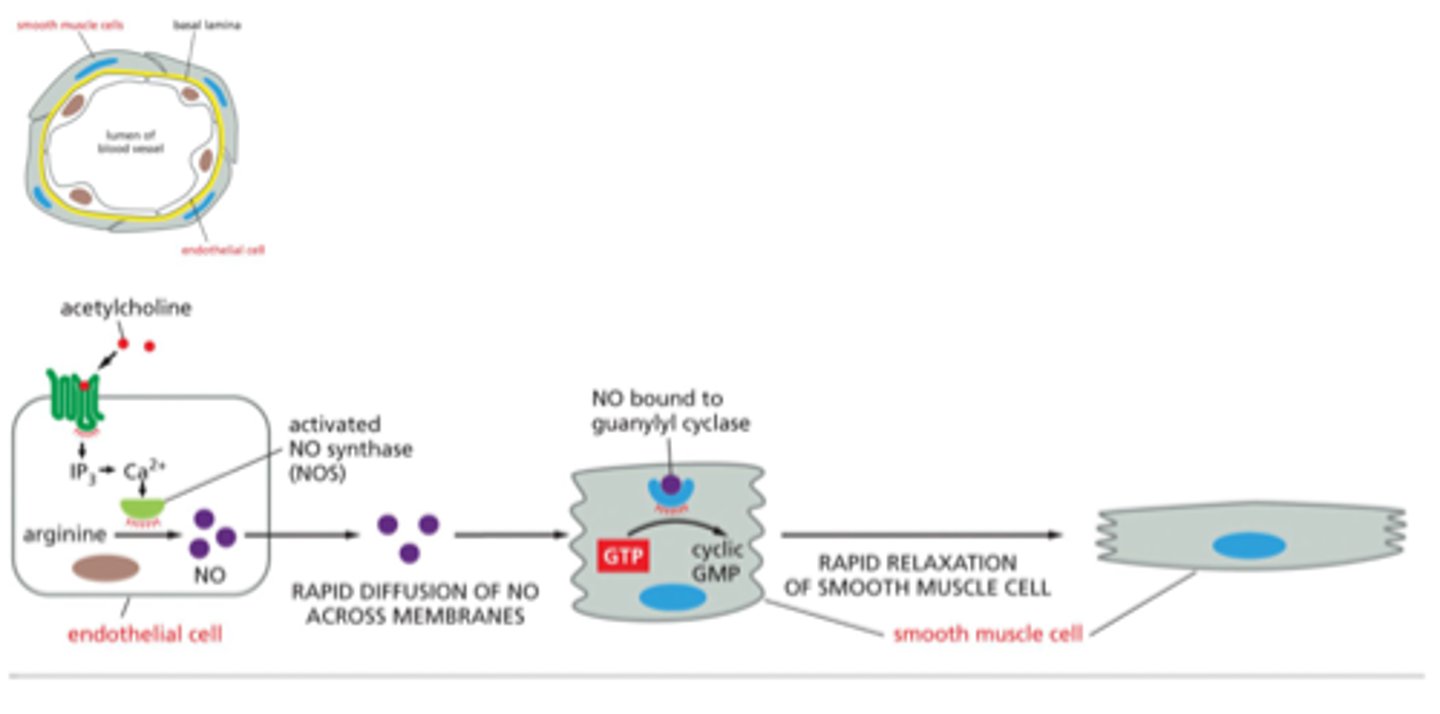
guanylyl cyclase
enzyme that catalyzes transformation of GTP to cyclic GMP
allows for muscle relaxtion
nitric oxide binds to guanylyl cyclase to make cGMP
GPCR desensitization
Receptor desensitization prevents sustained activation that could be harmful to cells
gets phosphorylated and then arrestin binds and ARRESTS
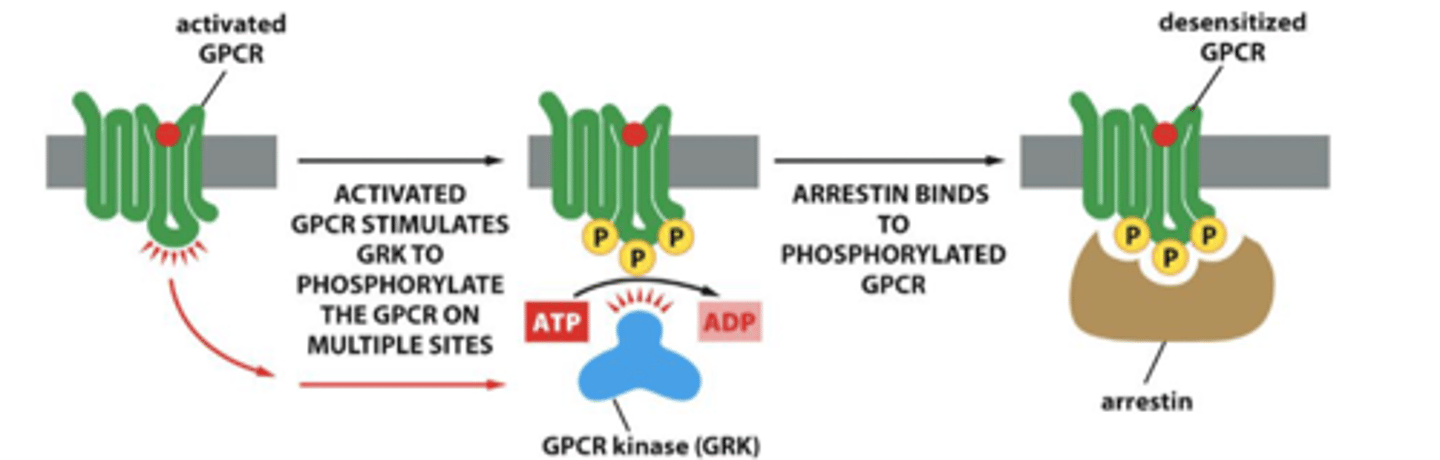
arrestin
proteins that bind phosphorylated GPRCs and deliver them to clathrin coated pits for endocytosis
enzyme that participates in desensitization of GPCRs by phosphorylating them after they have been activated by ligand binding
binds to GPCR so G proteins cannot interact w GPCR
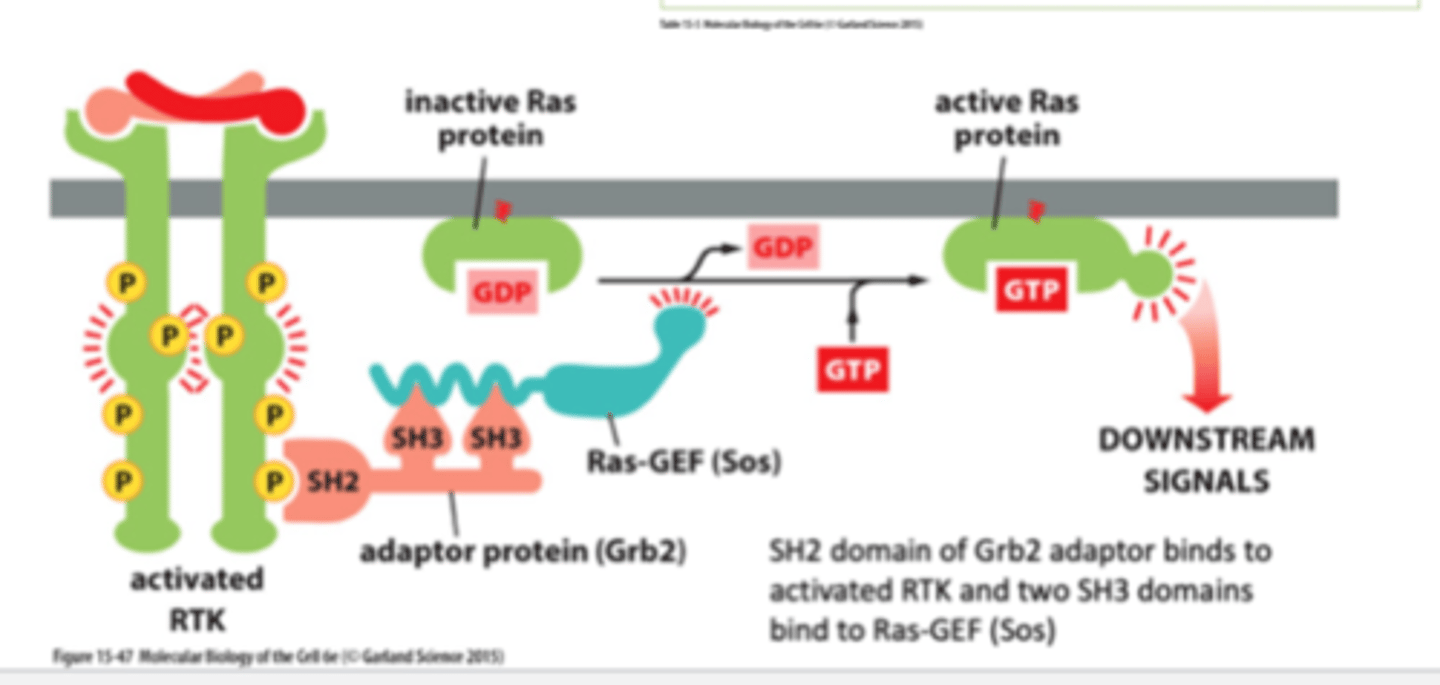
RTK activation of Ras
RTK is activated through autophosphorylation.
Adaptor proteins bind to the RTK which requires SH2 domains, which then binds SOS (Ras Guanosine Exchanger factor GEF), which promotes the exchange of GDP to GTP. Once GTP is bound, Ras is activated.
1. signal molecule binds and activates RTK - autophosphorylation
2. An adaptor protein (Grb2) docks on a particular phosphotyrosine on the phosphorylated RTK
3. the adaptor recruits a Ras guanine nucletide exchange factor (Ras-GEF) (Sos) that stimulates Ras to exchange its bound GDP for GTP (Ras is activated when in GTP form)
4. Activated Ras can now stimulate downstream signalling pathways→ex: cell division pathways
What can Ras signaling do?
activate MAP kinase
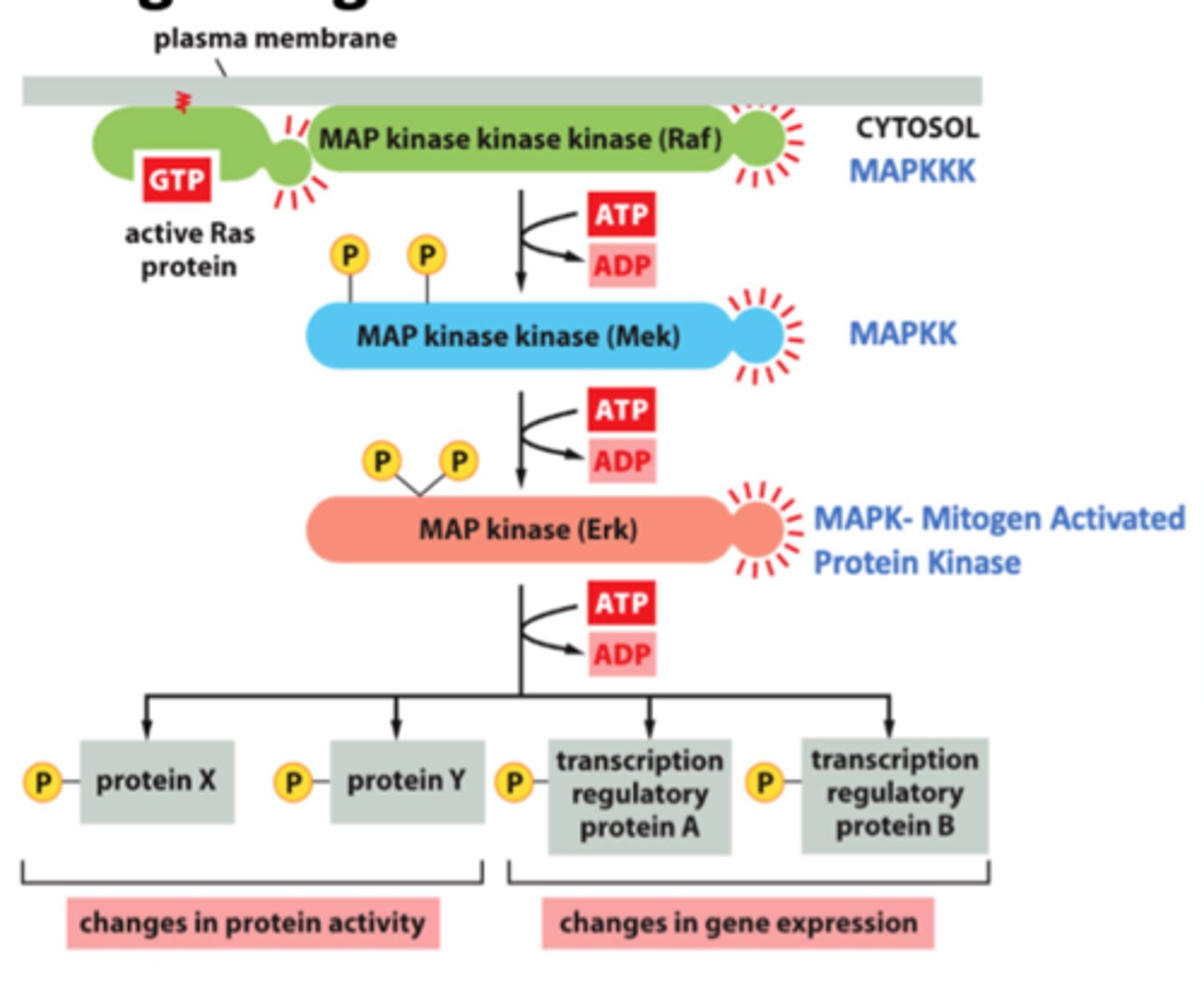
MAP kinase kinase kinase
MAPKKK
Raf
Phosphorylates and activates MAP-kinase-kinase.
- ATP → ADP
MAP kinase kinase
MAPKK
Mek
Phosphorylates and activates MAP-kinase.
- ATP → ADP
MAP kinase
MAPK
Erk
Mitogen-activated protein kinase. Protein kinase that performs a crucial step in relaying signals from cell-surface receptors to the nucleus.
It is the final kinase in a three-kinase sequence called the MAP kinase cascade.
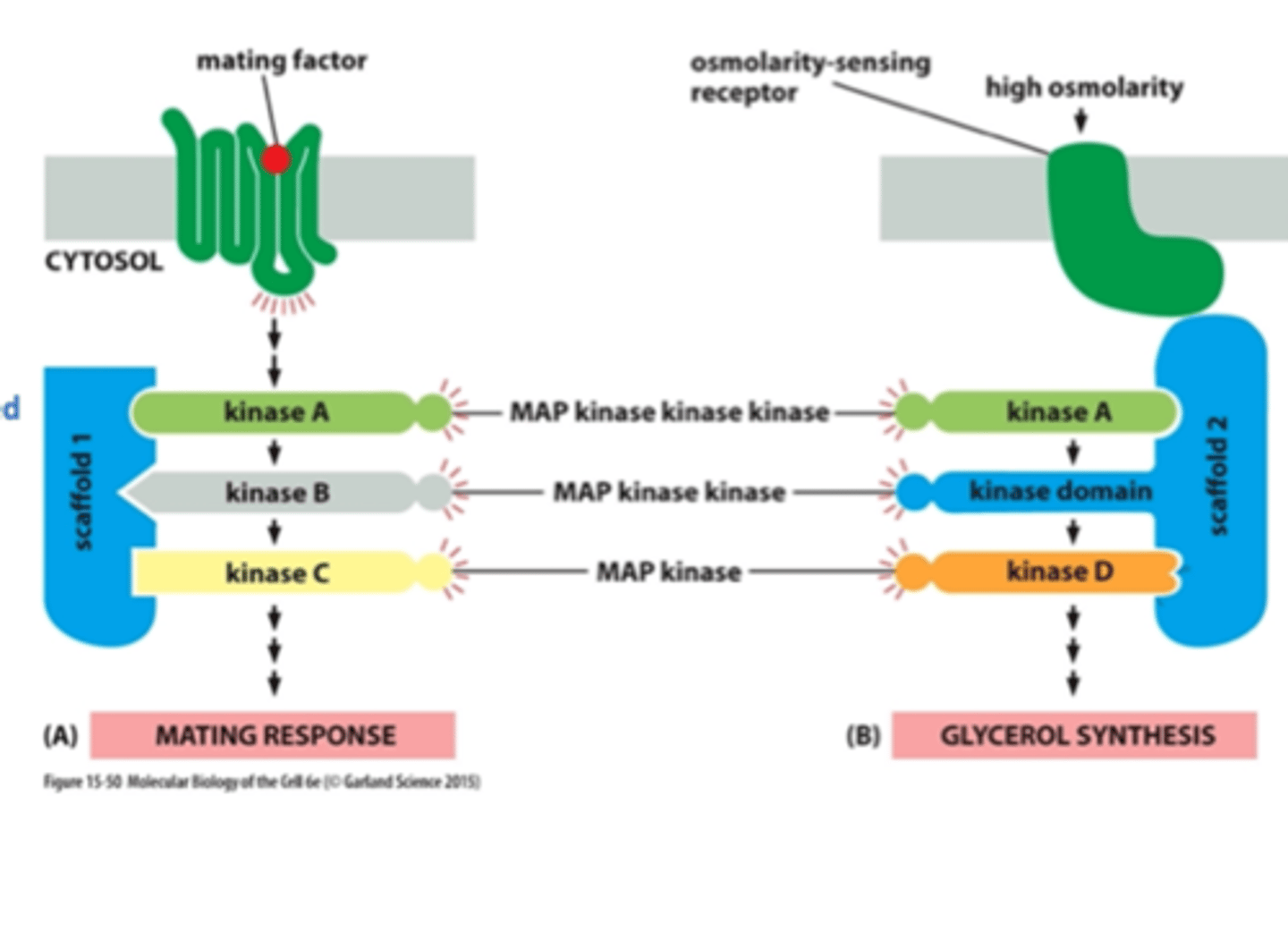
scaffold proteins
organize groups of intracellular signaling molecules into signaling complexes
improve the efficiency of a signaling cascade by holding all the participating enzymes in close proximity
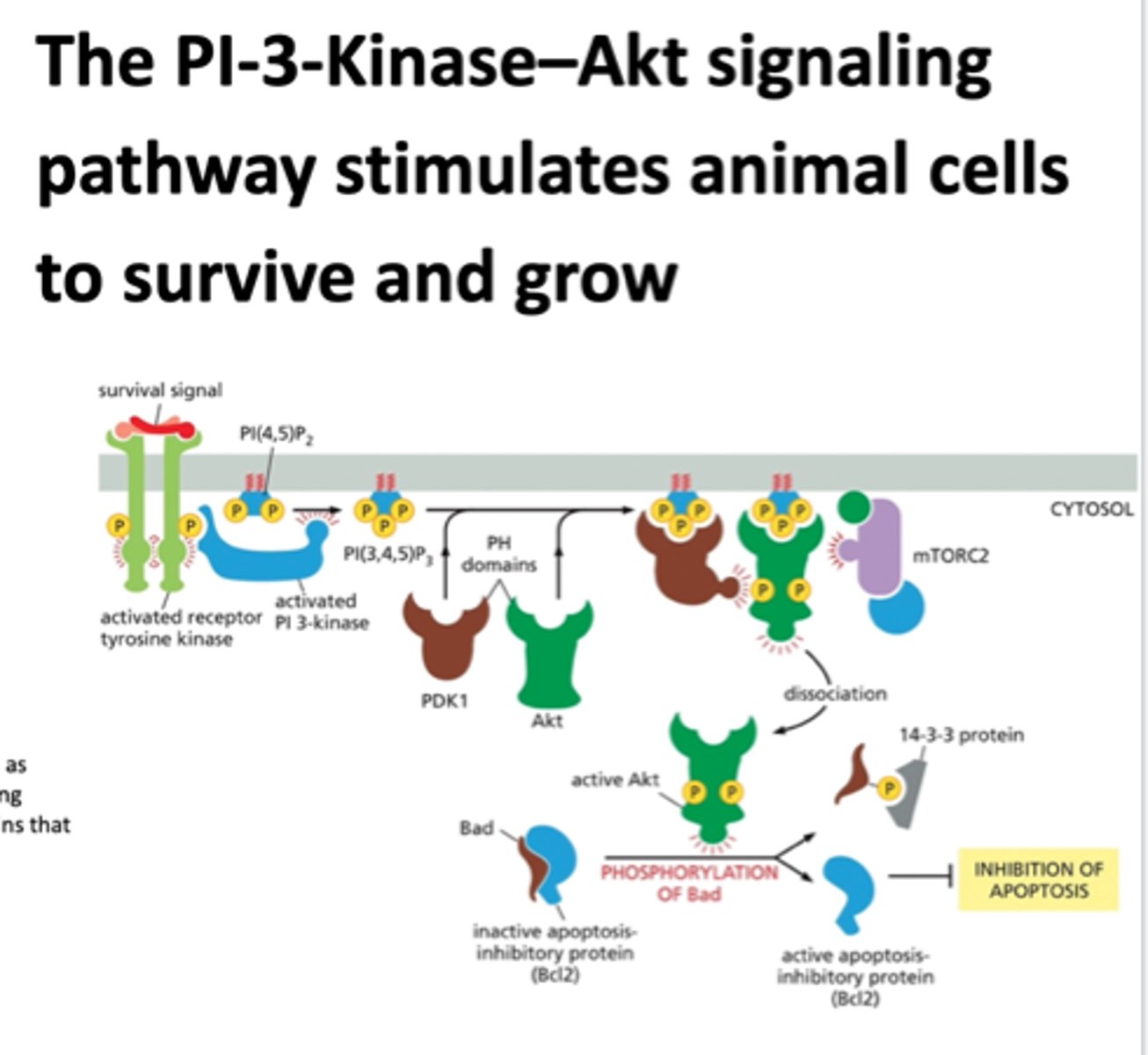
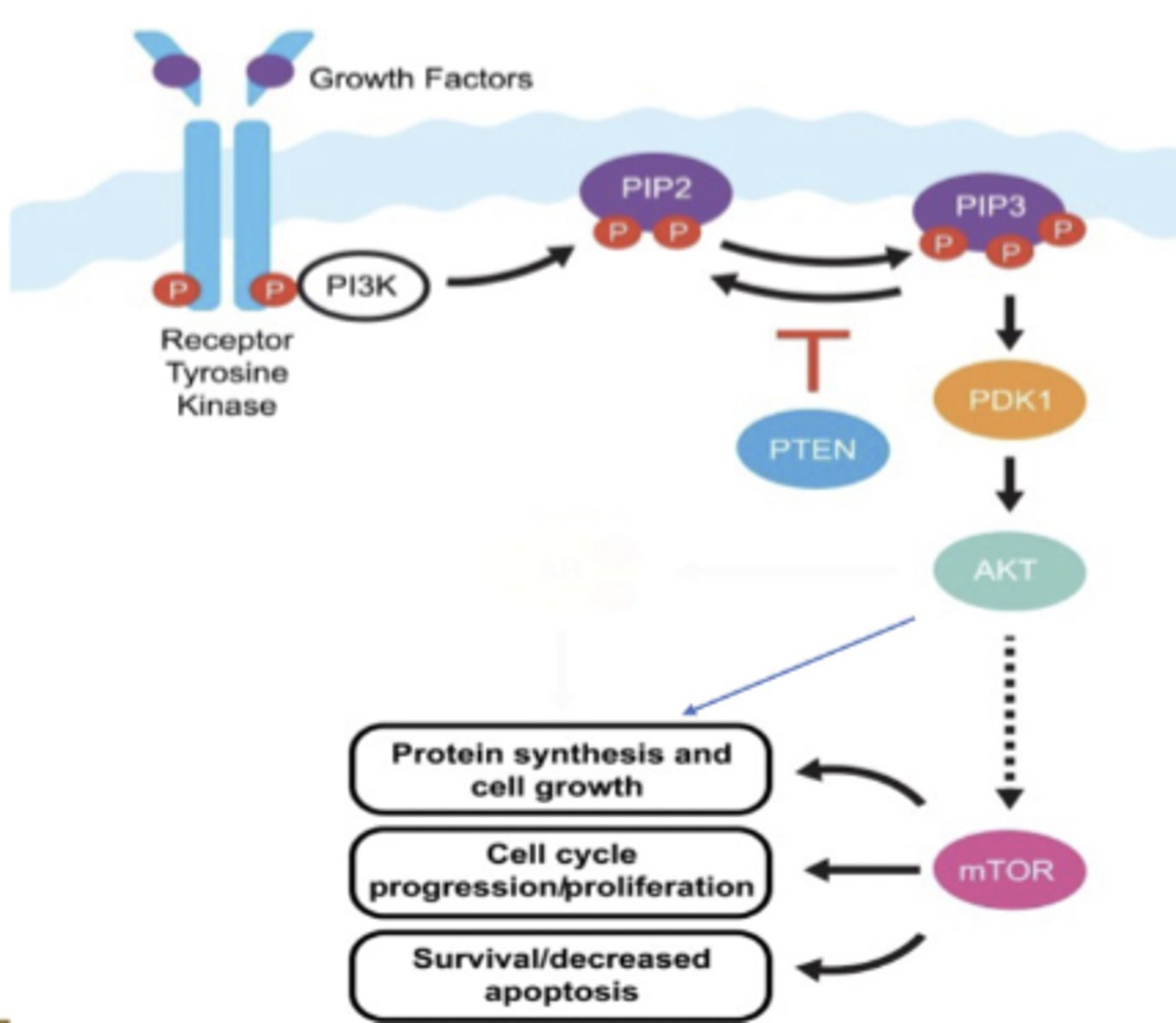
PI-3-kinase-Akt signaling pathway
this promotes cell survival and it is activated by a RTK.
phosphorylates an inositol phospholipid that is embedded in the cytosolic side of the plasma membrane. this attracts intracellular signaling proteins that have a special domain that recognizes them.
Akt is a protein kinase that is released from the plasma membrane and phosphorylates various downstream proteins on specific serines and threonines.
this promotes cell survival
PDK1
a kinase that phosphorylates another kinase Akt to become active
Akt
A serine/threonine kinase that is activated by PIP3 and plays a key role in signaling cell survival.
Bad protein
promotes apoptosis
inactivates apoptosis inhibitory protein
mTORC2
a protein complex that regulates cellular metabolism as well as the cytoskeleton
What can active Akt do?
can phosphorylated bad, allowing for an active apoptosis inhibitory protein, blocking apoptosis so NO CELL DEATH HOORAY
Bcl2
inhibits apoptosis
PI 3-kinase
phosphorylates PIP2 to PIP3
Enzyme that phosphorylates inositol phospholipids in the plasma membrane, which generates docking sites for intracellular signaling proteins that promote cell growth and survival.
PI-3 Kinase-Akt-mTORC1 signaling pathway
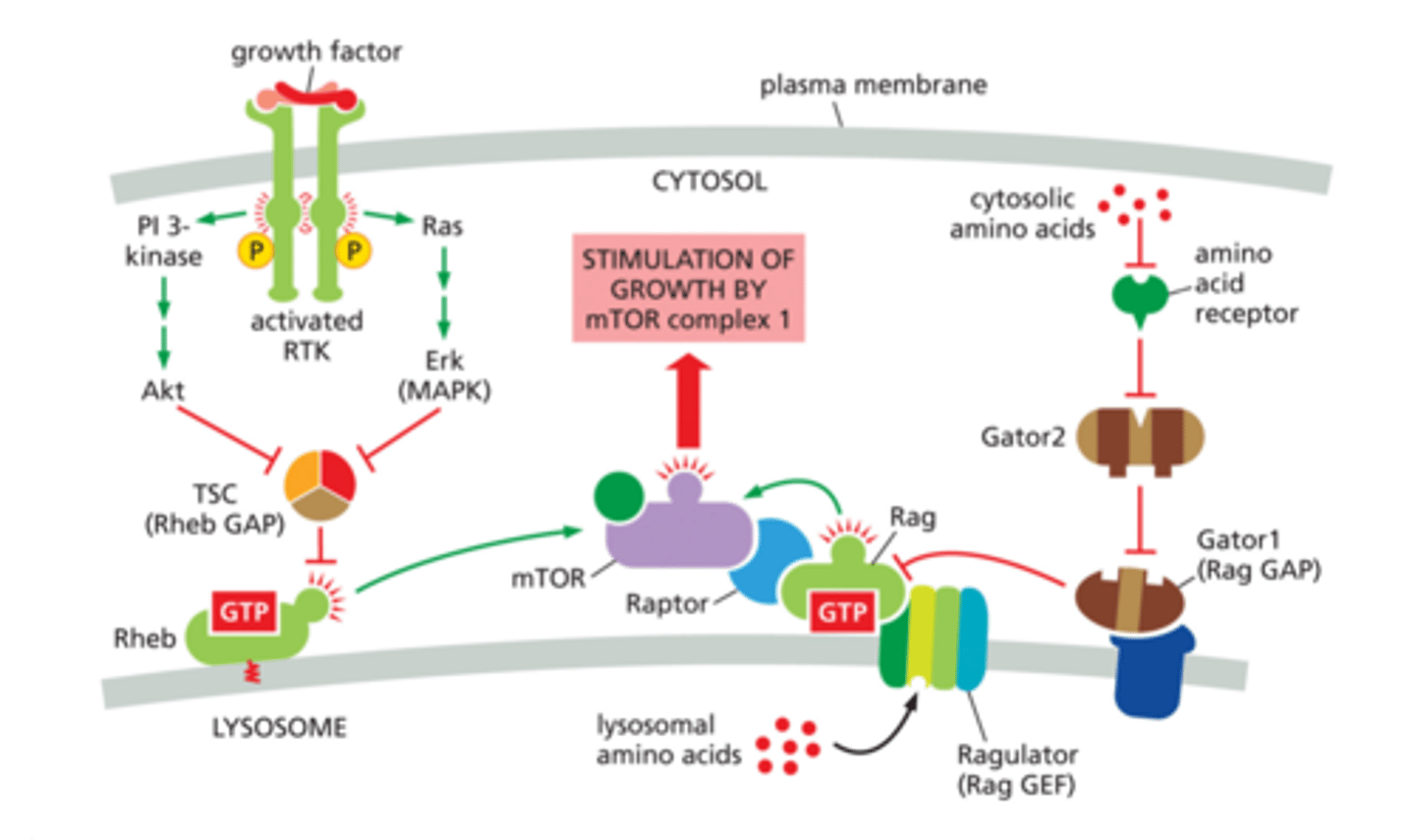
TSC
Rheb GAP - OFF
Rheb
Ras homolog that activates mTORc1
A monomeric Ras-related GTPase that in its active form (Rheb-GTP) activates mTOR, which promotes cell growth.
mTOR
mammalian target of rapamycin
mTORC1
Mechanistic target of rapamycin complex 1, kinase
Increases translation therefore, increases protein synthesis
Raptor protein
part of the MTORC1 COMPLEX
Rag
binds to GTP and raptor and mTOR making complex mTORC1!
Ragulator
Rag GEF - ON!
Gator1
Rag GAP - OFF!
Gator2
inhibits Gator1 allowing mTORC1 complex to be made and activated
Wnt/beta-catenin signaling pathway
without Wnt, beta catenin becomes unstable, and transcription DOES NOT occur
WITH Wnt signaling, beta catenin is stable YAY, and transcription DOES OCCUR!
with APC mutations beta catenin is always stable and results in UNCONTROLLED GROWTH AND POLYPS
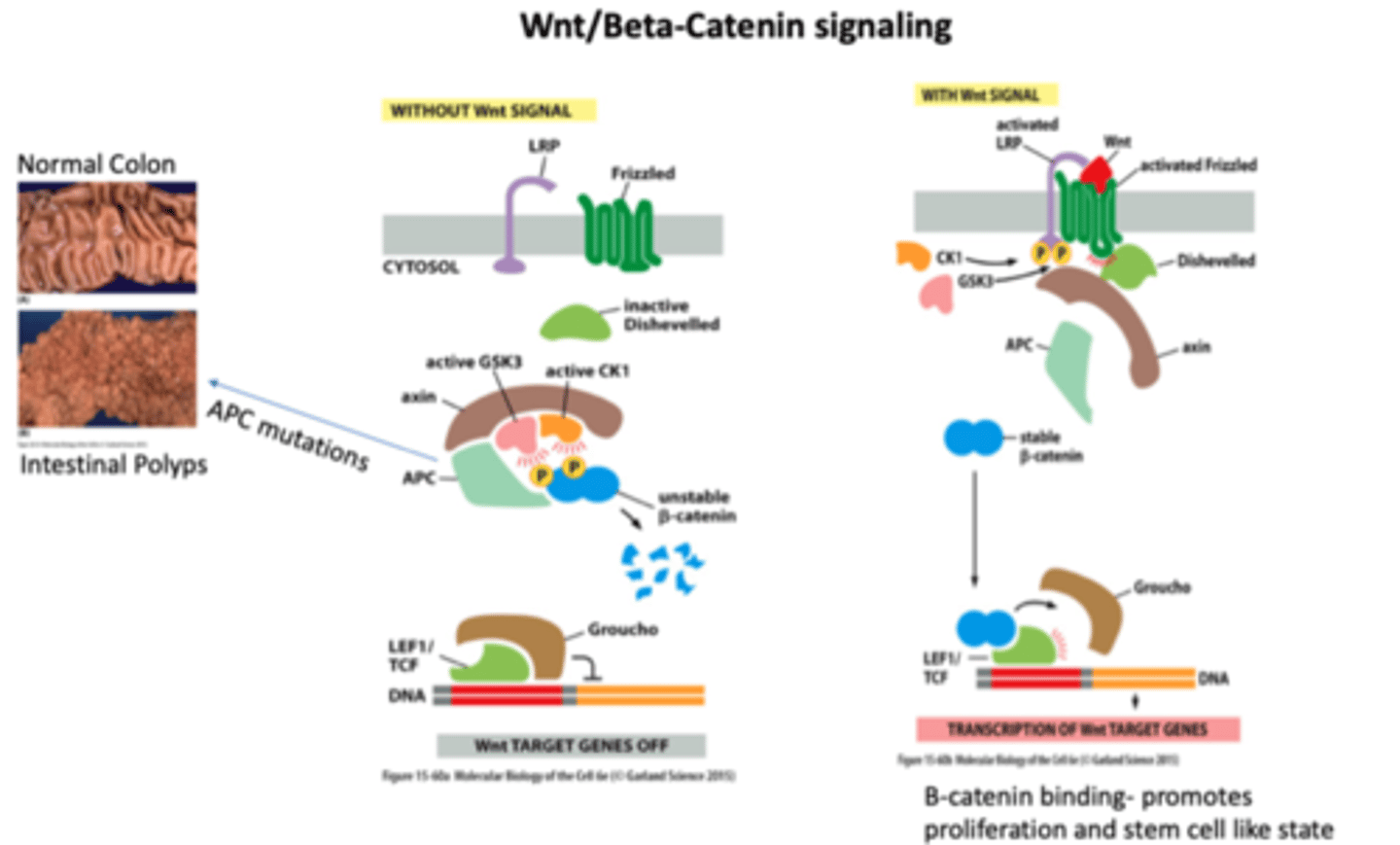
Wnt
Local signaling protein; activates receptors of the Frizzled family
regulates cell proliferation
soluble factor
resting cells: NO WNT; B catenin complexed with APC and gets destroyed
WNT stimulation deactivates destruction complex, increases beta catenin intracellular, which goes to nucleus and binds TCF, activates cell cycle genes and causes proliferation
Frizzled
receptor in the Wnt pathway
Groucho
protein that binds to LEF1/TCF and does NOT allow for transcription
LEF1/TCF
gets activated by beta catenin binding when its stable to bind
groucho gets kicked off
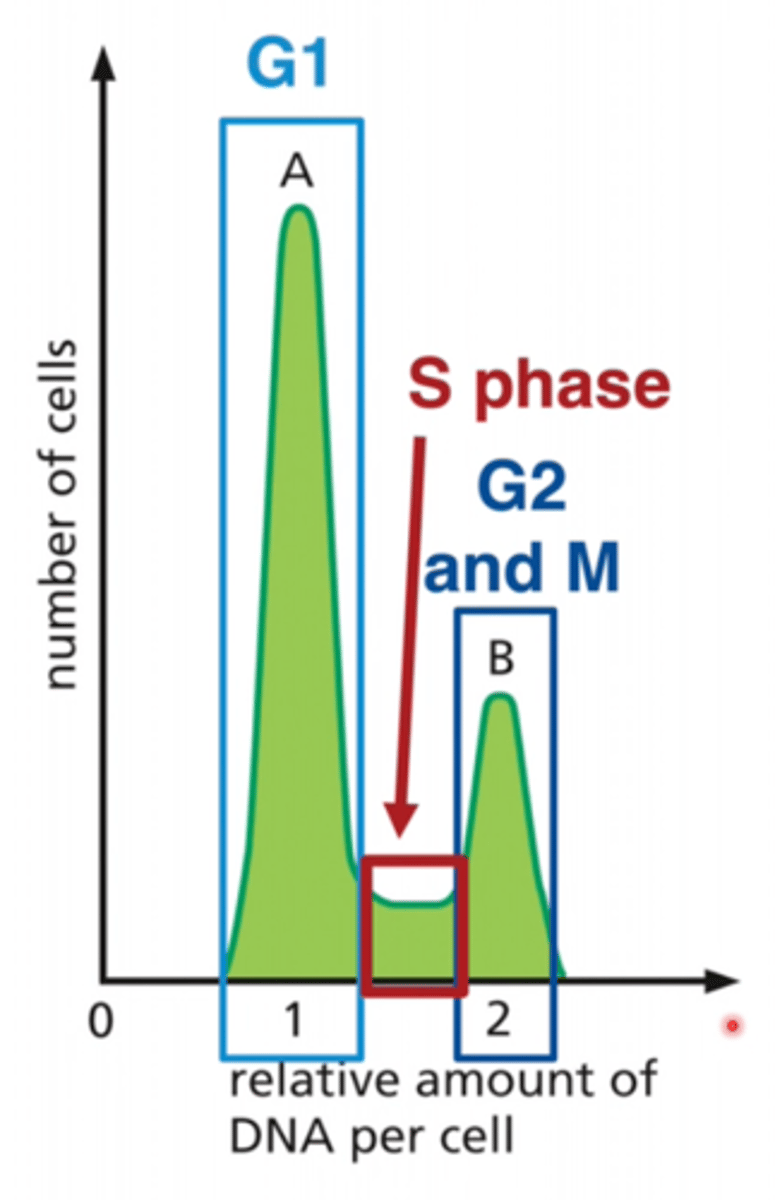
BrdU
A chemical that identifies newly-divided cells S PHASE
Flow cytometry
looking at number of cells expressing markers
ex: separate T cell populations using CD4/CD8 markers
method:
1. cells are stained with antibodies linked to fluorescent tag
2. cells passed through beam of light one at a time
3. light scattering and light emission is measured
4. fluorescence-activated cell sorting (FACS)- sort cells based on expression of markers
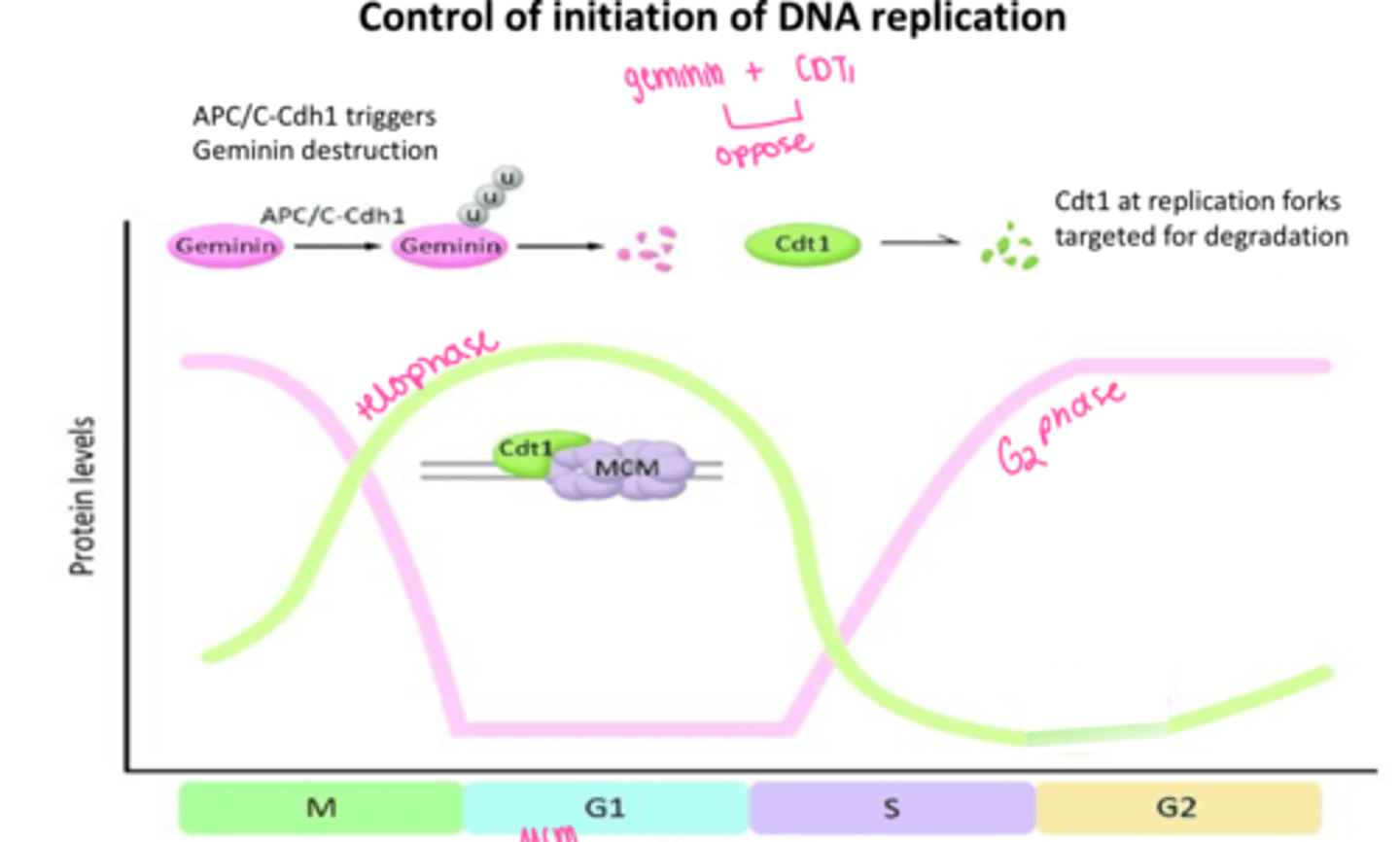
LOOK AT GRAPH
geminin
inhibits cdt1
Cdt1
Controls the timing of DNA replication
opposes geminin
Cell cycle control system
A cyclically operating set of proteins that triggers and coordinates events in the eukaryotic cell cycle.
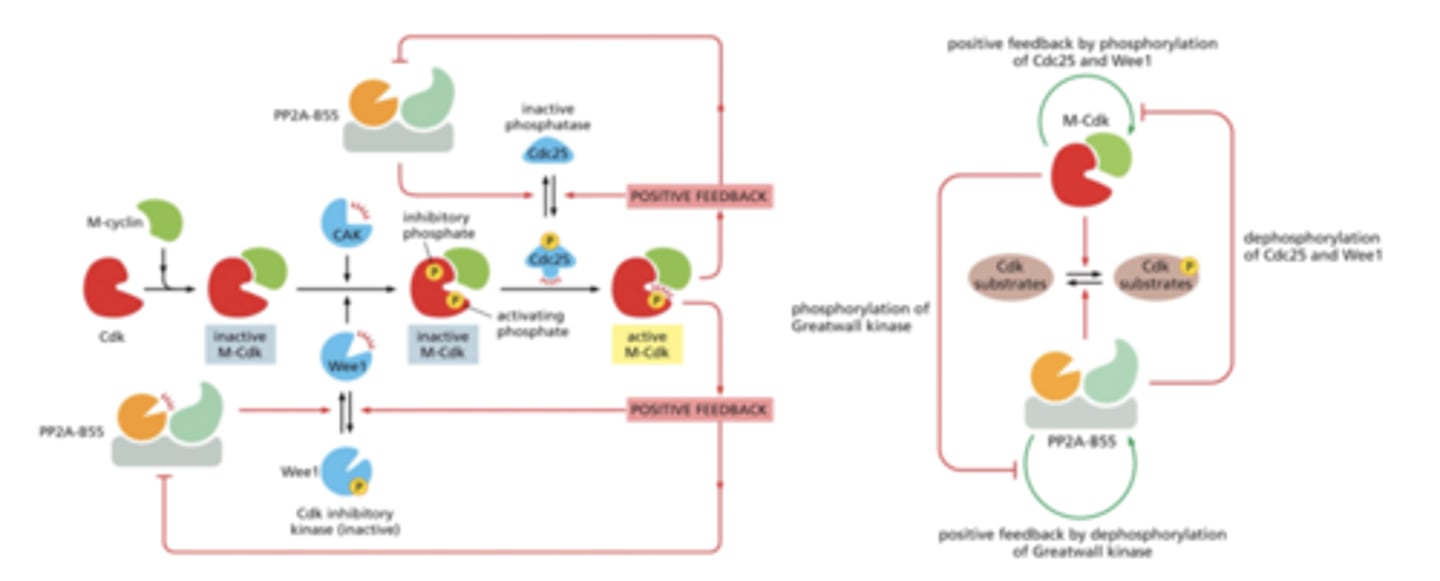
APC/C
anaphase-promoting complex/cyclosome
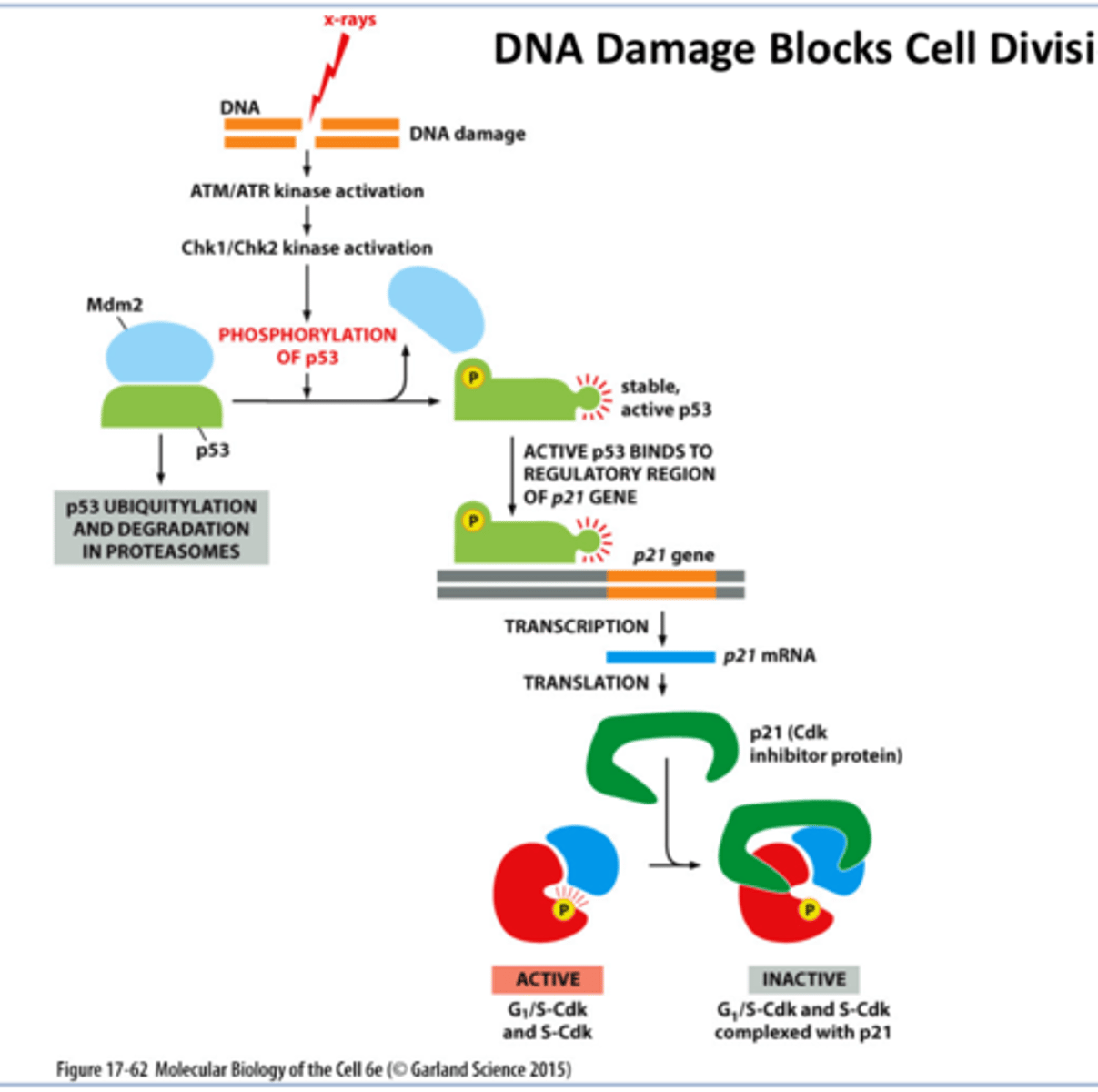
DNA Damage p53 and p21 response pathway
DNA damage
activates ATM/ATR
Chk1/Chk2 activation
that phosphorylates p53 releasing Mdm from p53
^^^Mdm degrades p53
then there is stable p53
this binds to p21 gene yay
that allows for transcription
then translation p21 cdk inhibitor
inhibits Cdk complex
• DNA damage can temporarily halt progression through G1 phase
• p53- Checkpoint protein and a tumor suppressor
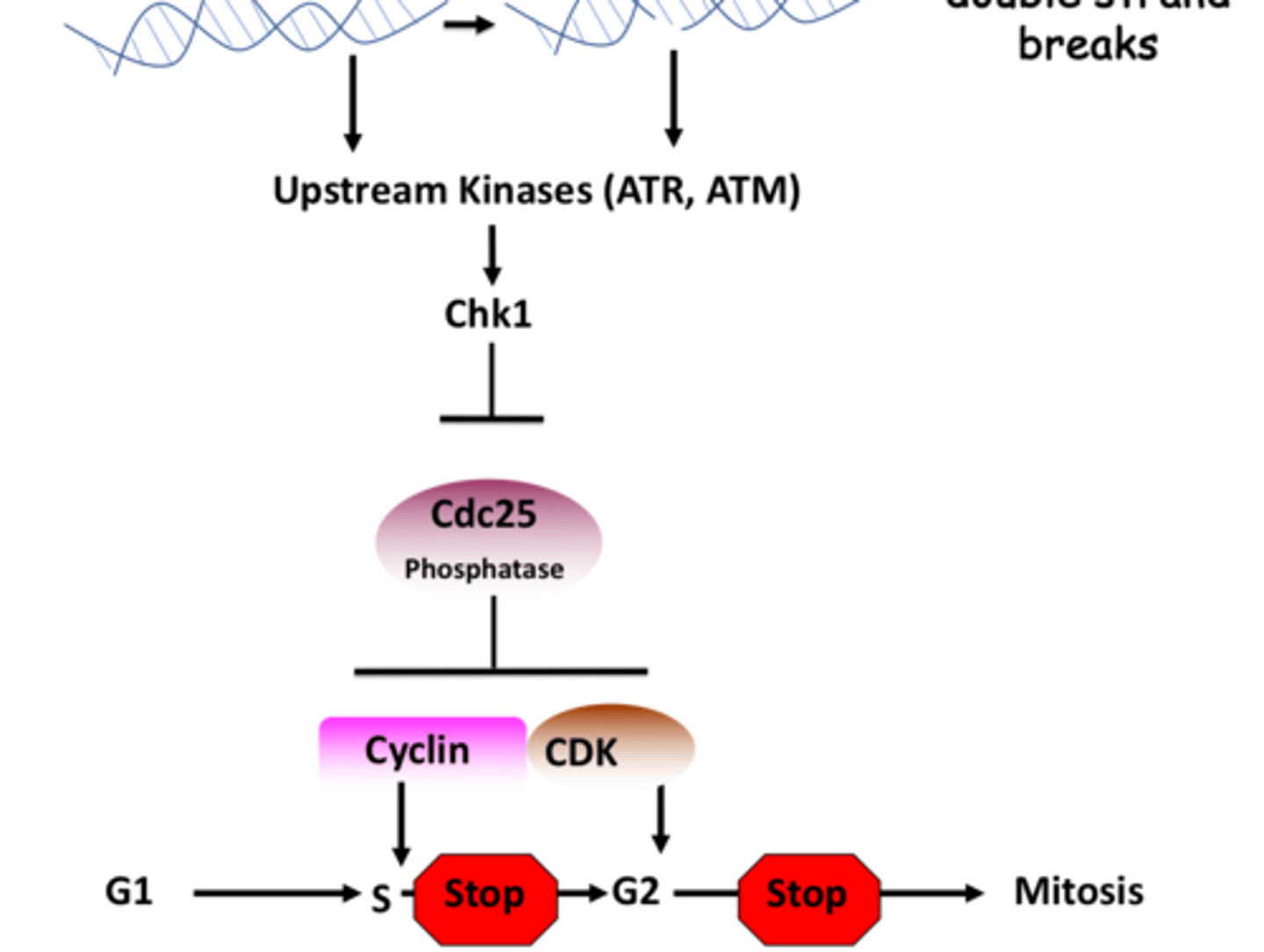
Activation of Checkpoint Kinase 1 (Chk1) by DNA damage stalls the cell cycle in S or G2 Phase by inhibiting the Cdc25 phosphatase
Chk1 inhibits Cdc phosphatase
^^^gets activated by ATR / ATM kinases
Protein Degradation by APC/C Complex
PTEN
PTEN = Tumor Suppressor and inhibits hyper activation of PI3K
NO PTEN→ hyperactivation of PI3K→ cell proliferationand growth
EMT
epithelial to mesenchymal transition
epithelial to mesenchymal transition
Acquisition by epithelial cells of the phenotypes of mesenchymal cells such as fibroblasts.
process by which epithelial cells lose their cell polarity and cell-cell adhesion, and gain migratory and invasive properties to become mesenchymal stem cells
CANCER
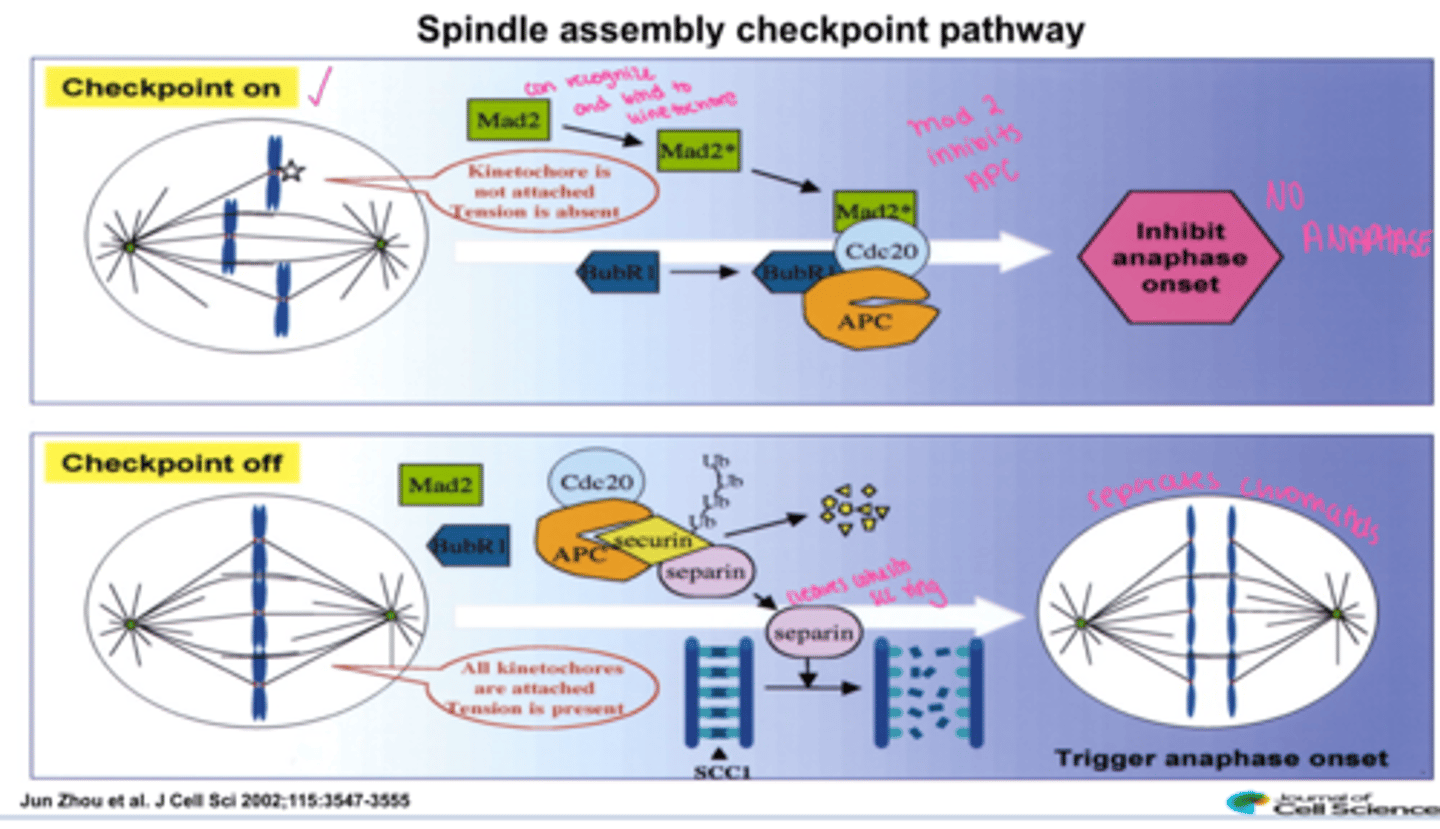
Mad2
(Mitotic Arrest Deficient) -component of Mitotic Checkpoint Complex (MCC) - inhibits CDC20
signal that chromosomes are not completely attached to microtubule spindles by blocking Cdc20 action
One of several proteins recruited to unattached kinetochores, it binds and inhibits Cdc20-APC/C and prevents cells from passing through the spindle assembly checkpoint until all chromatids are properly attached to spindle poles
Spindle Assembly Checkpoint (SAC)
Operates at the metaphase/anaphase transition to check for misaligned chromosomes (the third critical checkpoint).
Mad2 activated when misaligned and checkpoint is ON and inhibits anaphase
when all chromosomes are aligned hooray Mad2 is not active and securin is degraded and separin cleaves cohesin scc ring and anaphase occurs
anaphase A
movement of chromosomes toward the poles
anaphase B
spindle poles move apart
BOLES POLES
kinesin 5 and dyenin = astral
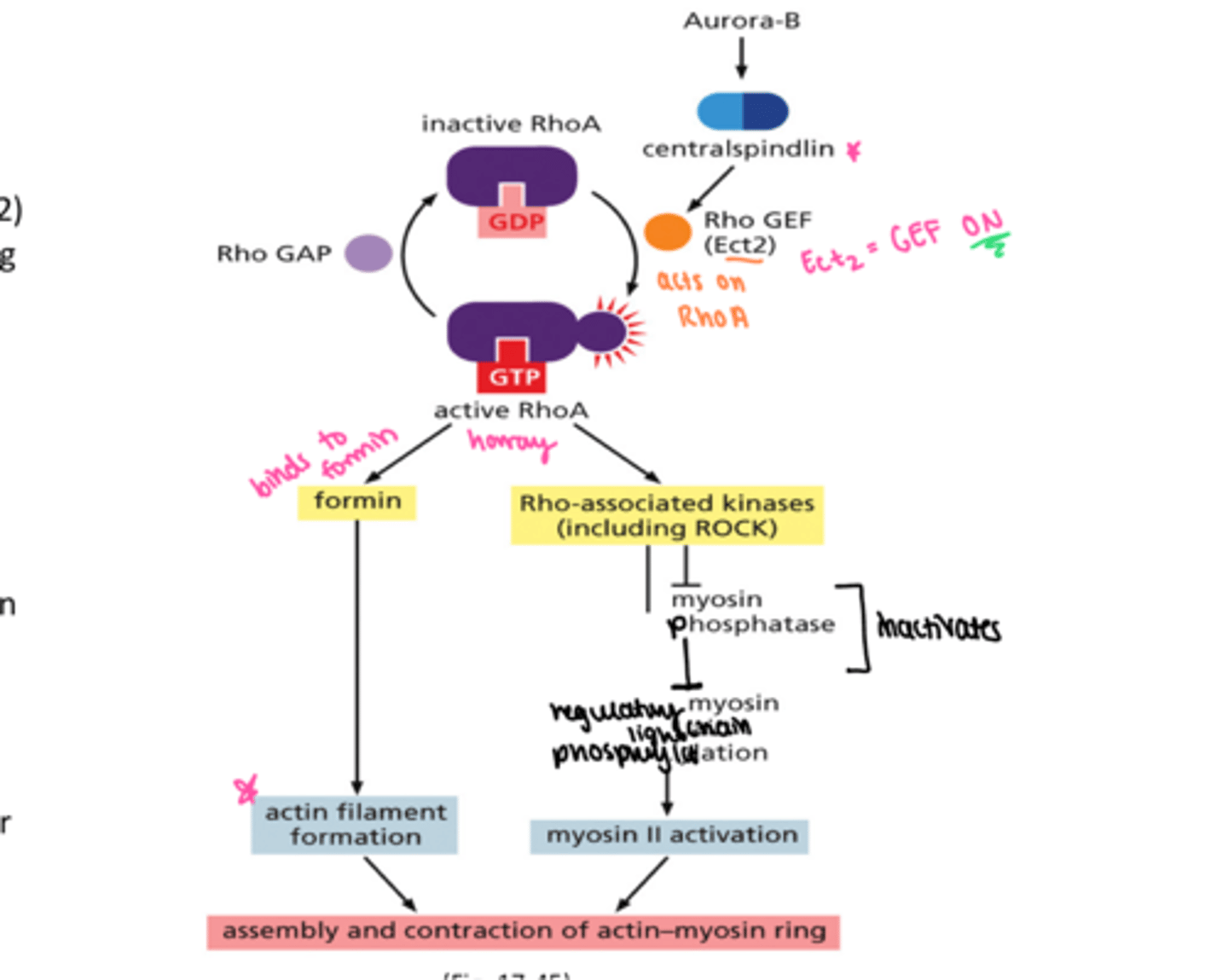
How is contractile ring assembled?
aurora b and central spindilin come first
Rho GEF = Ect2
Rho GAP
active RhoA binds to formin = actin
active RhoA binds to ROCK (kinases) = myosin
CONTRACTILE RING!
phragmoplast
In dividing plant cells, a structure formed by overlapping microtubules that guide vesicles containing cell wall components to the middle of the cell.
no astral microtubules since then have no centromeres