T3 - IE3 - Cardiology - Kaur - Venous Thromboembolism & Dyslipidemia Medicinal Chemistry
1/100
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
101 Terms
Warfarin has a ____________, ________ structure
- lactone
- coumarin
Warfarin has an ________ 4-hydroxyl group
- acidic
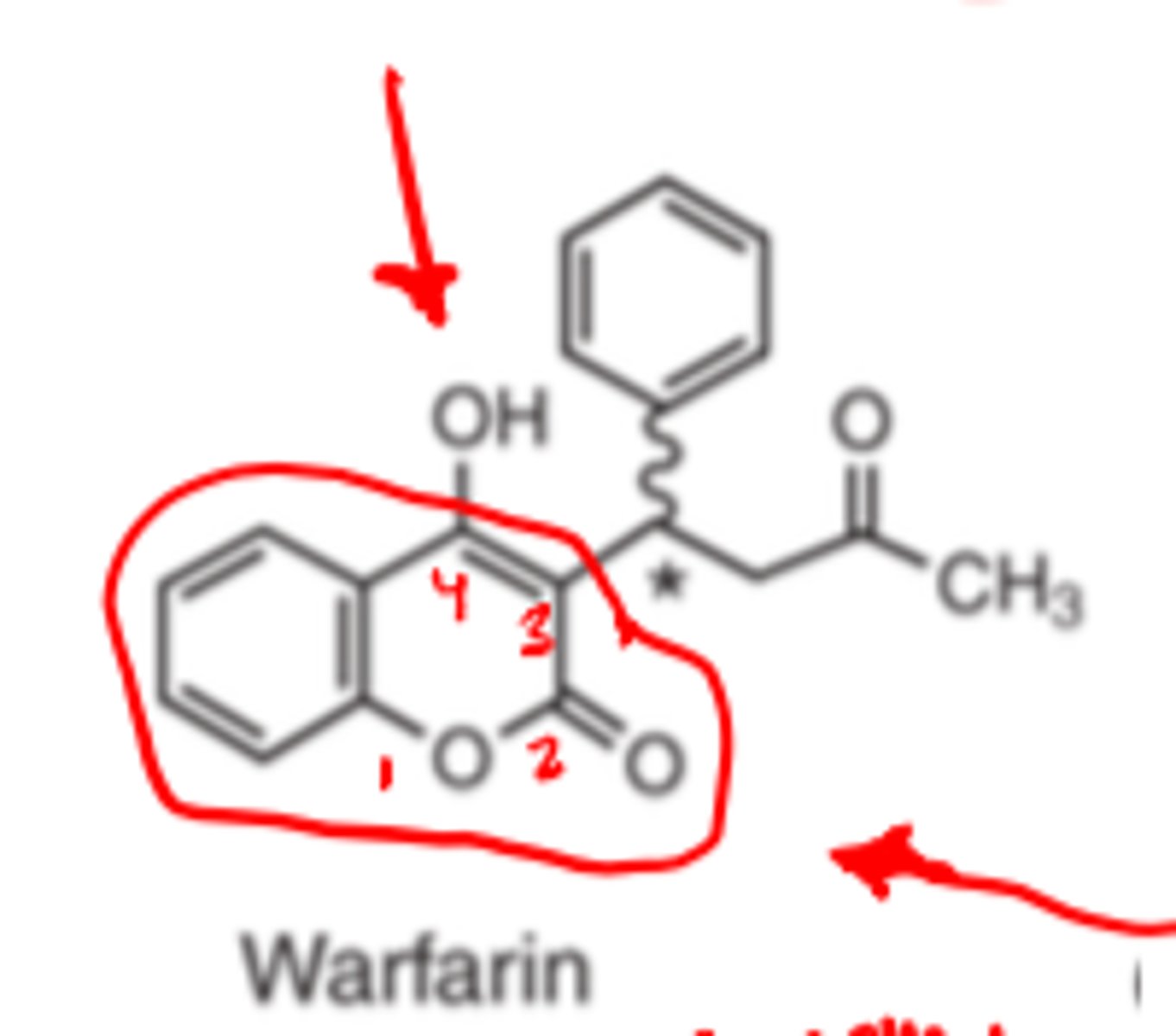
Warfarin has an acidic ___-_________ group
- 4-hydroxyl group
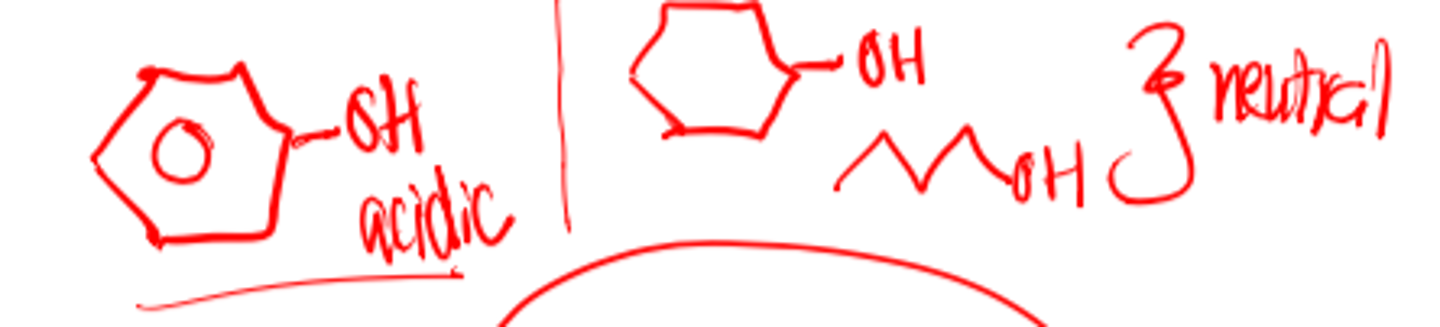
Most hydroxyl groups are neutral unless attached to a phenyl group
The (__)-warfarin is more potent than the (__)
- S
- R
Most hydroxyl groups are _________ unless attached to a benzene ring, which makes it ________
- neutral
- acidic
Warfarin has an acidic 4-hydroxyl group due to conjugation
Warfarin is _________ and _________ absorbed
- rapidly
- completely
~100% after administration
Warfarin peak plasma concentration in ____ _______
- three hours
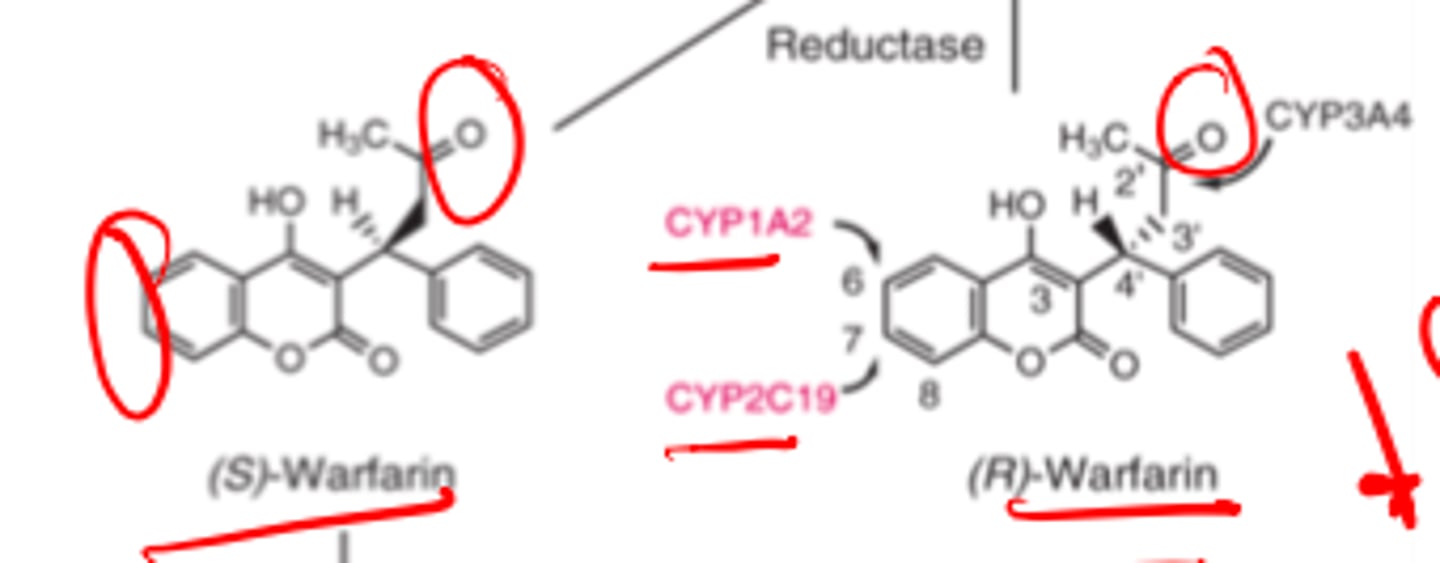
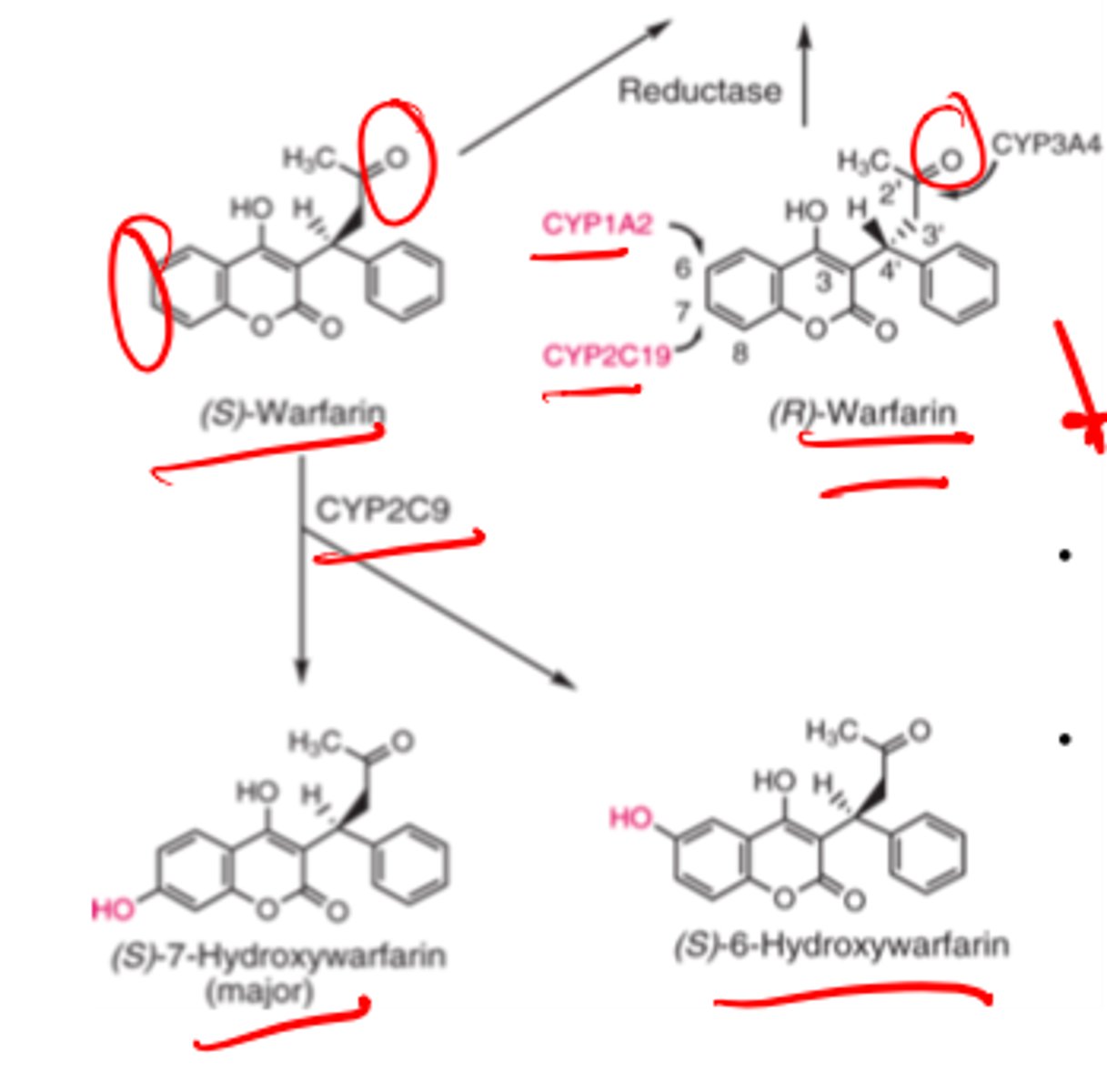
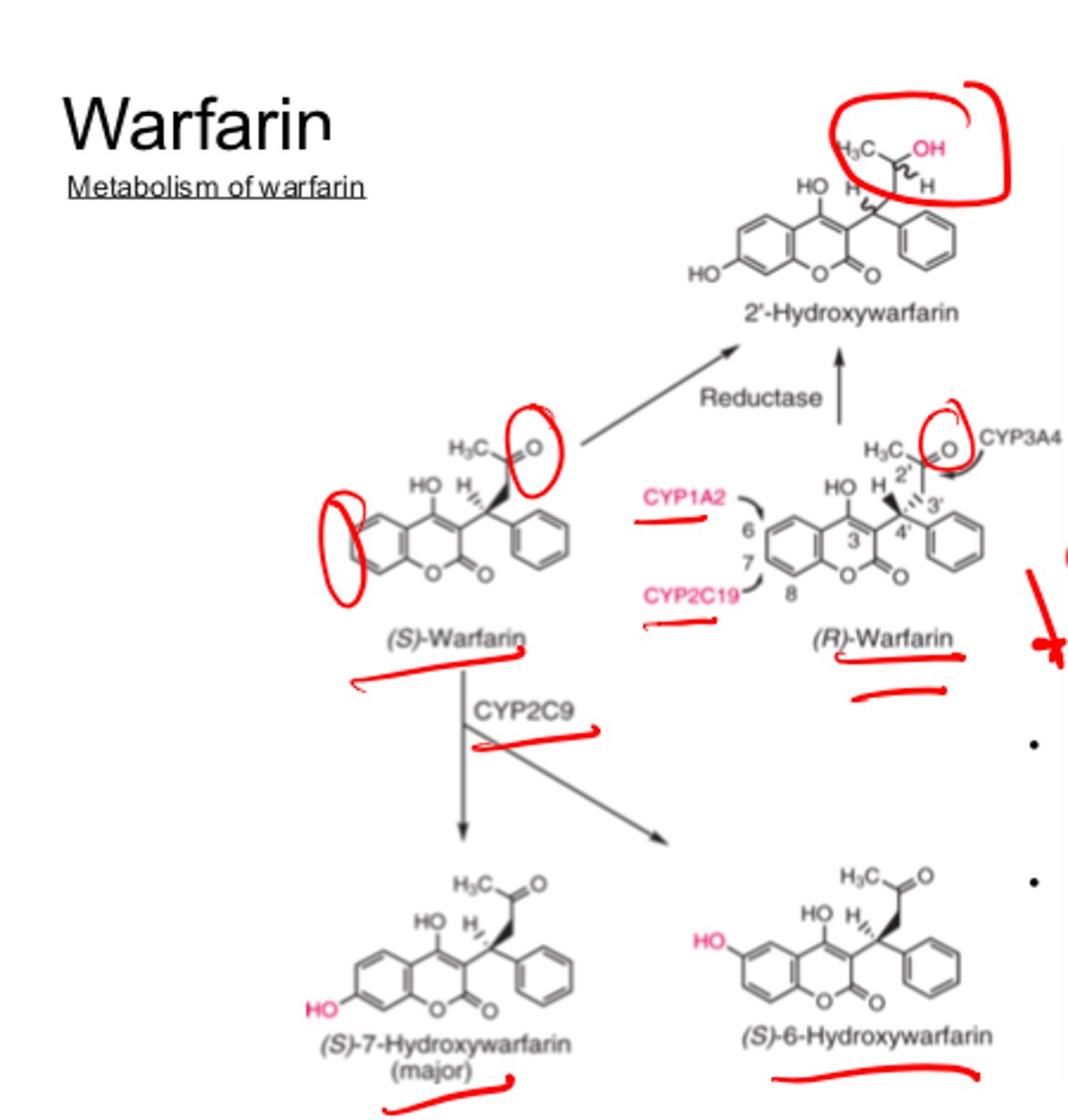
(S)-warfarin is metabolized by ___________ to inactive metabolites __- and ___-_______________
- CYP2C9
- 6
- 7-hydroxywarfarin
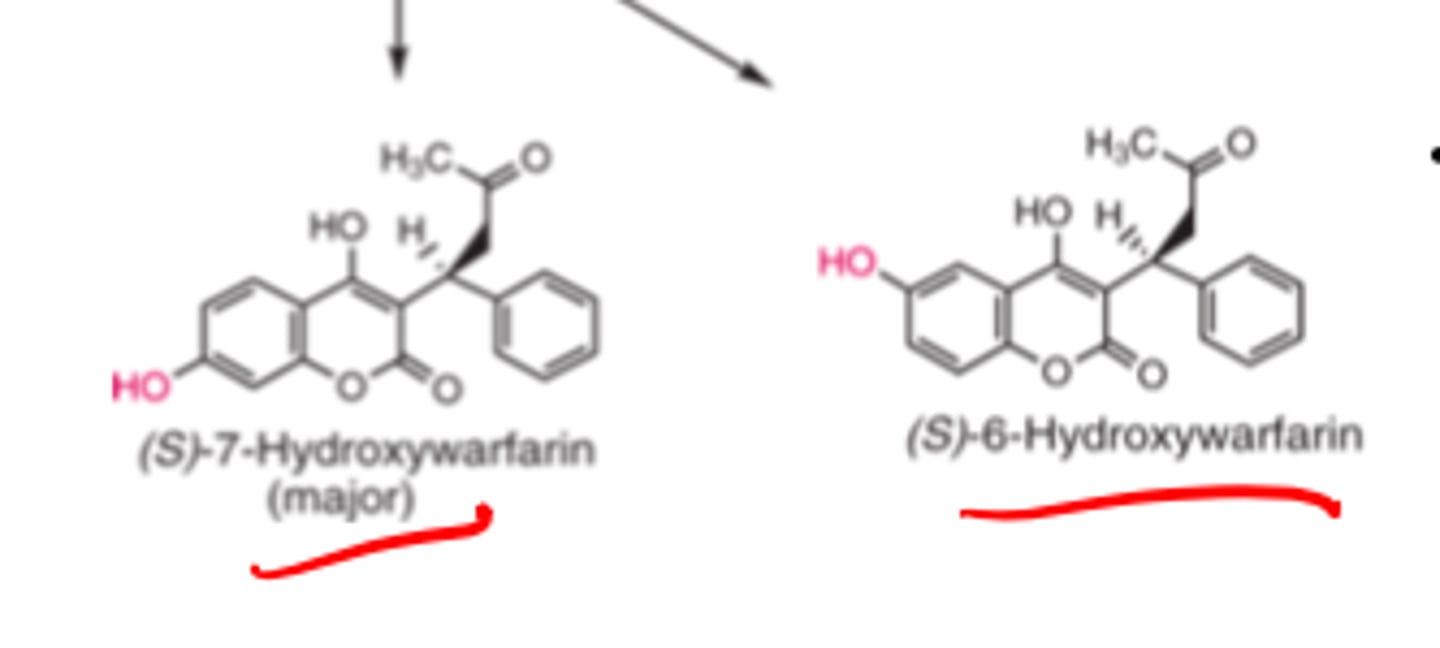
Reductive metabolism of Warfarin side chain carbonyl gives...
diastereometric Z-hydroxyWarfarin
Drug-Drug Interactions of Warfarin
Associated with enhanced or inhibited metabolism of warfarin (induction or inhibition of CYP2C9)
HMW
high molecular weight
LMW
low molecular weight
______________ has almost no unchanged drug excreted in urine
- Warfarin
Everything is relatively metabolized / changed when excreted.
Warfarin has almost ___ _______ drug excreted in urine
- no unchanged
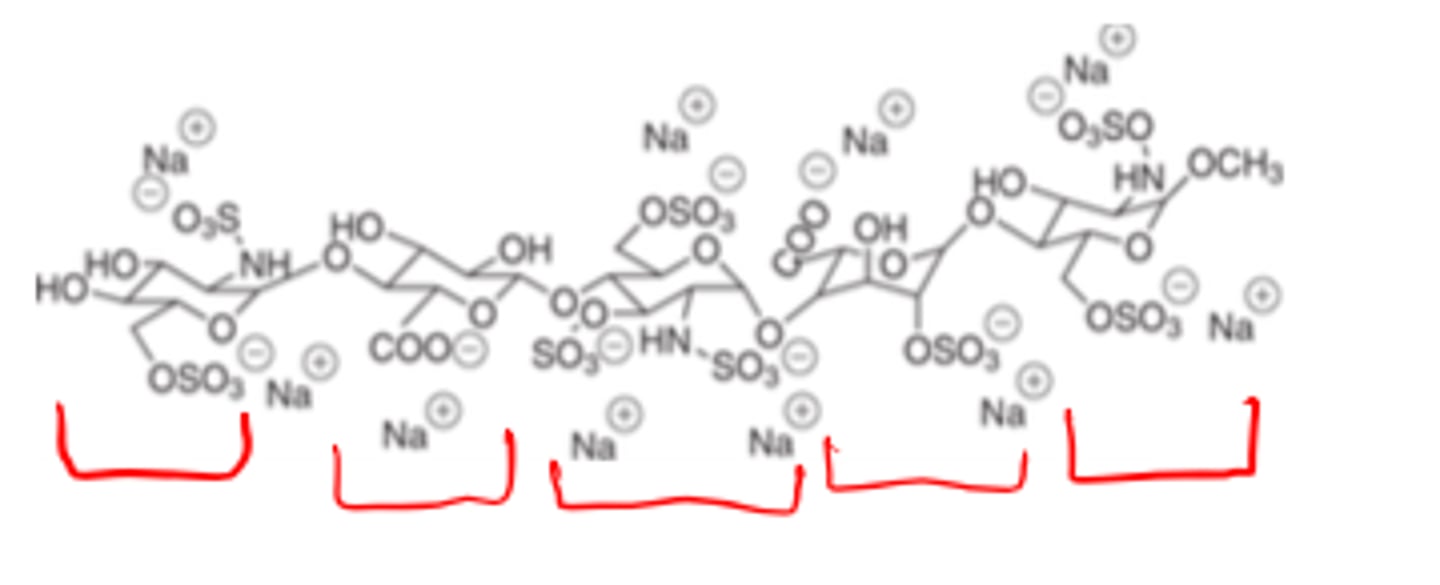
Heparin has a _______ ________ structure
- HMW polysaccharide (5-30 kDa)
Heparin is administered...
IV or subcutaneous but not orally due to PK
Heparin Physicochemical properties
sulfated, negatively charged, acidic molecule mostly used as sodium salt
Same physiochemical properties as Enoxaparin, Dalteparin, Fondaparinux
Heparin-based Anticoagulants
Heparin
Enoxaparin
Dalteparin
Fondaprinux
_________ and ________ are more selective compared to heparin
- Enoxaparin
- Dalteparin
Enoxaparin and Dalteparin are _______ _______ compared to heparin
- more selective
Both are LMW polysaccharides (4-6kDA) compared to (5-30 kDA) Heparin
Fonaparinux (heparin-based anticoagulant) has a _________ _________ structure
- synthetic pentasaccharide
Whereas, Heparin has a HMW polysaccharide structure (5-30 kDa); Enoxaparin and Dalteparin have LMW (4-6 kDa) polysaccharide structures.
Enoxaparin and Dalteparin have _________ _________ PK/PD profile in comparison to heparin
- more favorable
Fondaparinux is a prototype of a novel class of anticoagulants with significant advantages compared to their structurally related heparin. It is the first _________ ________ _____ _______
- selective factor Xa inhibitor
Fondaprinux is a ________, ________ sulfonated pentasaccharide. The immediate advantage of fondaprinux is that as a synthetic drug, its composition will _________ _________, which results in improved pharmacokinetics and more selective anticoagulant action
- synthetic
- highly
- not change
Fondaprinux is a synthentic, highly sulfonated pentasaccharide. The immediate advantage of fondaprinux is that as a synthetic drug, its composition will not change, which results in __________ __________ and _________ _________ anticoagulant action
- improved pharmacokinetics
- more selective
Fondaparinux is administered via ___________ _______ with a _______ _____ dose and shows complete absorption
- subcutaneous
- single daily
Fondaparinux has _________ _________ effect
- predictable anticoagulant
Does not require routine coagulation monitoring
Fondaparinux is ______ _______ and is excreted in the urine _______ in patients with normal renal function
- NOT metabolized
- unchanged
Elimination half-life of 17 hours
Fondaparinux Half-life
17 hours
Hirudin structure
small protein (65 amino acids)
Lepirudin structure
recombinant protein (65 amino acids)
Same as desirudin
Desirudin structure
recombinant protein (65 amino acids)
Same as lepirudin
Bivalirudin Structure
Peptide (20 amino acids)
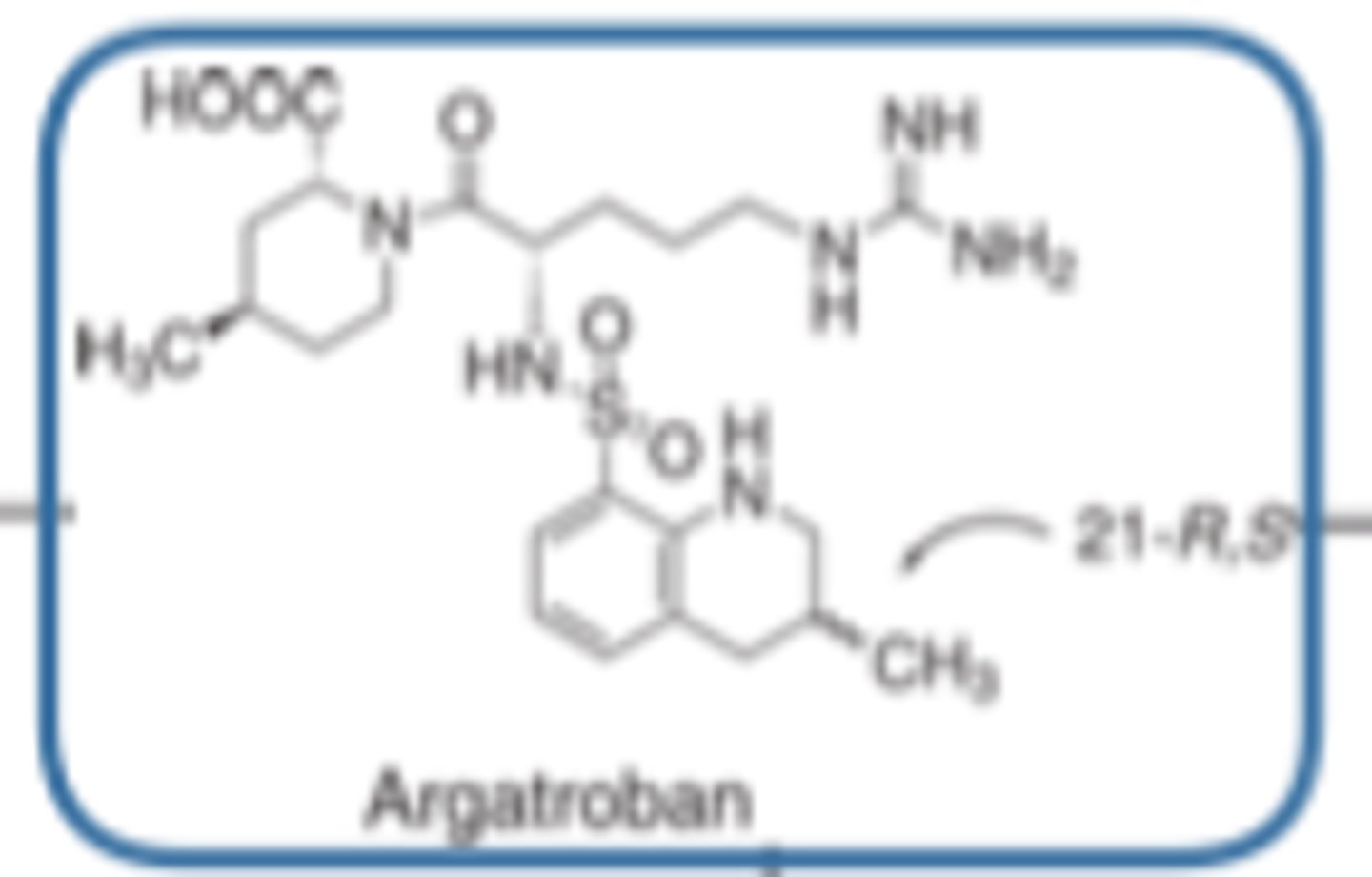
Argatroban has a _________-_______ structure
- peptide-mimetic
Looks a lot like a peptide but not a peptide
Dabigatran etexilate structure
non-peptide mimetic prodrug
Direct Thrombin Inhibitor Drugs
Hirudin
Lepirudin
Desirudin
Bivalirudin
Argatroban
Dabigatran etexilate
Hirudin MOA
irreversible inhibition of thrombin (both free thrombin and thrombin bound to fibrin)
Binds active and exosite of thrombin
Lepirudin and Desirudin have same mechanism of action
Bivalirudin MOA
reversible inhibitor, therefore less risk of bleeding
Argatroban and Dabigatran are also reversible inhibitors
Bivalirudin binds to both _______ _____ of thrombin and _____ of thrombin
- active site
- exosite

______ amino acids of Bivalirudin structure that binds to active site. Whereas ____ amino acids bind to the exosite
- 4
- 12
Active Site:
D-Phe
Pro
Arg
Pro
Exosite:
Asn
Gly
Asp
Phe
Glu
Glu
Ile
Pro
Glu
Glu
Tyr
Leu
Hirudin PK
IV
Lepirudin PK
IV
Half-life: 1.3 hours
Desirudin PK
SC
Half-life: 2-3 hour
Bivalirudin PK
IV
Rapid onset and short duration of action
Argatroban PK
IV or SC
Dabigatran is ________ ______, with RAPID onset and SHORT duration of action
- orally active (however, low bioavailability)
Argatroban has ___________ metabolism
- oxidative
Argatroban's ____________ metabolite retains 20-30% of activity
- aromatic
Whereas the hydroxylated metabolite is inactive
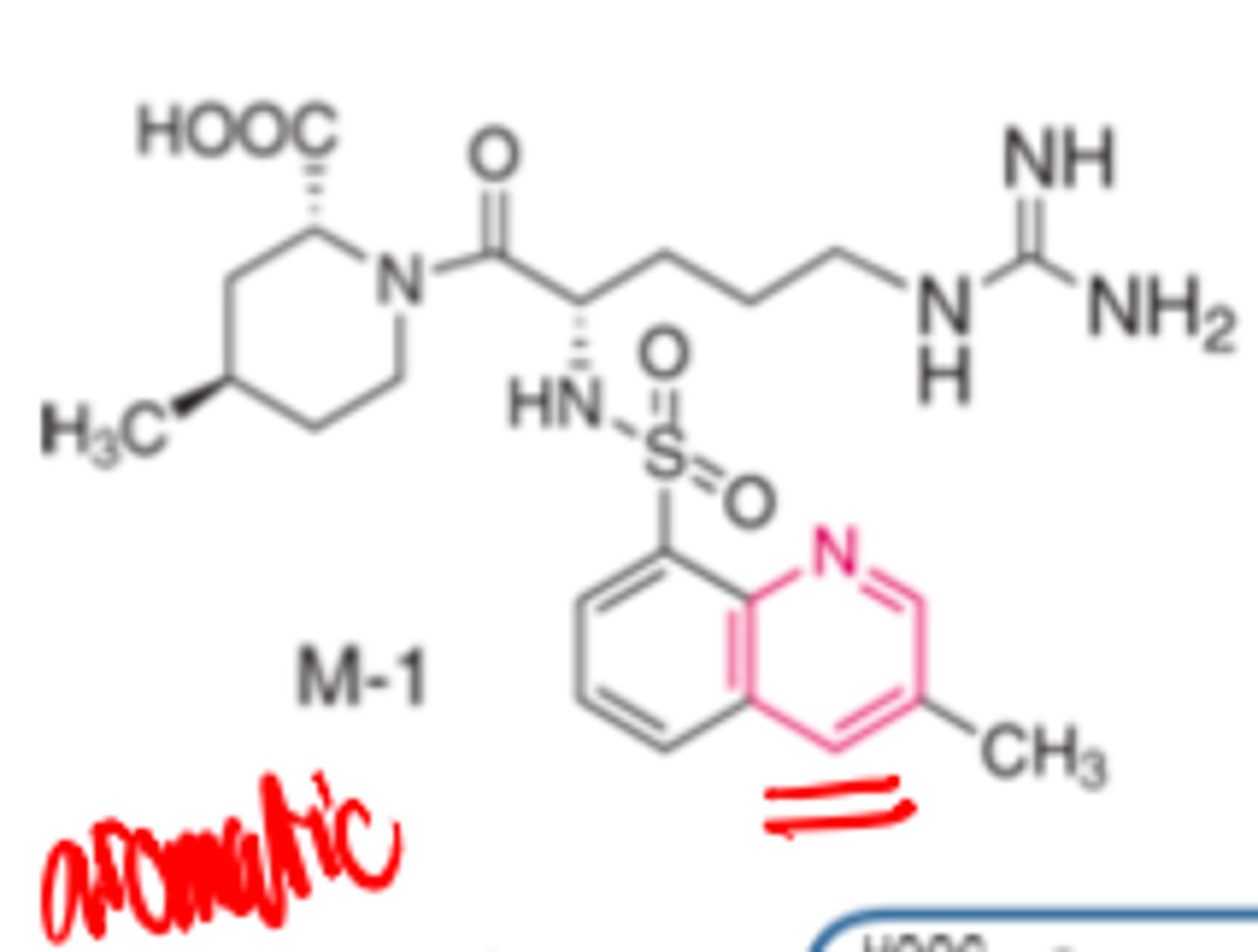
Argatroban's _________ metabolite is inactive
- hydroxylated
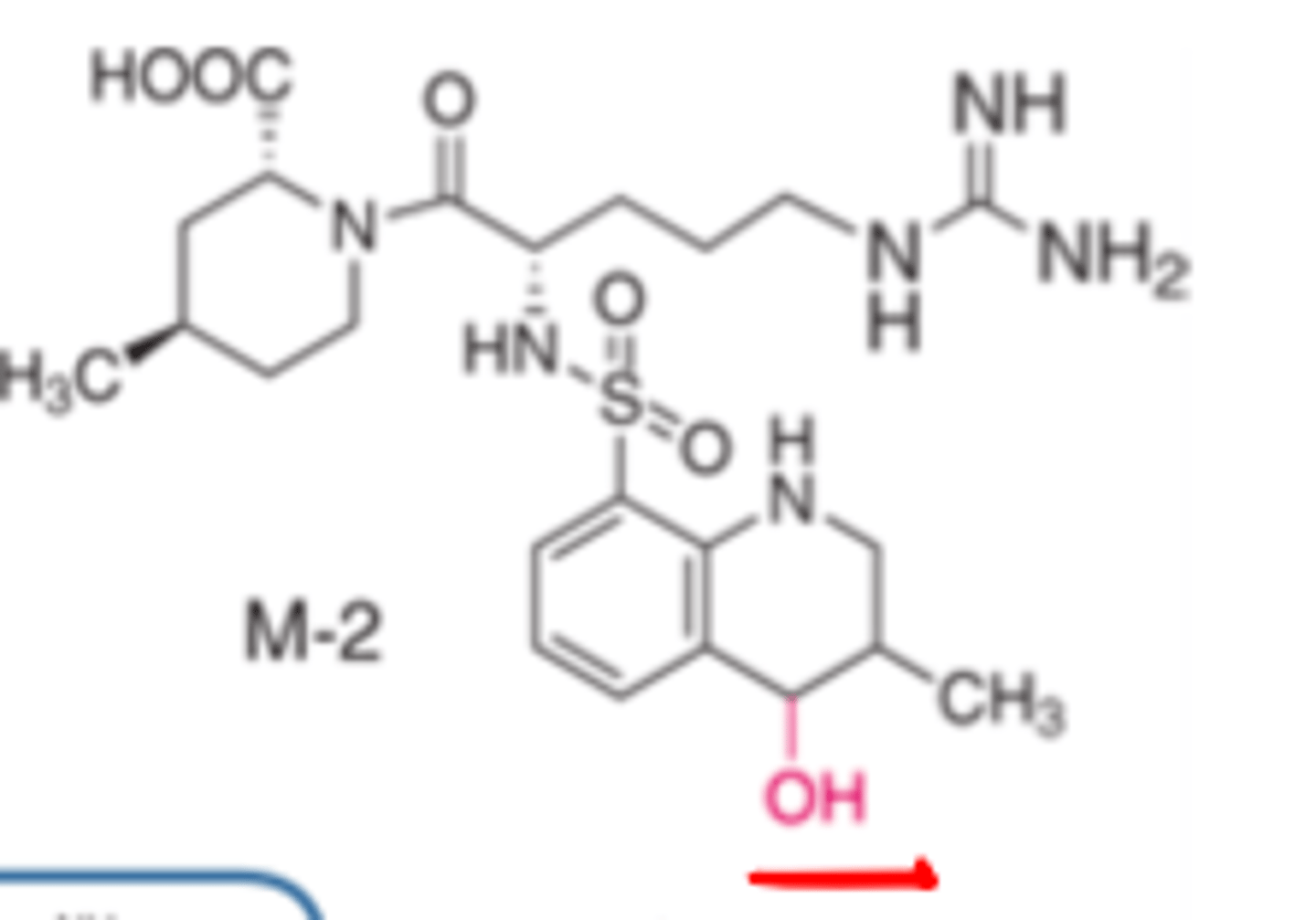
Argatroban aromatic metabolite retains...
20-30% activity
Argatroban's hydroxylated metabolite is ____________
- inactive
Due to dabigatran's rapid onset and rapid offset, the drug does _________ __________ coagulation monitoring, which represents a major advantage over other anticoagulant therapy
- not require
However, the drug is reported to produce increase in risk of major GI bleeding, and the patients should be aware
Dabigatran etexilate is metabolized to the prodrug by ____________
- esterases
Prodrug is fairly neutral because charged group have esters, after esters hydrolyze group
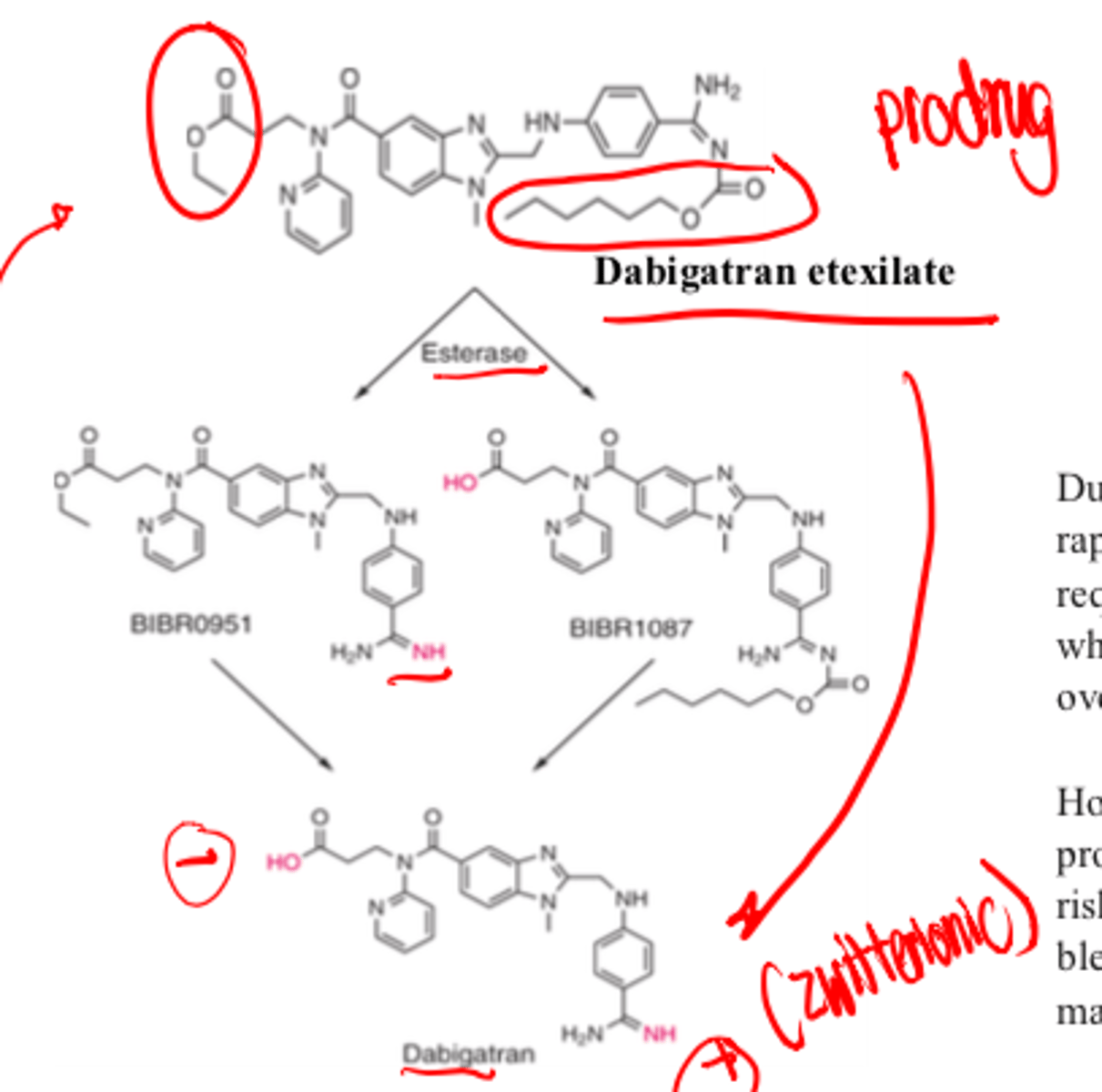
Dabigatran (non-prodrug) has _______________ charge
- zwitterionic
However, due to its charged groups - it has difficulty for absorption. It's prodrug form allows it to be orally active
Thrombolytic Drugs
Streptokinase (1st generation)
Alteplase (2nd generation)
Tenecteplase (3rd generation)
Streptokinase has a _____ _____ ______ ________ structure
- 414 amino acid protein
Alteplase has a _______ ______ structure
- serine protease (527 amino acid protein)
Tenecteplase has a _______ _______ structure
- serine protease (527 amino acid protein)
However, it has a three point mutations compared to 2nd generation alteplase. That allows for higher fibrin specificity, improved activity
Alteplase is ______-_______ _____ _____
- fibrin specific selective agent
Whereas, the 1st generation streptokinase is fibrin NON-specific
Streptokinase MOA
fibrin-non specific agent
Alteplase has LOW affinity for ______ ________ and very HIGH affinity for plasminogen _________ to fibrin
- free plasminogen
- bound
However, tenecteplase has HIGHER fibrin specificity, improved activity
Tenecteplase has similar structure to alteplase but contains _____ _______ _______, which gives it _______ fibrin specificity and _______ activity
- three point mutations
- higher
- improved
Alteplase PK
very short half-life (~5 minutes)
Tenecteplase PK
prolonged half-life (~17 minutes); allows for a single bolus application
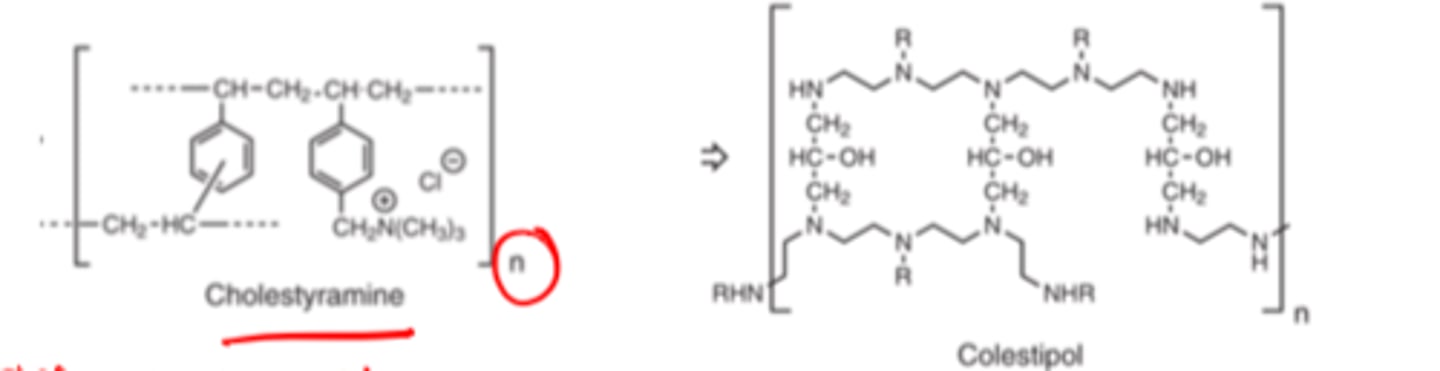
Bile Acid Sequestrants structure contains ________ ________ ________ and ___________ ________ groups and ________/_________ ________
- positively charged resins
- quaternary ammonium
- secondary/tertiary amines
Normal pKa values for the amines range from 9.0 to 10.5, thus ALL of these groups should be primarily ionized at intestinal pH
Bile Acid Sequestrants amines have normal pKa values of _____ to _____. Thus, all of the these groups should be primarily _______ at intestinal pH
- 9.0
- 10.5
- ionized
Bases are ionized from the beginning but lose their charge once pH > pKa
Bile Acid Sequestrants positively charged resins form ________ ______ with bile acids
- electrostatic interactions
Bile acids are negative, thus, are attracted to the positive bile acid sequestrants
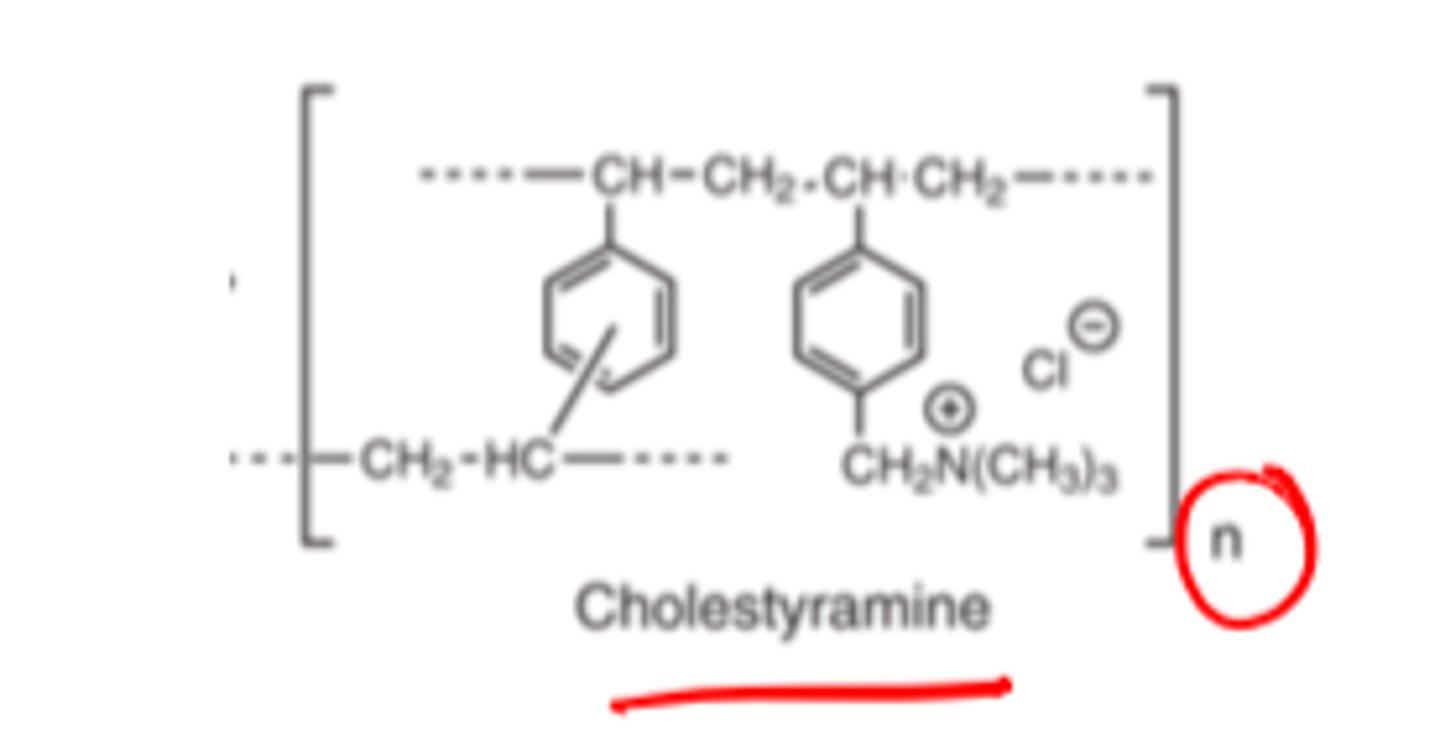
Bile Acid Sequestrants PK/Metabolism
Taken orally, but NOT absorbed (thus, minimal side effects)
Bile Acid Sequestrants Onset of Action
24-48 hours; can take up to 1 month to achieve peak response
Bile Acid Sequestrants are excreted in _____ as insoluble complex with bile acids
- feces
Bile Acid Sequestrants Drug Interaction
Potentially bind and sequester ANY acidic drug such as warfarin
HMGRIs (Statins)
HMG-CoA Reductase Inhibitors
Statins pharmacophore consists of...
3,5-dihydroxyheptanoic acid and decalin or OTHER aromatic ring
3R, 5R stereochemistry at 3- and 5- positions are very important
In their active forms, all HMGRIs contain a...
carboxylic acid
In their _______ forms, all HMGRIs contain a carboxylic acid
- active
Lovastatin is _________, whereas simvastatin is _________
- natural (R = H)
- synthetic (R = CH3)
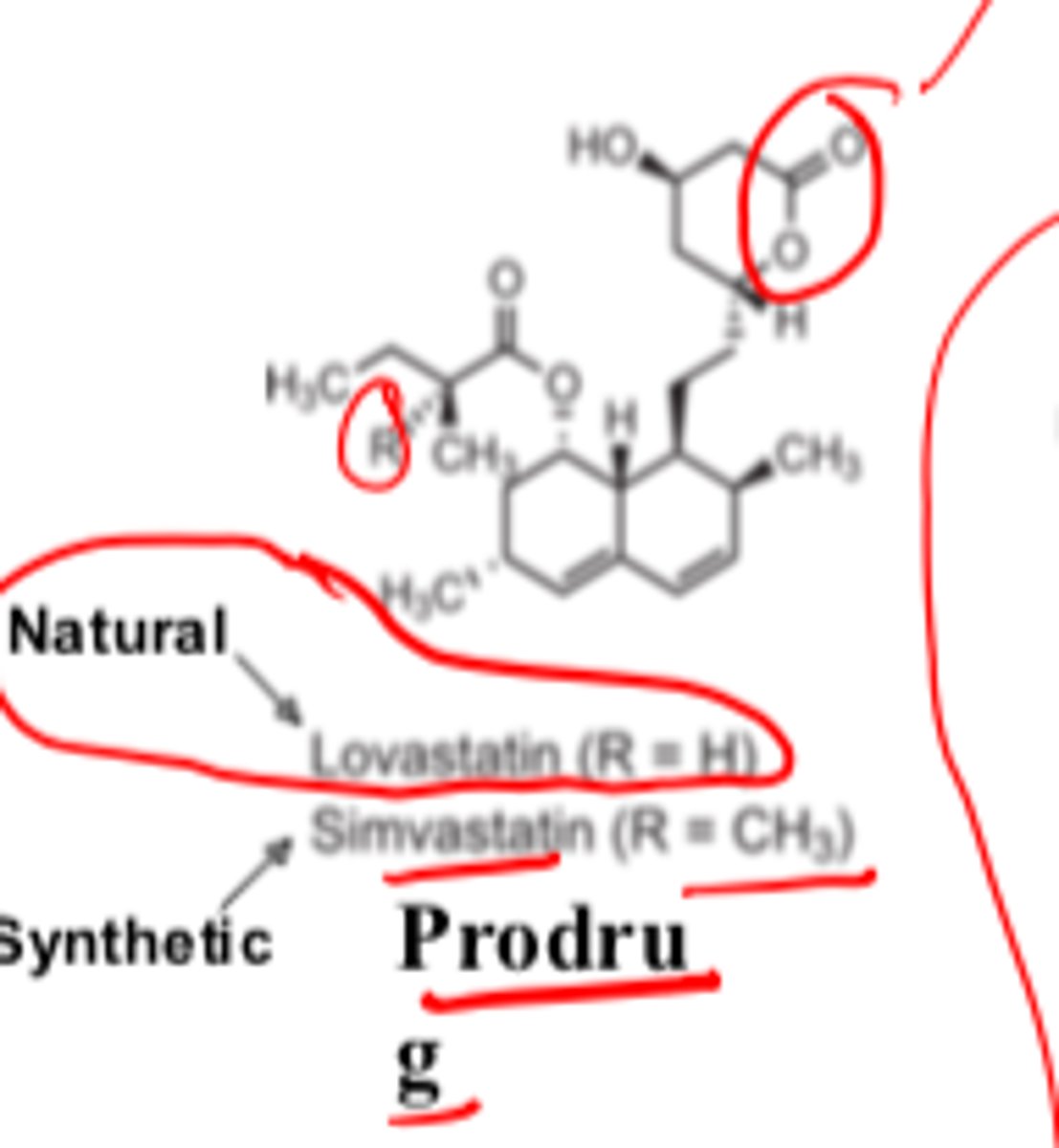
Lovastatin and simvastatin are __________
- prodrugs
Their active forms contain a carboxylic acid
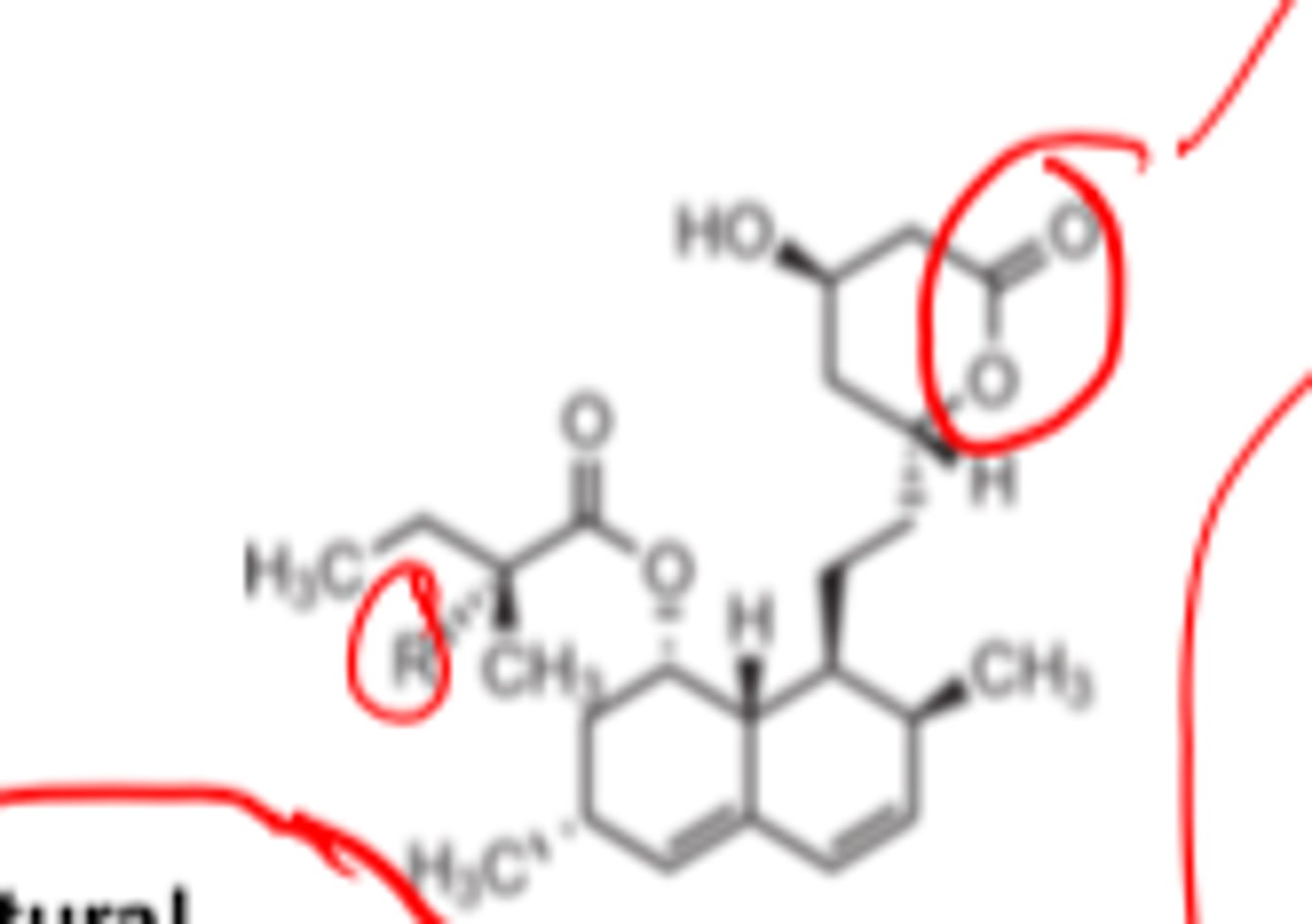
The inactive lactone prodrugs of HMGRIs must undergo in vivo __________ to produce effects
- hydrolysis
Good first-pass metabolism so, it has low oral bioavailability: 5-20% except pitavastatin (51%)
HMGRIs have good ____-_____ _______
- first-pass metabolism
Thus, low oral bioavailability (5-20%) with the exception of pivastatin (51%)
HMGRIs peak reduction of plasma cholesterol:
4-6 weeks of therapy (except atorvastatin - only 2 weeks)
HMGRI elimination half-life
1-4 hours
Except atorvastatin and rosuvastatin (19 hours)
HMGRIs (statins) should be administered at _________
- bedtime
Except atorvastatin and rosuvastatin
_________ and _________ are the only HMGRIs that do not need to be administered at bedtime due to their _______ ______ ___-______
- Atorvastatin
- Rosuvastatin
- long elimination half-life (19 hours)
Cholesterol Absorption Inhibitor Drugs
Ezetimibe
After oral administration, ezetimibe is ______ and _____ metabolized in the intestinal wall and the liver to its _________ _______
- rapidly
- extensively
- active metabolite (phenol glucuronide)
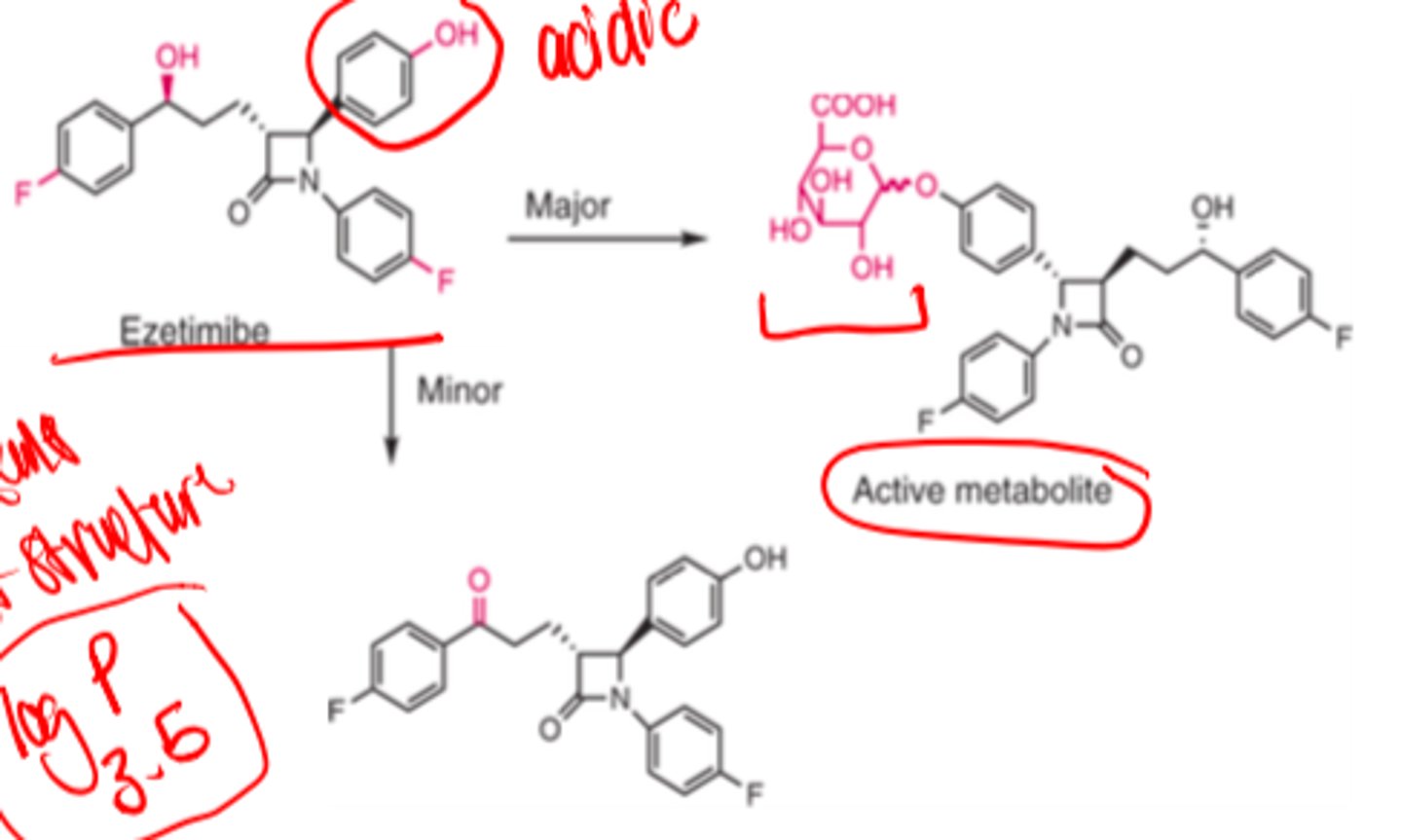
Ezetimibe has a ______ ______ structure
- small molecule
A small amount (<5%) of ezetimibe undergoes __________ to convert the benzylic hydroxyl group to a ketone
- oxidation
Ezetimibe is adminsitered _______; however, its absolute bioavailability cannot be determined because of its ________ _______ and lack of an ________ formulation
- orally
- aqueous insolubility
- (lack of) injectable formulation
Both ezetimibe and its glucuronide conjugate have a half-life of approximately...
22 hours
Fibrates Pharmacophore
phenoxy, spacer group, isobutyric acid
Compounds containing esters are prodrugs

_______-_________ of fibrates produces compounds with significantly longer half-life
- Para-substitution
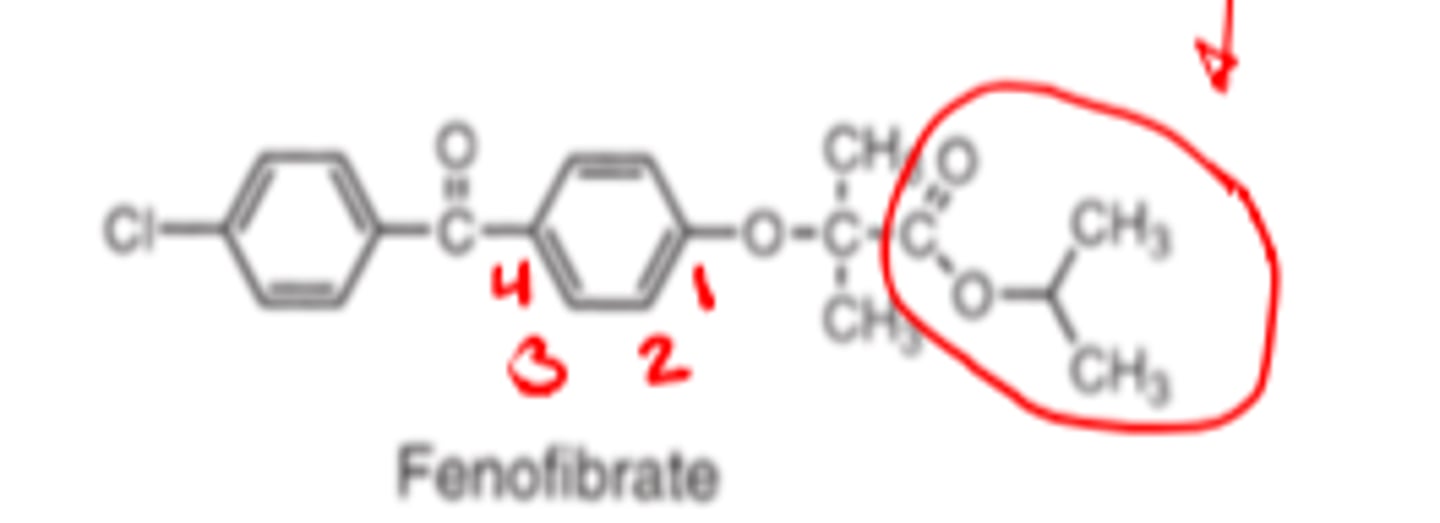
Fibrates inactive ester prodrugs must undergo ____ _______ ______ to produce effects
- in vivo hydrolysis
Oxidation of the aromatic methyl groups produces inactive hydroxymethyl analogs
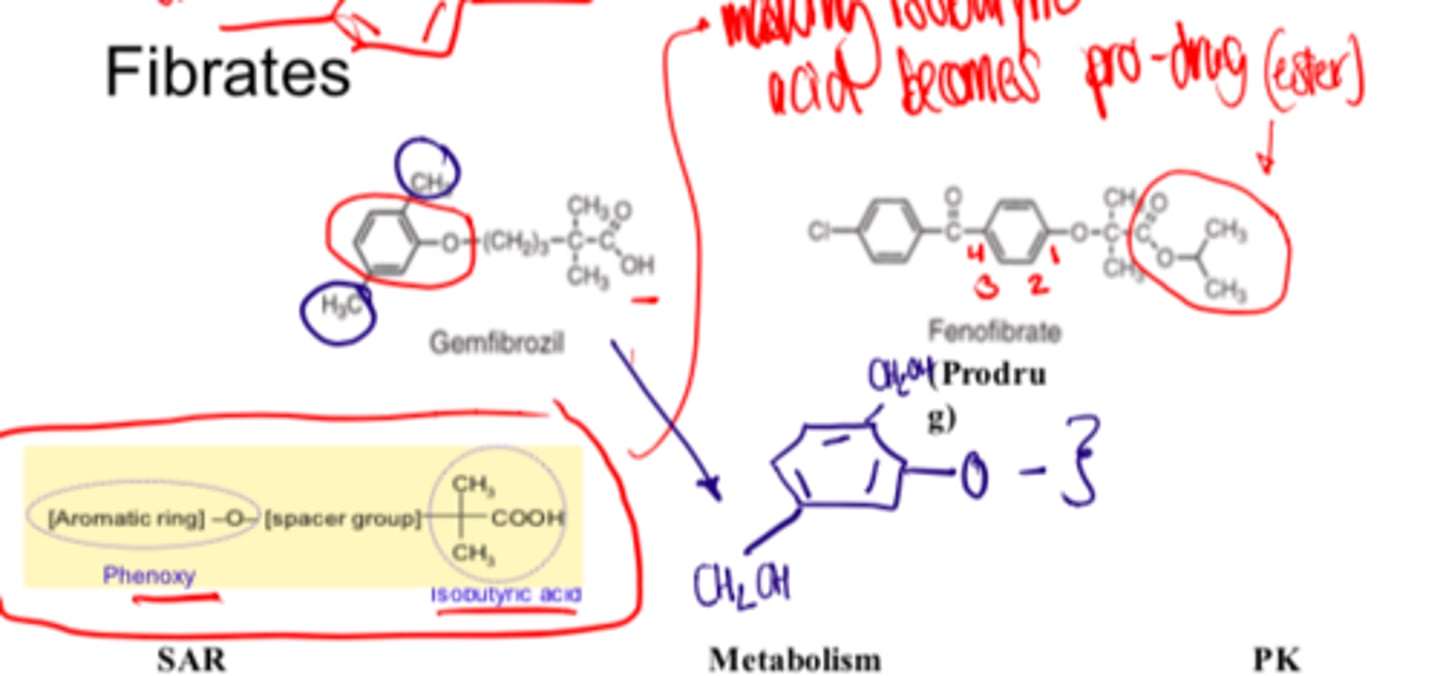
As a drug class, fibrates and their oxidized analogs are primarily excreted as...
glucuronide conjugates in the urine
Fibrates PK
excellent bioavailability (60-90%) food can ENHANCE oral absorption (should be taken with or just BEFORE meals)
Nicotinic acid is freely soluble in ________ _______
- alkaline solutions
Nicotinic acid is a _______, ________, _______, ______ powder
- stable
- nonhygroscopic
- white
- crystalline
Nicotinic Acid pKa
4.76
Therefore, it is primarily ionized at physiologic pH.
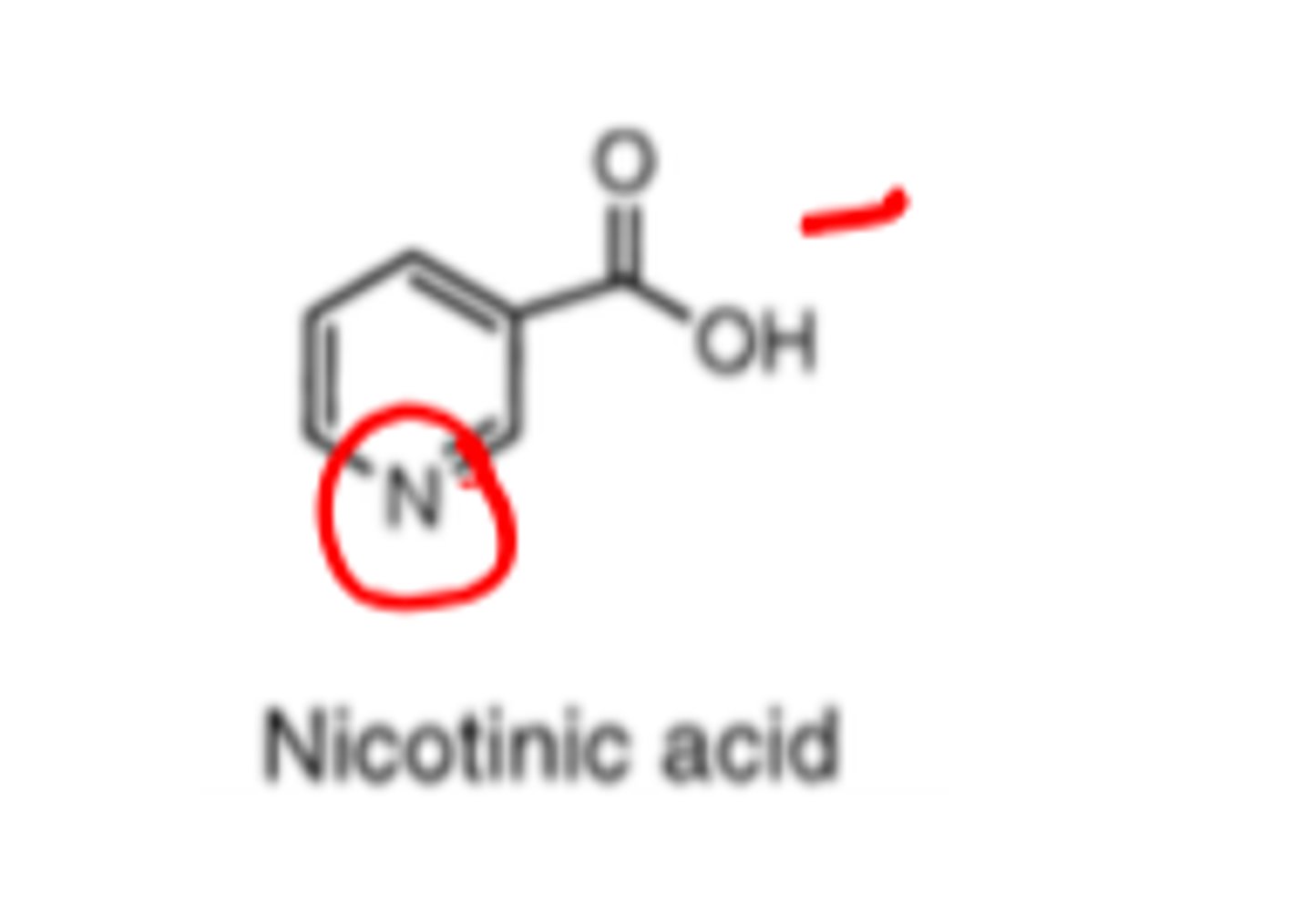
The pyridine nitrogen is a very weak base (pKa = 2.0) and primarily exists in un-ionized form.
Niacin's pyridine nitrogen is a _______ _________ base
- very weak (pKa = 2.0_
Therefore exists in the un-ionized form
Small doses of niacin used for dietary supplementation are primarily excreted as metabolites, whereas large doses used for hyperlipoproteinemia, are primarily excreted...
unchanged by the kidney
Niacin is readily ________, with peak plasma concentration occurs within ____ ______
- absorbed
- 45 minutes