Topic 6a - Rate of Reaction
1/18
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
19 Terms
What 3 things must particles do in order to react
Collide
Must have at least the minimum required energy to react (activation energy)
Collide with the correct orientation
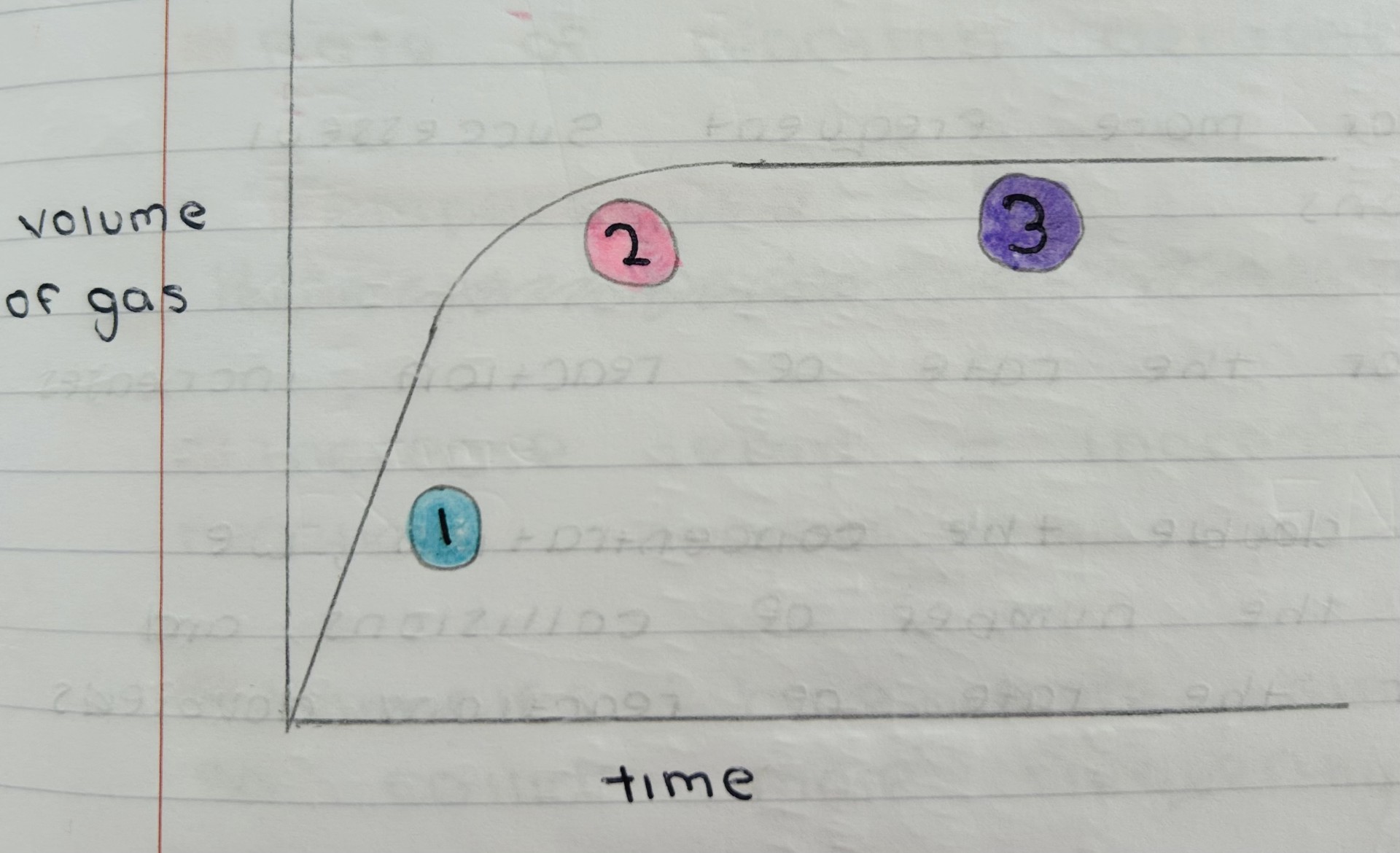
What happens in this reaction progress graph
1) Initial rate, fast, lots of reacting, particles = lots of collisions
2) Rate slows, successful collisions occur less frequently
3) Rate = 0, one or more of the reactants have been used up
4 factors that affect the rate of reaction
Concentration/pressure
Temperature
Surface area
Catalyst
How does concentration affect the rate of reaction
Increased concentration means more particles in the same space therefore more collisions
Therefore more frequent successful collisions
Therefore the rate of reaction increases
What happens to the rate of reaction when the concentration is doubled
If we double the concentration, we double the number of collisions and therefore the rate of reaction doubles
How does temperature affect the rate of reaction
Increase temp = increase rate of reaction
Particles have more kinetic energy so collide more frequently (and with more energy)
More particles have at least the minimum required energy to react
Increased frequency of successful collisions
How does surface area affect the rate of reaction
Increasing SA provides particles with more space to collide
Increases number of collisions
Increases frequency of successful collisions
Rate increases
What is a catalyst
A catalyst is a chemical that speeds up a reaction by offering an alternate reaction pathway with a lower activation energy requirement
How does catalysts affect the rate of reaction
Lower the minimum energy required to react
More particles now have at least minimum required energy
Increased frequency of successful collisions
Rate increases
Using collision theory, what happens to the rate of reaction when the volume the reaction takes place in is increased
Increasing the volume decreases the pressure
This causes particles to collide less frequently (as they are further apart)
Therefore less frequent successful collisions
Rate of reaction decreases
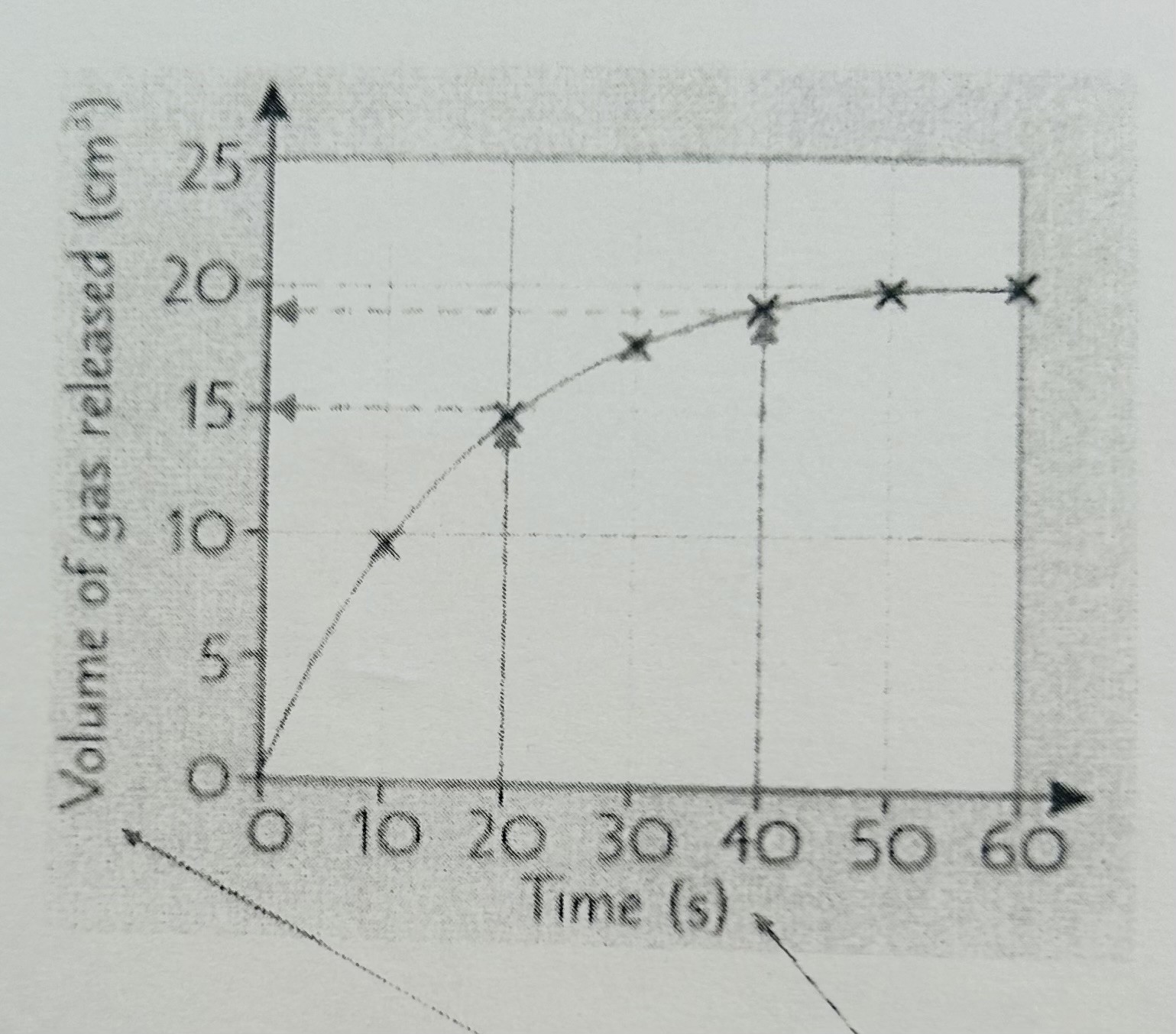
How to find rate of reaction from a graph:
Calculate the mean rate
The mean rate after 20 seconds is:
15/20 = 0.75
The mean rate after 40 seconds is:
19/40 = 0.48
The mean rate between two times eg 20 & 40 seconds is:
Volume at 40 seconds - Volume at 20 seconds/ difference in time
19-15/40-20 = 4/20 = 0.2
Why is mean rate called mean rate
It’s the average
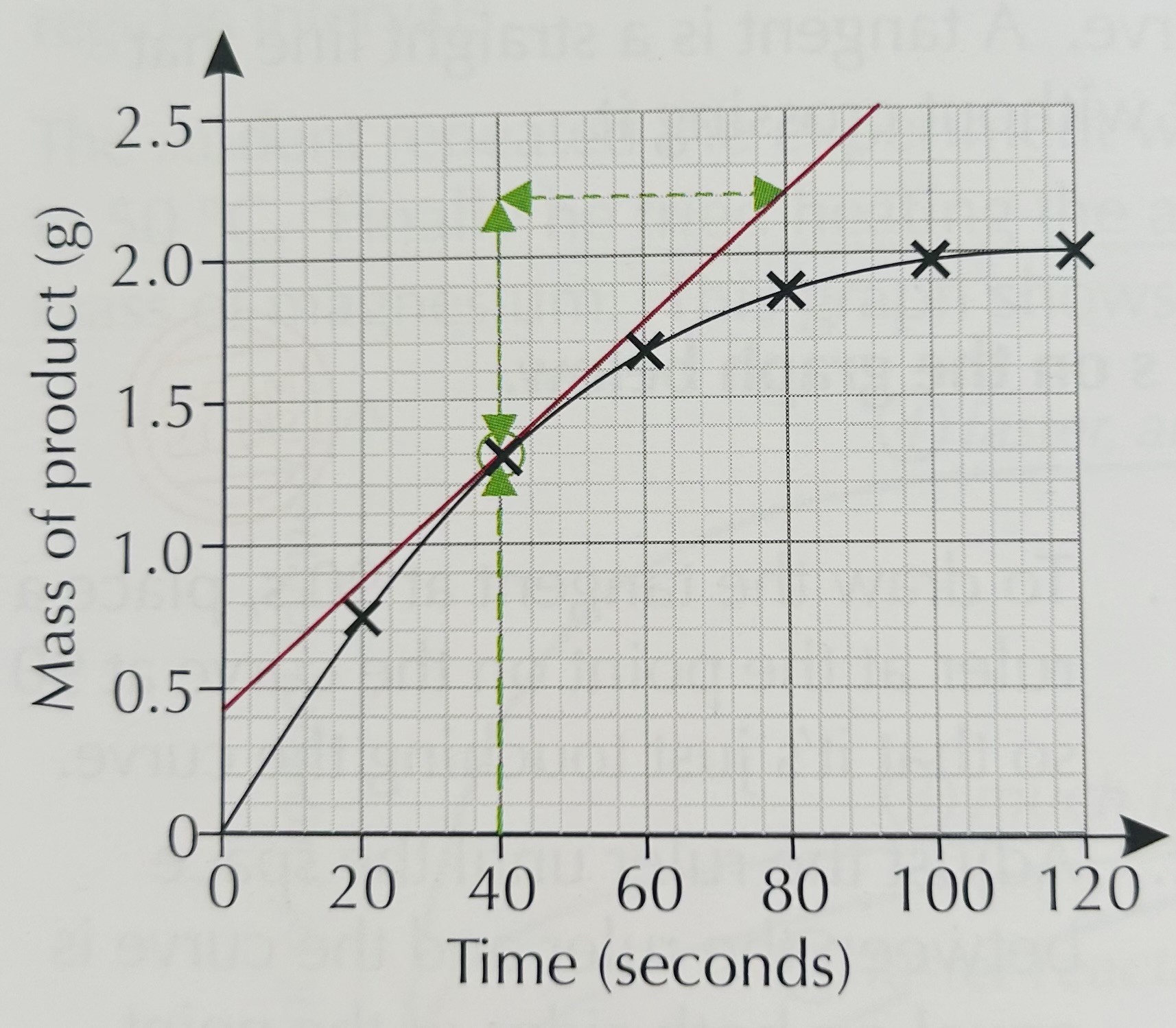
Calculate rate of reaction from a tangent
How to do it:
Draw a straight line that touches the curve at the point you are asked about
The middle of your straight line must touch the curve
Draw a triangle
Gradient = change in y / change in x
For example for this graph:
= (2.2 - 1.3) / (80-40)
= 0.90 / 40 = 0.023
Gas syringe experiment
Reaction between magnesium metal with dilute hydrochloric acid
The equation for this reaction: 2HCl + Mg → MgCl + H2
This is used to investigate the effect of increased reactant concentration on the rate of a reaction
Independent variable - concentration
Dependent variable - volume of gas produced
Control - time, length of magnesium, total volume
Method of gas syringe experiment

Cloudy experiment
Reaction between sodium thiosulfate and hydrochloric acid
The equation for this reaction: 2HCl + Na2S2O → 2NaCl + SO2 + S + H2O
This is used to investigate the effect of changing the reactant concentration of the sodium thiosulfate on the rate of a reaction
Why does the reaction go cloudy - SOLID SULFUR
Independent variable - concentration of sodium thiosulfate
Dependent variable - rate of reaction
Control - temp, person observing
Method of cloudy experiment

Why the control variable had to be controlled in the experiments
So its accurate and so you know what causes the changes
Example questions for experiments -
Describe what the results tell us how changing the concentration affects the rate of reaction
When the rate of concentration increases the rate of reaction increases too. When the rate is … the concentration is … and when the rate is … the concentration is …