T cells
1/53
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
54 Terms
How can you confirm MHC II
-Contains p56lck, ITAM, CD3, CD4+
-TCR is alpha beta
Explain how HSC cells differentiate to myeloid lineage and lymphoid lineage
The development from a hematopoietic stem cell (HSC) to a mature immune cell involves several distinct stages and branching pathways, primarily taking place in the bone marrow and thymus.
1. The Stem Cell and Progenitor Phase
The process begins in the bone marrow with a Long-Term Hematopoietic Stem Cell (LT-HSC), which can self-renew or differentiate into a Short-Term HSC (ST-HSC) and then into a Multipotent Progenitor (MPP). From the MPP stage, the pathway splits into two major lineages: Myeloid and Lymphoid.
2. The Myeloid Lineage
Cells committed to the myeloid lineage follow the Common Myeloid Progenitor (CMP) pathway. This branch produces most of the cells of the innate immune system:
Megakaryocyte-Erythrocyte Progenitor (MEP): This path leads to the formation of Erythrocytes (red blood cells) and Platelets (via Megakaryocytes).
Granulocyte-Monocyte Progenitor (GMP): This path generates Monocytes, which mature into Macrophages in tissues, as well as Neutrophils, Eosinophils, and Basophils.
Dendritic Cells: These professional antigen-presenting cells can arise from either the myeloid or lymphoid lineage.
T Cell Development (Thymus)
T cell precursors migrate from the bone marrow via the blood to the thymus to complete maturation.
Double Negative (DN) Stages: Early thymocytes (DN1-DN4) lack CD4 and CD8 co-receptors. During these stages, the cell chooses between the $\gamma\delta$ lineage and the $\alpha\beta$ lineage.
Double Positive (DP) Stage: $\alpha\beta$ thymocytes express both CD4 and CD8 as well as a complete T cell receptor (TCR).
Selection: DP cells undergo positive selection (to ensure they recognize MHC) and negative selection (to delete self-reactive clones).
Single Positive (SP) Mature Cell: Surviving cells become either CD4+ Helper T cells or CD8+ Cytotoxic T cellsand exit to the periphery.
4. Activation and Terminal Differentiation
Mature cells that have not yet encountered antigen are called naive cells. Upon recognizing an antigen presented by a Dendritic Cell in peripheral tissues (like lymph nodes), these cells undergo clonal expansion and differentiate:
Effector Cells: B cells become plasma cells that secrete antibodies. CD4+ T cells differentiate into specialized subsets such as Th1, Th2, Th17, Tfh, or Tregs to coordinate the immune response. CD8+ T cells become Cytotoxic T Lymphocytes (CTLs) that kill infected cells.
Memory Cells: A portion of the activated cells become long-lived memory cells, which provide a rapid response upon re-exposure to the same pathogen.
Exhaustion: Under conditions of chronic antigen persistence, T cells may enter a state of exhaustion, losing their effector functions.
Stem cell and progenitor phase
Myeloid lineage
Lymphoid lineasge
T cell development in thymus
Explain the T cell development pathway
In bone marrow: HSC develops into common lymphoid precursor
In blood: T cell precursor migrates to the thymus via the blood
In thymus: T cell precursor develops in thymus to SP cells by a series of steps:
→DN1: Its c-kit/CD44 positive, and CD25 negative
→DN2: Its c-kit/CD44/CD25 positive. Gamma, delta and beta gene recombination occurs, beta mature product associates with pre-alpha to form pre-TCR. Can develop to DP cell where alpha recombines for selection to occur and deletes delta locus. OR it makes a gamma delta T cell (very rare), which prevents alpha recombination.
→DN3: Its c-kit/CD44 negative, and CD25 positive.
→DN4: Its c-kit/CD44/CD25 positive
→Double positive: Selection occurs, expresses both CD4+ and CD8+ and recognizes either MHC class.
→Single positive: T cell is selected to be either a TH cell with CD4+ or a Tc cell with CD8+.
In blood / peripheral tissues: The mature CD8+ and CD4+ T cells, and gama delta T cells migrate to the peripheral tissues via the blood.
With biotechnology, how do we generate T cells for experimental purposes?
Stem cells are cultured with stromal cells that express a ligand for the mTCR Notch, which is a critical factor for committing a stem cell to the T cells lineage.
The growth factors IL-7 and FLT3L are added to the culture to support growth/development of cells for 1-2 weeks.
The T-lineage cells can now be harvested for study.
Explain the maturation of T cells (up to immature T cell development)
Stem cells
Double negative (CD4-CD8-) Pro-T cell: Thymocyte does not express CD4 or CD8 and does not have a TCR.
Pre-T cell: Develops Pre-TCR
Double positive (CD4+CD8+) immature T cell: Thymocytes express both CD4 and CD8 co-receptors and has a complete TCR on cell surface. Ready to undergo positive/negative selection to determine its final functional fate. Ensures that mature T cells are MHC-restricted and self-tolerant
Explain T cell selection (mature T cells)
T cell is a DP immature T cell ready to undergo selection
Positive selection: TCR has weak recognition of either class I or class II MHC + peptide. This causes either a mature CD4+ TH cells arise or mature CD8+ Tc cell that survive.
Negative Selection: TCR has strong recognition to either class I or class II MHC + peptide, reacting to self-antigens. These self-reactive cells receive signals to trigger apoptosis.
Failure of Positive Selection: TCR fails to recognize an MHC + peptide. It does not receive signals to survive, thus undergoes apoptosis bc it failed to be selected (“death by neglect”)
Explain the Avidity hypothesis
Suggests that selection is determined by the strength of the TCR signal
→No signal/too weak: Failure of positive selection, undergoes apoptosis (death by neglect)
→Optimal signal: Positive selection, gets signal to survive
→Strong signal: Negative selection, undergoes apoptosis
Explain the differential-signaling hypothesis
suggests that qualitatively different signals (e.g., TCR vs. CD8 signaling) may lead to survival or deletion, though selection is likely a combination of both avidity and differential signaling.
Suggests that instead of different levels of one signal (thru TCR), different signals (TCR vs CD8 vs CD28..) may lead to survival or deletion. Selection is likely a combination of avidity and differential signaling
→Example: Mice are genetically engineered to express CD8 at twice the normal levels and had 1/13 the typical level of CTLs. This inappropriate high signal level resulted in negative selection.
γδ vs αβ thymocyte fetal to adult development
The γδ and αβ thymocytes are in a running competition, where the timing of their appearance and dominance is tied to development. The % of CD3+ cells in the thymus shifts significantly during fetal development
→Days 14-17: γδ thymocytes are the dominant population, making up nearly 100% of the CD3+ cells in the early thymus.
→Day 16: αβ thymocytes begin to appear
→Day 18: αβ thymocytes are now 25% of the population
→Day 19: The 2 populations cross, αβ thymocytes become the dominant lineage and rise toward 100%, while γδ thymocytes decline to 5%.
What are the critical events in the DN stage and how are they caused?
-Critical events in DN stage are caused by signaling of the pre-TCR, the signals cause these 4 events:
Cell becomes permissive for TCR α-chain locus arrangement
Stimulates expression of CD4 and CD8 coreceptors
Stimulates proliferation
Stops additional TCR β-chain locus arrangements (allelic exclusion)
→If they do not receive signals, they undergo apoptosis
How to choose between γδ vs αβ?
→Commitment to γδ: Occurs when thymocyte successfully rearranges both its γ and δ genes before a functional β chain is produced, expressing a complete γδ TCR. Signals sent thru this receptor downregulates β expression, committing the cell to the γδ lineage.
→Commitment to αβ: Occurs when thymocyte successfully rearranges a functional β chain, it associates with a pre-Tα to form a pre-TCR. Signals thru the pre-TCR downregulate γδ expression and promote the rearrangement of the α chain, deleting δ.
How are nonproductive TCR α-chain’s rescued?
The rescue of nonproductive VαJα joins is a "rescue" mechanism that allows T cells to successfully develop even if their first genetic rearrangement fails to produce a functional protein. This is possible bc the TCR α-chain locus contains a multiplicity of inherited V and J gene segments.
→Leapfrog mechanism: If an initial join is nonproductive, the cell can "leapfrog" over the failed segments to attempt a new rearrangement. The cell can undergo multiple, successive rearrangement events bc theres multiple V and J segments.
What are the 4 steps of the leapfrog mechanism
Begins with the random selection and joining of one Va segment to one Ja segment.
The resulting gene is expressed, if the rearrangement is nonproductive it doesnt produce a functional a-chain protein.
Instead of the cell immediately undergoing apoptosis, the presence of additional V and J segments allows for another attempt. The recombination selects a Va and Ja segment of the failed joins.
The new Va and Ja segments are brought together, the intervening DNA is deleted from the chromosome.
→Success: If a productive rearrangement is achieved and resulting TCR can bind to an MHC molecule, the cell undergoes positive selection and survives.
→Failure: If the cell exhausts all possible gene segments without creating a functional receptor, it undergoes apoptosis.
What cell types do presentation during selection in the thymus?
Thymic epithelial cells: Play role in positive selection, interacts with DP thymocytes via Class I or II MHC molecules
Dendritic cells & Macrophages: Play role in neg selection, by presenting self-antigens to trigger apoptosis of self-reactive T cells.
How do thymic epithelial cells express all antigens/proteins from all of the different cell types (>150) in humans?
Thymic epithelial cells expresses AIRE protein, which allows the thymus to test developing T cells against antigens they will encounter throughout the body, preventing autoimmunity.
What are the two pathways of apoptosis, include caspases
The Extrinsic (Death Receptor) Pathway
The Intrinsic (Mitochondrial) Pathway
→FINAL STAGE (after either pathway): Once Caspase-3 is activated by either pathway, it leads to the final stage of apoptosis. It cleaves vital structural and functional proteins and activates apoptotic effectors. Cell undergoes physical changes and then breaks apart into several apoptotic bodies to be phagocytosed.
What are the 4 steps of the extrinsic (Death Receptor) apoptosis pathway?
Begins when FasL ligand binds to its receptor, Fas, on the target cell.
The intracellular "death domain" of the Fas receptor recruits FADD
FADD recruits and activates Procaspase-8, cleaving it into active caspase-8
→Caspase 8 can directly cleave/activate Caspase 3 OR cleave Bid into Trancated Bid to move to the mitochondrion (intrinsic pathway) to amplify apoptotic signal
Caspase 3 is activated which leads to final stage of apoptosis.
What are the 4 steps of the intrinsic (mitochondrial) apoptosis pathway?
Internal signals cause mitochondrion to release cytochrome c into the cytosol, which then binds to Apaf-1 and Procaspase-9.
This complex forms the apoptosome, which activates caspase-9
Active Caspase-9 cleaves and activates Caspase-3.
Caspase 3 is activated which leads to final stage of apoptosis.
What are zygomens, what does procaspase zygomen do?
Zygomens are inactive precursors of enzymes that must be cleaved to become catalytically active.
→Caspases are synthesized as inactive zygomens called procaspases.
What are CARD and DED
→CARD is found in apoptosome in the intrinsic pathway, and DED is found in procaspase-8 which is used in the extrinsic apoptotic pathway. These domains facilitate the proteolytic cleavage of the zygomens into active initiator caspases.
→The presence of these domains distinguishes initiator caspases
What are the 5 markers of apoptosis?
Cells undergo morphological changes such as cell shrinkage, chromatin condensation and nuclear fragmentation
Cell surface develops blebs, protrusions of the membrane that contain intact, functional organelles.
Cell breaks into apoptotic bodies
DNA is extensively nicked and fragmented
TUNEL assay can recognize these cells, TdT adds labeled nucleotides to the 3' ends of the DNA fragments to visualize.
What are the differences between necrosis and apoptosis
→Features: nature, blebbling, organelles, nucleus, membrane integrity, final outcome
Feature | Necrosis | Apoptosis |
|---|---|---|
Nature | Accidental/Passive death | Programmed/Active suicide |
Blebbing | Small blebs fuse to become large | Small blebs form and remain discrete |
Organelles | Not located in blebs; become non-functional | Located within blebs; remain functional |
Nucleus | Structure changes but remains largely intact until rupture | Breaks apart; DNA fragments into small pieces |
Membrane Integrity | Ruptures, releasing contents into environment | Cell breaks into apoptotic bodies |
Final Outcome | Cellular contents leak, causing inflammation | Bodies are phagocytosed |
What does Canale-Smith Syndrome (CSS) cause?
Patients with CSS have a mutated Fas receptor that cant transmit death signals. Thymic selection fails, leading to an abnormal accumulation of DN T cells and a lack of proper immune tolerance.
→Fas (protein) FasL are the primary signals for apoptosis from the T cell
How do cancer cells evade Fas-mediated apoptosis?
Cancer cells are expected to develop mechanisms that evade Fas-mediated apoptosis to survive and proliferate despite being targeted by the immune system.
Express decoy receptors: Cancer cells produce decoy Fas receptors to bind to FasL on CTLs but lack the intracellular death domain, soaking up the FasL and preventing it from binding to actual functional FasR’s on cell surface.
Inhibitory proteins (v-FLIP): Cancer cells express v-FLIP to inhibit the activation of caspase-8, preventing apoptosis.
Fas mutation: Similar to CSS, render cells non-responsive to death signal
Inducing T cell exhaustion: They contribute to T-cell exhaustion, making them lose their effector functions.
What are the two phases of cell-mediated immunity?
Induction phase in lymphoid organs: T cell activation and differentiation
Effector phase in peripheral tissues: Elimination of microbes
What are the 3 steps in the Induction phase in lymphoid organs (CMI)
Naive CD4+ and CD8+ T cells recognize antigen-peptides presented by DCs, activated by second signal (costimulation) by binding B7 (on DCs) to the CD28 receptor on the T cell.
Activated T cells secrete cytokines, like IL-2, and express IL-2 receptors. This cytokine acts as a growth factor, driving proliferation.
The proliferated cells differentiate into effector cells and memory cells.
→Effector CD4+ cells become specialized subsets (like Th1, Th2, or Th17),
→Effector CD8+ cells become Cytotoxic T Lymphocytes (CTLs).
What are the 2 steps in the Effector phase in peripheral tissues (CMI)
Differentiated effector T cells enter the circulation and migrate to the site of antigen
Effector T cells encounter antigens in peripheral tissue and are activated
CD4+ TH cells: Recruit and activate phagocytes to destroy ingested microbes. Th1 cells secrete IFN-γ, the primary cytokine for activating macrophages for phagocytosis
CD8+ TC cells: Directly kill infected target cell by releasing granules to trigger apoptosis.
What happens if T cells dont receive the costimulation signals?
Costimulation is the second signal thats required for T cell activation. If they dont receive this second signal, the T cells may become unresponsive or tolerance
What are all the receptors and ligands involved in T cell activation
Antigen recognition
Costimulatory receptors
Adhesion molecules
Inhibitory receptors
Antigen recognition (signal 1)
→TCR αβ: Recognizes MHC-peptide complex
→CD3 and ζ chains: Associated with TCR for signal transduction. Contains ITAMS which serve as docking sites for kinases
→CD8+ or CD4+ receptors: Bind to class I or II MHC molecules respectively. Bring Lck kinase to phosphorylate ITAMs
Costimulatory Receptors (Signal 2)
→CD28: Costimulatory receptor on resting T cells, binds to B7-1 or B7-2 on APCs
Adhesion Molecules
→LFA-1: Found on T cell, it binds to ICAM-1 on the APC
Inhibitory Receptors (Negative Regulation)
→CTLA-4: Expressed on activated T cells and competes with CD28 for binding to B7. Dampens immune response.
→PD-1: This receptor binds to PD-L1 or PD-L2, inhibis T cell response
(extra: CD40L binds to CD40 on APCs like B cells)
What is the difference between single-chain anti-CTLA-4 and anti-CTLA-4 Ab?
Features: Valency, cross-linking, primary effect
Feature | Anti-CTLA-4 Antibody | Single-Chain Anti-CTLA-4 |
|---|---|---|
Valency | Bivalent (2 binding sites) | Monovalent (1 binding site) |
Cross-linking | Can cross-link receptors | Cannot cross-link receptors |
Primary Effect | Acts as an agonist, triggers inhibitory signal to suppress the T cell | Acts as an antagonist, blocks the "brake" to activate the T cell |
what is the first event in signaling activation
→Phosphorylation of ITAMs: The CD4/8-associated p56Lck kinase phosphorylates the ITAMs of ζ chains, creating docking site for ZAP-70
(besides T cell recognizing MHC-peptide complex)
What are lipid rafts?
Theyre sections in the membrane that are more structured and rigid, serving as a platform for signaling events. They have 300-500% more cholesterol and 50% more sphingolipids. Theyre mobile in the surrounding fluid membrane.
What do small secondary messengers do?
small secondary messengers like calcium amplify and diffuse a signal throughout the cell.
What do cascades of kinase do?
They amplify signals. Signal transduction pathways are OFF unless activated, but since the proteins are all synthesized but inactive, they can be rapidly activated, a “hair-trigger”. Signaling relines on protein post-translation modifications.
What are immediate, early and late genes?
Immediate genes: Focused on transcription factors and replication
→ex: c-Fos and c-Jun (TFs form AP-1 complex), NFAT (transcriptional activation)
Early genes: Involved in T cell growth and signaling to other cells
→ex: IL-2 (proliferation)
Late genes: Involved in T cell migration
→ex: HLA-DR (class II MHC molecule), VLA-4 (adhesion molecule)
What is the contraction phase (homeostasis) of T cells
As the infection is successfully eradicated, the T-cell population undergoes a dramatic contraction to restore the system to its resting state. Theres a withdrawal of survival signals leading to apoptosis, and regulatory “brakes”.
→This decline occurs withing 1-2 weeks after the infection is cleared, preventing prolonged inflammation/tissue damage. Memory T cells remain.
Explain naive, resting and activated T cells
Naive: Mature T cells that have yet to encounter their specific antigen, strictly activated by DCs. Theyre the primary immune response
Resting: Theyre memory or effector T cells that are not currently active. They can be re-activated by any APC, theyre the secondary immune response, faster and more potent than the primary one.
Activated: T cells that have recognized their specific MHC-peptide complex and received costimulation. Lead to proliferation and differentiation
What is T cell exhaustion?
The stepwise and progressive loss of T-cell functions when subjected to chronic antigen stimulation. Seen in chronic viral infections (HIV) and tumours
→Determining severity: duration/magnitude of antigenic activation, availability of CD4 T-cell help, levels of stimulatory/suppresive cytokines, expression of activatory&inhibitory receptors.
What are superantigens?
Superantigens trigger a massive, non-specific activation of T cells independent of antigen. Such stimulation causes immune dysregulation due to overactivation and overstimulation.
Why can non-specific activation of the immune system be bad?
This overwhelming inflammatory response results in systemic diseases such as Toxic Shock Syndrome or severe food poisoning
what is CD45R0?
A specific surface protein used as a marker to identify memory (M) and effector (E) T cells. This marker distinguishes these "experienced" cells from naive T cells, which have not yet encountered their specific antigen.
What are the 3 professional APCs
They express Class II MHC molecules and provide the costimulatory signals (B7) necessary to activate TH cells.
Dendritic cells: The most potent APCs, presenting to naive, effector and memory T cells, perform phagocytosis.
Activated macrophages: Must be activated by IFN-γ, present to effector and memory T cells, perform phagocytosis.
B cells: Can be resting or activated presenting to memory and effector TH cells for antibody production and isotype switching. (resting do not require B7)
What are the non-professional APCs
Nearly all nucleated cells, involved with Class I MHC to activate TC cells. (macrophage must be resting)
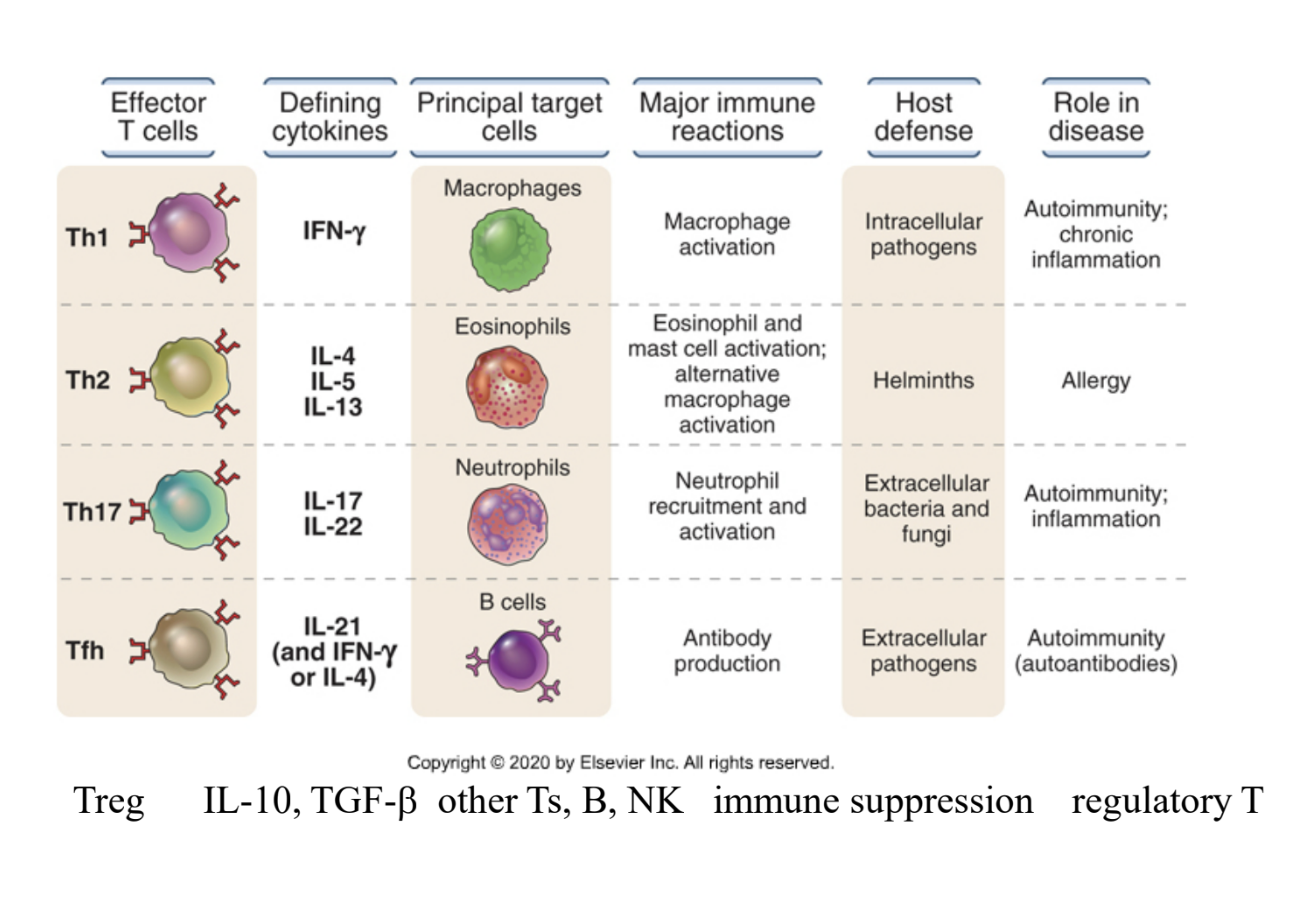
Provide the following for Th1, Th2, Th17, Tfh, and Treg
→Defining cytokines
→Principal target cells
→Major immune reactions
→Host defense
→Role in disease
What is the function of Th1 cells (APC)
They produce IFN-γ, which activates macrophages to kill phagocytosed microbes (classical pathway). Sometimes IFN-γ stimulates the production of IgG antibodies, but TH cells may be the source of this cytokine
(primary mediator of CMI against intracellular pathogens)
Provide 5 steps of macrophage activation by Th1 cells
The Th1 TCR recognizes a specific peptide presented on the Class II MHC molecule of the macrophage.
Th1 cell expresses CD40L on its surface, binding to the CD40 receptor on the macrophage
Th1 cell secretes IFN-γ which binds to IFN-γ receptors on the macrophage
The combination of the CD40 signal and the IFN-γ signal triggers a biochemical cascade within the macrophage that "activates" it.
Activated macrophage releases pro-inflammatory cytokines like TNF, IL-1, and IL-12. Or produce ROS, NO to kill the microbes.
Explain the development of Th1, Th2, and Th17 effector cells
→Th1 cells: Induced by IL-2 (from DC and macrophages) and IFN-γ (from NK) in response to intracellular microbes. Activates STAT1 or STAT4 transcription factors which commit the cell to the Th1 lineage.
→Th2 cells: Differentiation into the Th2 subset is triggered by IL-4 in response to helminths or protein antigens. This environment activates the transcription factors GATA-3 and STAT6
→Th17 cells: Develops in the presence of pro-inflammatory cytokines (IL-1), in response to extracellular microbes. Express STAT3 transcription factors.
What is the 5 functions of Th2 cells
Antibody Production (IgE): Th2 cells produce IL-4, which stimulates B cells to undergo heavy-chain isotype switching to produce IgE antibodies.
Mast Cell Activation: The IgE produced binds to mast cells, and further secretion of IL-4 and IL-13 by Th2 cells promotes mast cell degranulation, releasing mediators that contribute to inflammation.
Eosinophil Activation: Th2 cells secrete IL-5, the primary cytokine for the activation and recruitment of eosinophils to the site of infection to destroy helminths.
Intestinal Response: IL-4 and IL-13 act on the gut to increase intestinal mucus secretion and peristalsis, helping to physically expel parasites from the body.
Alternative Macrophage Activation (M2): Use IL-4 and IL-13 to induce "alternative" macrophage activation, promoting tissue repair and fibrosis following injury.
(coordinate against helminthes and key players in allergic reactions)
Explain the classical and alternative macrophage activation
Classical Macrophage Activation (M1): Induced by microbial products binding to TLRs and cytokines (IFN-γ) which are produced by Th1 cells. Produces high levels of ROS, NO and lysosomal enzymes to destroy microbes in its phagosomes. It can also secrete cytokines (IL-1, IL-12) to promote inflammation.
Alternative Macrophage Activation (M2): Induced by IL-4 and IL-13 which are produced by Th2 cells. They promote tissue repair and fibrosis. They secrete suppressive cytokines (IL-10) to inhibit inflammation.
What are the 3 functions fo Th17 cells
Inflammation/neutrophil response: It produces IL-17 which induces production of chemokines (TNF), which recruit neutrophils into the site of inflammation
Antimicrobial peptides: It produces IL-17 & IL-22 which stimulates the production of antimicrobial peptides to directly kill microbes.
Increased barrier integrity: It produces IL-22 which act on epithelial cells in barrier tissues, and promoted the repair of damaged epithelia.
(prevents infections in tissues like skin and mucosal surfaces)
Explain the development of regulatory T cells
Tregs maintain immune balance by suppressing immune responses and preventing autoimmunity. They differentiate from CD4+ T cells that recognize self-antigens in the thymus or peripheral tissues, theyre dependent on FoxP3.
→They inhibit the activation of naive T cells and their differentiation into effector T cells by contact-dependent mechanisms or by secreting cytokines that inhibit T cell responses.
What is the mechanism of killing infected cells by TC cells
CD8+ CTL recognize peptides on Class I MHC molecule of the infected cell. The CTL forms conjugate with target cell via LFA-1 (adhesion molecule) on the T cell binding to ICAM-1 on target cell.
CTLs release their granule contents toward the infected cell. Granzymes are delivered to the cytosol of the target cell by perforin-dependent mechanism, then inducing apoptosis.
Explain the central tolerance of self antigens
Central tolerance occurs in the lymphoid organs—the thymus for T cells and the bone marrow for B cells. During this phase, immature lymphocytes that recognize self-antigens with high affinity are handled through several mechanisms:
Apoptosis (Deletion): This is the primary mechanism for both T and B cells. Through a process called negative selection, immature lymphocytes that react strongly to self-MHC molecules or self-peptides are signaled to undergo programmed cell death.
Receptor Editing: This mechanism is specific to B cells. If an immature B cell recognizes a self-antigen, it may be given a chance to change its specificity by undergoing further rounds of light-chain gene rearrangement.
Development of Regulatory T Cells (Tregs): Some immature CD4+ T cells that recognize self-antigens in the thymus do not die but instead differentiate into regulatory T cells. These Tregs then migrate to the periphery to suppress any remaining self-reactive cells.
Explain the peripheral tolerance of self antigens
Peripheral tolerance occurs in the peripheral tissues and acts as a secondary "safety net" for mature self-reactive lymphocytes that managed to escape central tolerance. The mechanisms include:
Anergy (Functional Inactivation): If a mature T cell recognizes an antigen but does not receive the necessary costimulation (Signal 2, such as CD28 binding to B7), it enters a state of long-term unresponsiveness called anergy.
Apoptosis (Deletion): Mature lymphocytes that encounter self-antigens repeatedly or in a particular context may be signaled to undergo apoptosis, physically deleting them from the repertoire.
Suppression: Regulatory T cells (Tregs) that were generated in the thymus or periphery actively inhibit the activation and effector functions of self-reactive T and B cells through contact-dependent mechanisms or the secretion of suppressive cytokines like IL-10 and TGF-β.
Inhibitory Receptors: Activated T cells express inhibitory receptors like CTLA-4 and PD-1. These receptors provide a "brake" on immune responses, and their induction is critical for maintaining tolerance to self-antigens in peripheral tissues.