Thermochemistry
0.0(0)
Studied by 5 peopleCard Sorting
1/57
Earn XP
Last updated 11:51 PM on 1/17/23
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
58 Terms
1
New cards
thermochemistry
study of energy changes during physical, chemical, and nuclear changes
2
New cards
energy
ability to do work (J or kJ)
3
New cards
thermal energy
total kinetic and potential energy of substance. can be transferred to surroundings as heat
4
New cards
heat
transfer of thermal energy from one substance to another
5
New cards
system
the chemical process whose heat we’re examining
6
New cards
surroundings
anything outside the system (e.g. water)
this gets measured
this gets measured
7
New cards
3 types of thermodynamic systems
open, closed, isolated
8
New cards
open system
energy and matter (reactants/products) enter and leave freely
9
New cards
closed system
energy enters and leaves freely but matter (reactants/products) does not
10
New cards
example of closed system
glowstick, ice cooler
11
New cards
example of open system
barbeque
12
New cards
isolated system
neither energy nor matter can leave or enter the system. ideal theoretical state
13
New cards
2 types of thermochemical processes
exothermic and endothermic
14
New cards
exothermic reaction
heat released to surroundings from the reactant bonds/IMFs, potential energy converted to kinetic energy
15
New cards
exothermic reaction example
fire, combustion
16
New cards
endothermic reaction
heat absorbed from surroundings (requires energy input), kinetic energy converted to potential energy
17
New cards
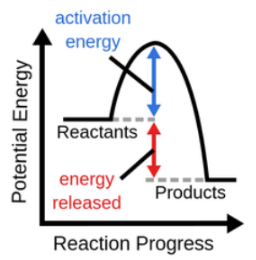
\
exothermic
18
New cards
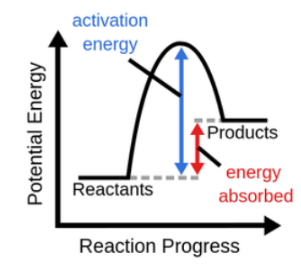
\
endothermic
19
New cards
temperature
average kinetic energy of particles in a substance
20
New cards
calorimetry
measuring energy changes in a chemical system
21
New cards
parts of calorimeter
well-insulated container (ideally isolated system), water, thermometer
22
New cards
general heat equation (q of surroundings)
q = mcΔT
23
New cards
enthalpy
thermal energy in a chemical substance
24
New cards
types of thermochemical kinetic energy
translational, rotational, vibrational
25
New cards
types of thermochemical potential energy
bonds, IMFs
26
New cards
how does the enthalpy change of the system compare to the heat of the system and surroundings?
ΔH of sys = q of sys = -q of surr
27
New cards
molar enthalpy
change in enthalpy per mol
28
New cards
specific heat capacity of water
4\.18 J/(g)(C)
29
New cards
molar enthalpy equation
ΔH per mool = (q of sys) / n (moles)
30
New cards
three ways to show enthalpy changes
potential energy diagrams, adding energy as reactant/product, stating the ΔH separately
31
New cards
who won the 1918 nobel prize in chemistry for his work in ammonia synthesis?
haber
32
New cards
name the fertilizer made by Haber using his namesake proccess
Zyklon A
33
New cards
hess’s law of summation
The change in molar enthalpy is the same regardless of whether the change happens in one step or multiple steps.
34
New cards
reasons to use Hess’s law
1. reaction is too high/low energy
2. reaction is too fast/slow
35
New cards
what is standard enthalpy of formation
energy needed to make one mol of a substance at SATP (25°C, 100 kPa)
36
New cards
what should be noted regarding cancelling out common compounds using Hess’s law?
they must be the same state (i.e. enthalpy of state changes not accounted for)
37
New cards
equation to find enthalpy change using enthalpy of formation
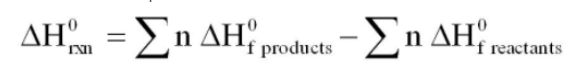
38
New cards
three things required for reactions to happen
1. reactant particles must collide
2. they must collide with enough kinetic energy
3. they must collide with the right orientation
39
New cards
transition state
state of reactants during the reaction where the chemical changes happen
40
New cards
transition state theory
1. bonds are broken and formed in chemical reactions
2. particles need enough kinetic energy to overcome the electrostatic forces between them
41
New cards
activation energy
minimum energy input needed to start reaction
42
New cards
when is the peak of potential energy in a reaction
during the transition state
43
New cards
5 ways to change rate of reactions
1. temperature
2. add a catalyst
3. change reactant concentrations
4. change reactants surface area
5. nature of reactants (reactivity differences in different substances)
44
New cards
how does temperature affect reaction rate
Temperature ∝ kinetic energy of particles ∝ # of collisions with enough energy
and
Temperature ∝ kinetic energy of particles ∝ # of total collisions ∝ # of successful collisions
and
Temperature ∝ kinetic energy of particles ∝ # of total collisions ∝ # of successful collisions
45
New cards
how do catalysts affect reaction rate
makes reactions faster since it provides reaction with an alternative reaction mechanism that happens occurs with a lower Activation Energy (same amount of energy = more successful collisions = faster reaction)
46
New cards
how does concentration affect reaction rate
Concentration ∝ # of particles ∝ # of collisions ∝ # of successful collisions
47
New cards
how does reactant surface area affect reaction rate
Surface area ∝ space for collisions ∝ # of collisions ∝ # of successful collisions
48
New cards
unit for reaction rate
mol/L/s or mol/L\*s
49
New cards
what do maxwell-boltzman distributions show
what proportion of molecules will collide with enough energy
50
New cards
describe a maxwell-boltzmnn distribution graph
\# of molecules within a range of kinetic energy, with a set x value for the Activation Energy (particles with more activation energy can collide with enough energy)
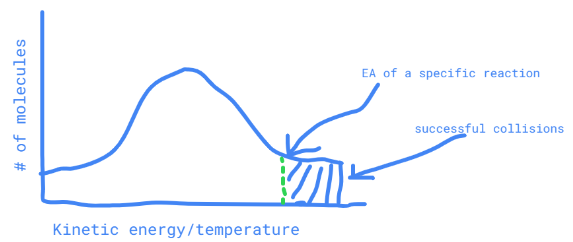
51
New cards
describe a maxwell-boltzmann distribution where energy has been increased
the curve is shifted right but the activation energy remains the same (the activation energy line stays in place)
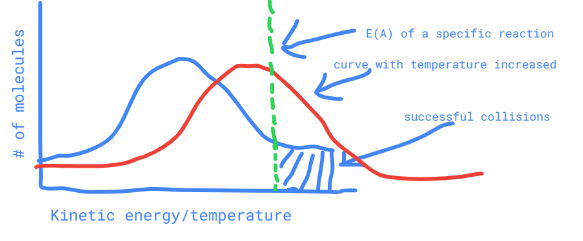
52
New cards
describe a maxwell-boltzmann distribution where a catalyst has been added
the activation energy line is shifted left while the curve stays in place
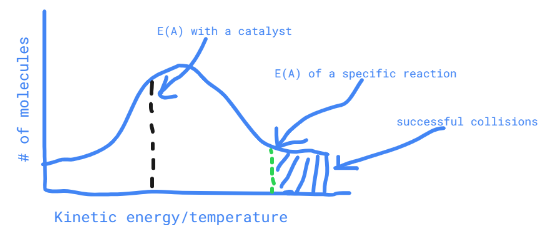
53
New cards
how do you find the average rate of a reaction?
r = Δ\[\]/Δt
54
New cards
what is the rate law?
r = k(\[A\]^n)(\[B\]^m)
55
New cards
rate symbol
r
56
New cards
properties of constant k
* determined experimentally and unique for each reaction
* units vary based on order (degree) of reaction
* 2nd derivative of reaction curve (mol of reactant vs time for reaction)
* units vary based on order (degree) of reaction
* 2nd derivative of reaction curve (mol of reactant vs time for reaction)
57
New cards
what is order (degree) of reaction?
sum of reactant exponents in rate law
58
New cards
how to find rate law?
1. find the reaction rate ratio and conc. ratio in two trials where the conc. of only 1 reactant changed
2. find the exponential relationship between the conc. ratio and reaction rate ratio
3. repeat 1-2 for each reactant (this is the order of the reaction with respect to each reactant)
4. find k by subbing in the values for any trial and isolating k (**remember to include units in the calculations and how they change**)