Bio ch 1
1/12
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
13 Terms
(Radioactive) Isotopes
two atoms of an element that differ in number of neutrons; when radioactive, they decay spontaneously, giving off particles and energy, often used as diagnostic tools in medicine
Radioactive tracers are used to track atoms through metabolism, in w imaging instruments such as PET scanners, which monitor cancer’s growth and metabolism
Radiometric Dating
parent isotope decays into daughter isotope at a fixed rate, the half-life of the isotope; scientists measure the ratio of different isotopes, calculating how many half-lives passed since the fossil/rock was formed
Concept 2.1: Matter consists of chemical elements in ___ form and in combinations called _____
Matter consists of chemical elements in pure form and in combinations called compounds
Concept 2.2: An element’s properties depend on the ______ of its atoms
An element’s properties depend on the structure of its atoms
Concept 2.3: The f______ and f______ of molecules and ionic compounds depend on chemical ______ing between atoms
Concept 2.3: The formation and function of molecules and ionic compounds depend on chemical bonding between atoms
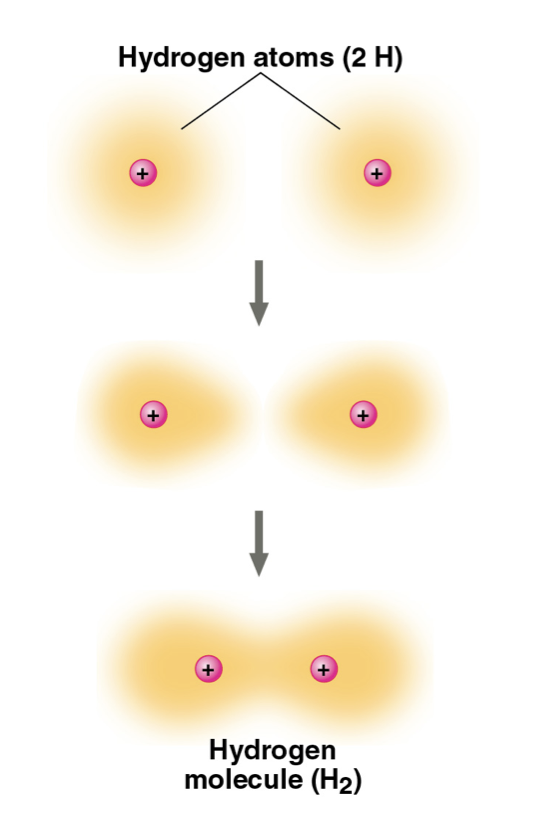
Covalent Bonds ★
the sharing of a pair of valence electrons by two atoms, which count as part of each atom’s valence shell
Single covalent bond = sharing of one pair of valence electrons
Double covalent bond = sharing of two pairs of valence electrons
Nonpolar covalent bond = atoms share electron equally
Polar covalent bond = atoms share electron unequally; one atom is more electronegative
(resulting in partial pos/neg charge for each atom/molecule)
(molecule = 2 or more atoms held tgt by covalent bonds)
Electronegativity
an atom’s attraction for the electrons in a covalent bond; an atom’s tendency to pull the shared electron towards itself
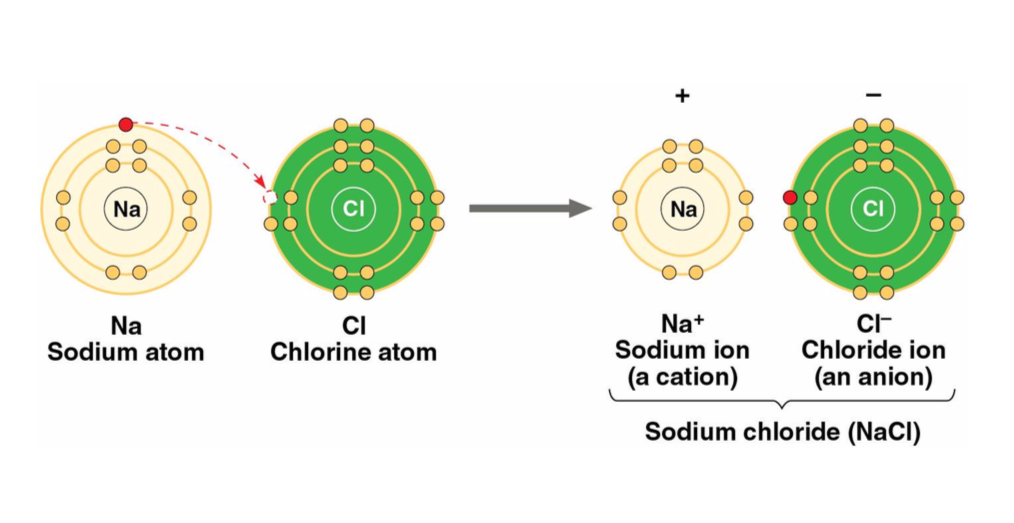
Ionic Bonds
the stripping of electrons from bonding partners resulting in oppositely charged atoms/molecules, ions, specifically cations and anions that attract each other
cation = positively charged ion
anion = negatively charged ion
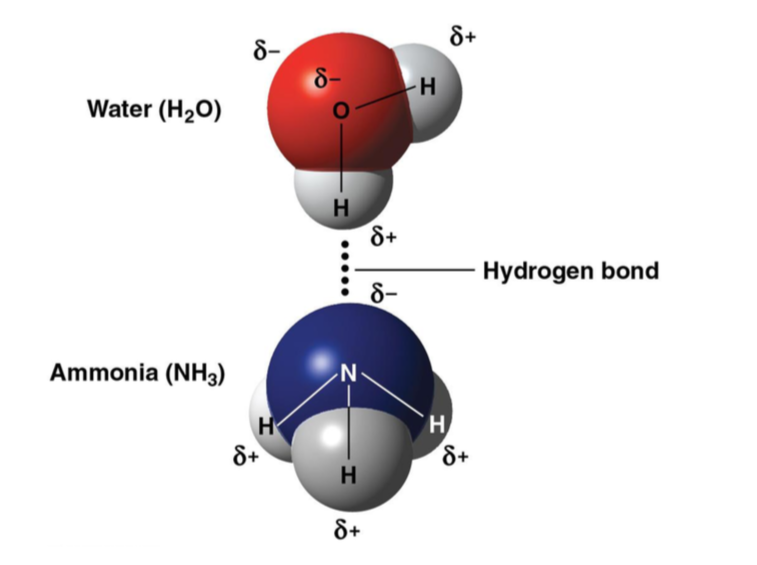
Hydrogen Bonds
bond formed when a hydrogen atom covalently bonded to one electroneg atom is also attracted to another electroneg atom (eg water and ammonia)

Vand der Waals Interactions
accumulation of electrons by chance in one part of the molecule due to unevenly distributed electrons, resulting in attractions between close tgt molecules from these charges
collectively strong (eg molecules of a gecko’s toe hairs and wall surface)
A molecule’s ___ and ___ are key to its function
A molecule’s size and shape are key to its function
key concept: structure determines function
A molecule’s shape is determined by the positions of its atom’s orbitals, and define how biological molecules recognize and respond to one another (eg opiates like morphine and natural morphine have similar effects bc their shapes are similar and thus bind the same brain receptors)
Concept 2.4: Chemical reactions ____ and ____ chemical _____.
Chemical reactions make and break chemical bonds
eg 6CO2 + 6H2O —> C6H12O6 [glucose] + 6O2 (sunlight is written above the arrow)
Chemical equilibrium
forward and reverse reaction occur at the same rate (while relative concentrations of reactants and products don’t change)