Hyperandrogenism, Infertility, Contraception, and Sterilization.
1/97
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
98 Terms
What is hyperandrogenism?
The clinical manifestation of elevated circulating levels of male hormones in women.
What is the clinical difference between hirsutism and virilization?
Hirsutism is excessive terminal hair in a male-patterned distribution, while virilization involves the acquisition of male characteristics like clitoromegaly, deepening voice, and changes in body fat distribution.
What are the three primary sources of androgen production in women?
Adrenals (25%), ovaries (25%), and peripheral tissues (50%) via conversion in adipose and skin.
What are inactive androgens mostly bound to?
Proteins like albumin and sex hormone binding globulin.
What enzyme converts testosterone into the more potent dihydrotestosterone (DHT)?
5a-reductase.
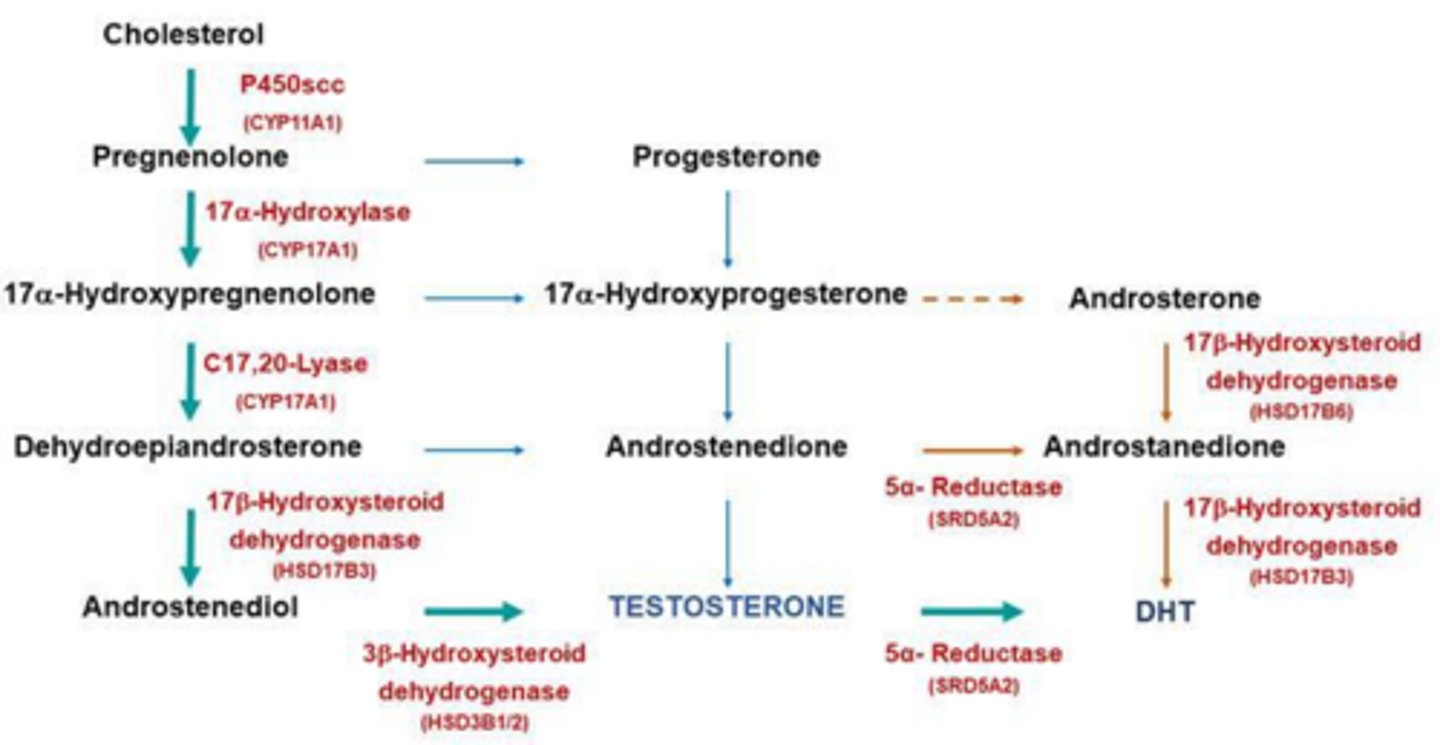
What is the most common genetic enzyme deficiency causing Congenital Adrenal Hyperplasia (CAH)?
21-hydroxylase deficiency, causes 95% of cases.
How does decreased cortisol lead to hyperandrogenism?
Decreased cortisol -> loss of negative feedback -> increased ACTH -> adrenal gland overstimulation -> adrenal hyperplasia. Accumulated precursors are shunted into androgen production -> hyperandrogenism.
When is class CAH (Congenital adrenal hyperplasia) diagnosed?
At birth or early infancy.
What are the clinical hallmarks of 'salt-wasting' CAH?
Complete enzyme deficiency leading to life-threatening hypotension, hyponatremia, and hyperkalemia.
What is the simple virilizing type associated with classic CAH?
Partial deficiency, presents with ambiguous genitalia in a female newborn but NO salt-wasting crisis.
What is the treatment for SW CAH (salt-wasting CAH)?
Lifelong hydrocortisone and aldosterone replacement therapy
What is the treatment for SV CAH (simple virilizing CAH)?
Hydrocortisone replacement.
When is non-classic CAH diagnosed?
At puberty or later in life when patient presents with hirsutism, acne, and irregular menses/infertility.
Do those with non-classic CAH have ambiguous genitalia or salt wasting?
No
How is non-classic CAH diagnosed?
Non-classic CAH is diagnosed by elevated 17-hydroxyprogesterone levels (specifically in the early AM)
What is the treatment for non-classic CAH?
OCPs and spironolactone. Add low dose dexamethasone when patient is seeking to restore fertility.
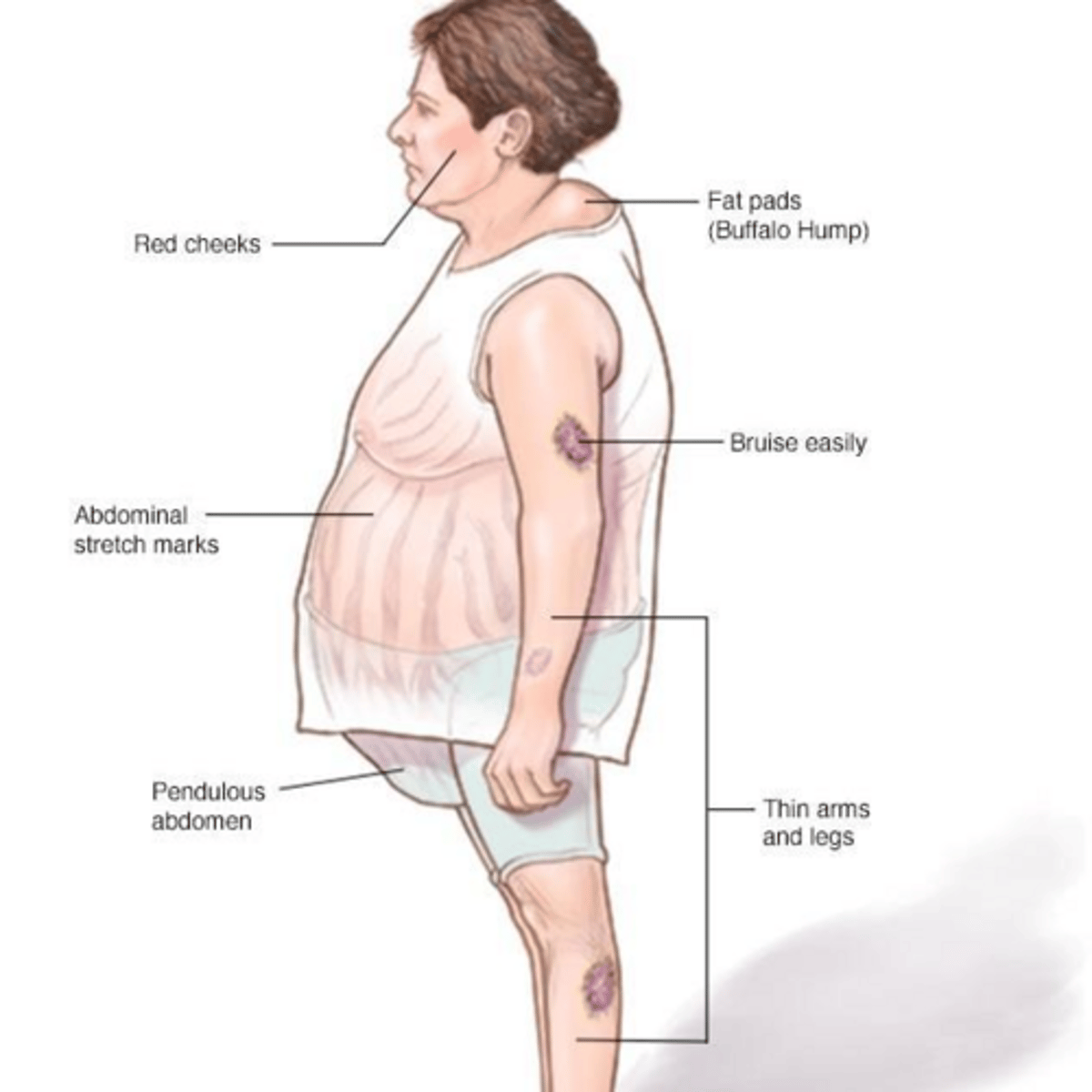
What are the 'classic' clinical signs of Cushing Syndrome?
Central obesity (moon facies, buffalo hump), purple abdominal striae, easy bruising, proximal muscle weakness, and hypertension.
What diagnostic marker strongly suggests an adrenal neoplasm (carcinoma)?
DHEA-S levels greater than 700 mcg/dL.
What are the red flags for adrenal carcinoma?
Rapid onset (weeks to months) of severe hirsutism, progression to virilization, and a palpable abdominal or pelvic mass.
What is PCOS?
A state of chronic anovulation associated with androgen excess (most common cause of hirsutism)
What is the gonadotropin mismatch observed in PCOS?
Increased GnRH pulse frequency leads to elevated LH and a mismatched LH:FSH ratio (typically 2:1 or 3:1).
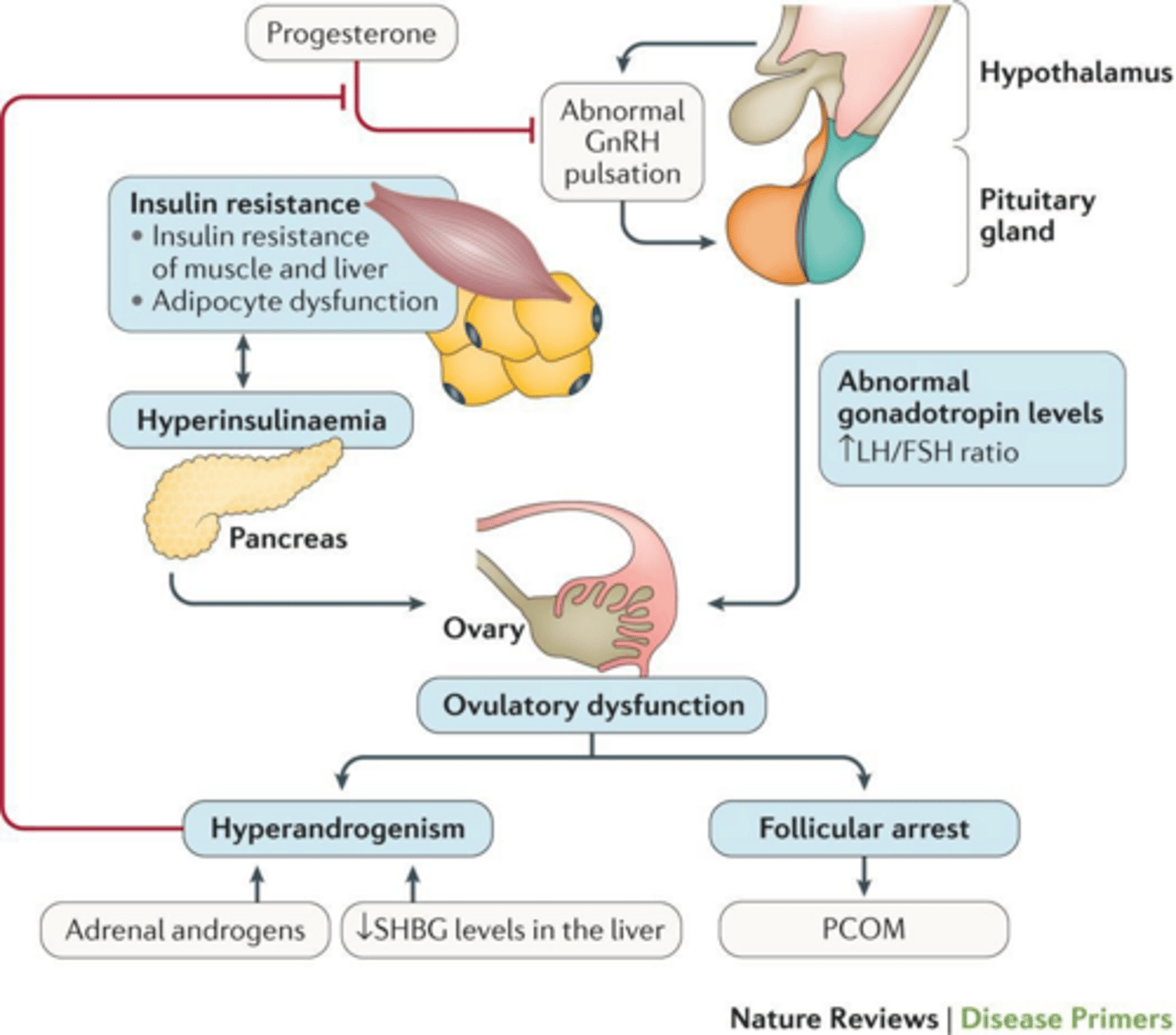
How does hyperinsulinemia contribute to the pathophysiology of PCOS?
It directly stimulates theca cell androgen production and decreases SHBG levels, which increases the amount of free, active testosterone.
What are the Rotterdam Criteria for diagnosing PCOS?
Requires 2 out of 3: 1) Oligo- or anovulation (<9 periods/year), 2) Clinical or biochemical hyperandrogenism, 3) Polycystic ovaries on ultrasound.
What is the classic ultrasound appearance of ovaries in PCOS?
The 'string of pearls' appearance, defined as 12 or more follicles (2-9 mm) or increased ovarian volume (> 10 mL).
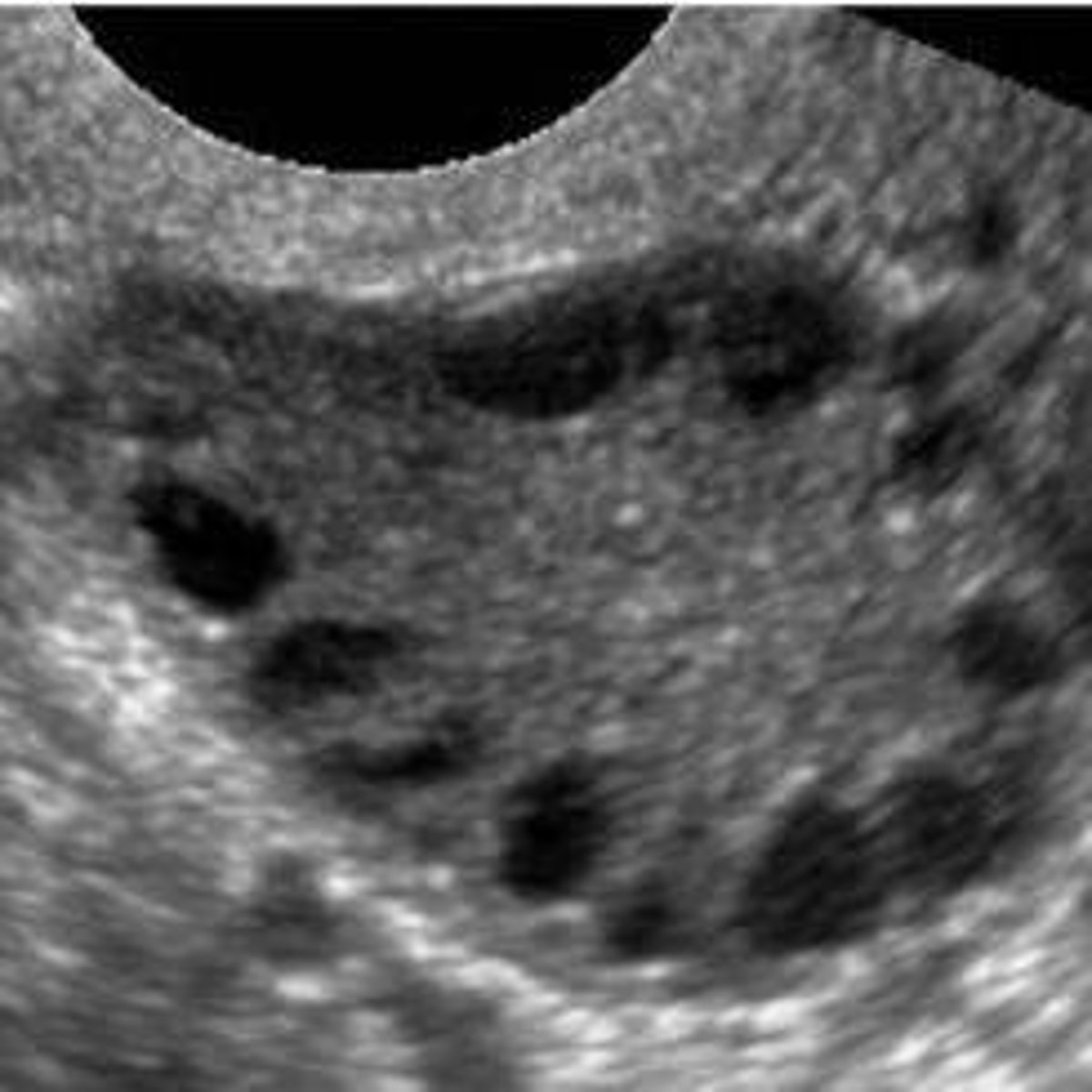
Why are women with PCOS at increased risk for endometrial cancer?
Chronic anovulation leads to unopposed estrogen exposure, causing endometrial hyperplasia.
What is the effect of low FSH levels in PCOS?
FSH levels are insufficient to aromatize androgens into estrogen, leading to follicle arrest.
What is the gold-standard treatment of PCOS for those not seeking pregnancy, to target specifically menstrual regulation and provide endometrial protection?
Combined OCPs.
Why are combined OCPs the gold-standard for PCOS?
They suppress LH (reducing androgen production) and increase SHBG (decreasing free testosterone); They prevent endometrial hyperplasia
What is the treatment for management of hyperandrogenism (hirsutism/acne)?
Combined use of COC (to lower free testosterone) and spironolactone (block androgen receptors at the follicle)
Why must a COC be used with spironolactone?
Spironolactone alone can cause teratogenicity (risk of feminizing male fetus)
What conditions must be ruled out before diagnosing PCOS?
Thyroid disease, hyperprolactinemia, and non-classic congenital adrenal hyperplasia.
What is the role of Metformin in PCOS management?
It acts as an insulin sensitizer to treat glucose intolerance and Type 2 Diabetes risk, often aiding in weight loss.
What is the primary goal of managing hyperandrogenism?
Treating the underlying cause while providing symptom control (e.g., OCPs, anti-androgens).
What long-term health screenings are recommended for patients with PCOS?
Annual blood pressure, BMI, lipid profile, periodic 2-hour OGTT or HbA1c for diabetes, and routine mental health screening for depression and anxiety.
What is the clinical significance of HAIR-AN syndrome?
It stands for Hyperandrogenism, Insulin Resistance, and Acanthosis Nigricans; it is a severe form of insulin resistance.
What is the classic triad of symptoms defining HAIR-AN syndrome?
Hyperandrogenism, Insulin Resistance, and Acanthosis Nigricans.
How does severe insulin resistance in HAIR-AN syndrome lead to hyperandrogenism?
Excess insulin binds to IGF-1 receptors on the ovaries, stimulating theca cells to produce excess androgens, while also suppressing SHBG.
What lab values are diagnostic indicators for HAIR-AN syndrome?
Insulin levels >80 μU/mL, 2-hour GTT >500 μU/mL, and Total Testosterone >200 ng/dL.
Which ovarian tumor is the most common androgen-secreting tumor in young women?
Sertoli-Leydig Cell Tumor.
What type of ovarian tumor is more common in postmenopausal women?
Hilus Cell Tumor.
What clinical signs suggest an ovarian neoplasm?
Rapid-onset hirsutism (weeks/months) and significant virilization (clitoromegaly, voice deepening).
What is the lab marker indicative of an ovarian neoplasm?
Total testosterone > 200ng/dL
What is the definition of Idiopathic Hirsutism?
Hirsutism in women with regular ovulatory menses and normal serum androgen levels.
What is the pathophysiology of Idiopathic Hirsutism?
Increased 5-alpha-reductase activity within the hair follicle, which converts testosterone to the more potent DHT.
What kind of diagnosis is idiopathic hirsutism?
Diagnosis of exclusion; must rule out PCOS, HAIR-AN, tumors.
What is the primary mechanism of Combined OCPs in treating hyperandrogenism?
Suppresses LH to decrease ovarian androgens and increases SHBG levels.
What is the primary mechanism of Spironolactone in treating hyperandrogenism?
It blocks androgen receptors and inhibits 5-alpha-reductase.
Which medication is used to suppress ACTH and decrease adrenal androgens?
Glucocorticoids.
When should a patient with hyperandrogenism be referred to an endocrinologist?
In cases of sudden onset/virilization, failure of primary care management, or when infertility requires ovulation induction.
How is clinical infertility defined in a general context?
The failure to conceive after one year of unprotected intercourse.
What is the recommended timeframe for seeking an infertility evaluation for women over 40?
No more than 3 months.
What is the recommended timeframe for seeking an infertility evaluation for women over 35?
6 months, or less if there is a known history of conditions like PCOS, PID, or oligomenorrhea.
What is the difference between primary and secondary infertility?
Primary infertility is the inability to ever conceive; secondary infertility is the inability to conceive after having been pregnant in the past.
What are the three main categories of infertility causes and their approximate prevalence?
Female factors (65%), Male factors (20%), and Unexplained (15%).
What is the first diagnostic test that should always be performed when investigating infertility?
A semen analysis to rule out male factors.
What is the most common correctable cause of male infertility?
Varicocele (often described as a 'bag of worms').
What lab profile is associated with testicular (primary) male infertility?
High FSH/LH and low testosterone.
Why should a second semen analysis be performed if the first is abnormal?
Due to the high variability in sperm production; a second test should be done 1-2 weeks later.
What does a high FSH level indicate in the context of a male infertility workup?
Primary testicular failure, which generally carries a poor prognosis.
What are the three main pillars of female infertility causes?
Ovulatory factors, Tubal/Pelvic factors, and Uterine factors.
What is the most common cause of female infertility?
Ovulatory dysfunction.
How can ovulation be confirmed in a female patient?
Basal body temperature tracking, ovulation prediction kits (LH surge), or mid-luteal serum progesterone levels.
What is the first-line imaging modality for female infertility?
Transvaginal ultrasound.
Which diagnostic test is used to evaluate uterine shape and tubal patency?
Hysterosalpingogram (HSG).
What is the gold standard for diagnosing tubal or peritoneal disease, such as endometriosis?
Laparoscopy.
Which medications are commonly used for ovulation induction?
Letrozole (Aromatase Inhibitor) or Clomiphene Citrate (SERM).
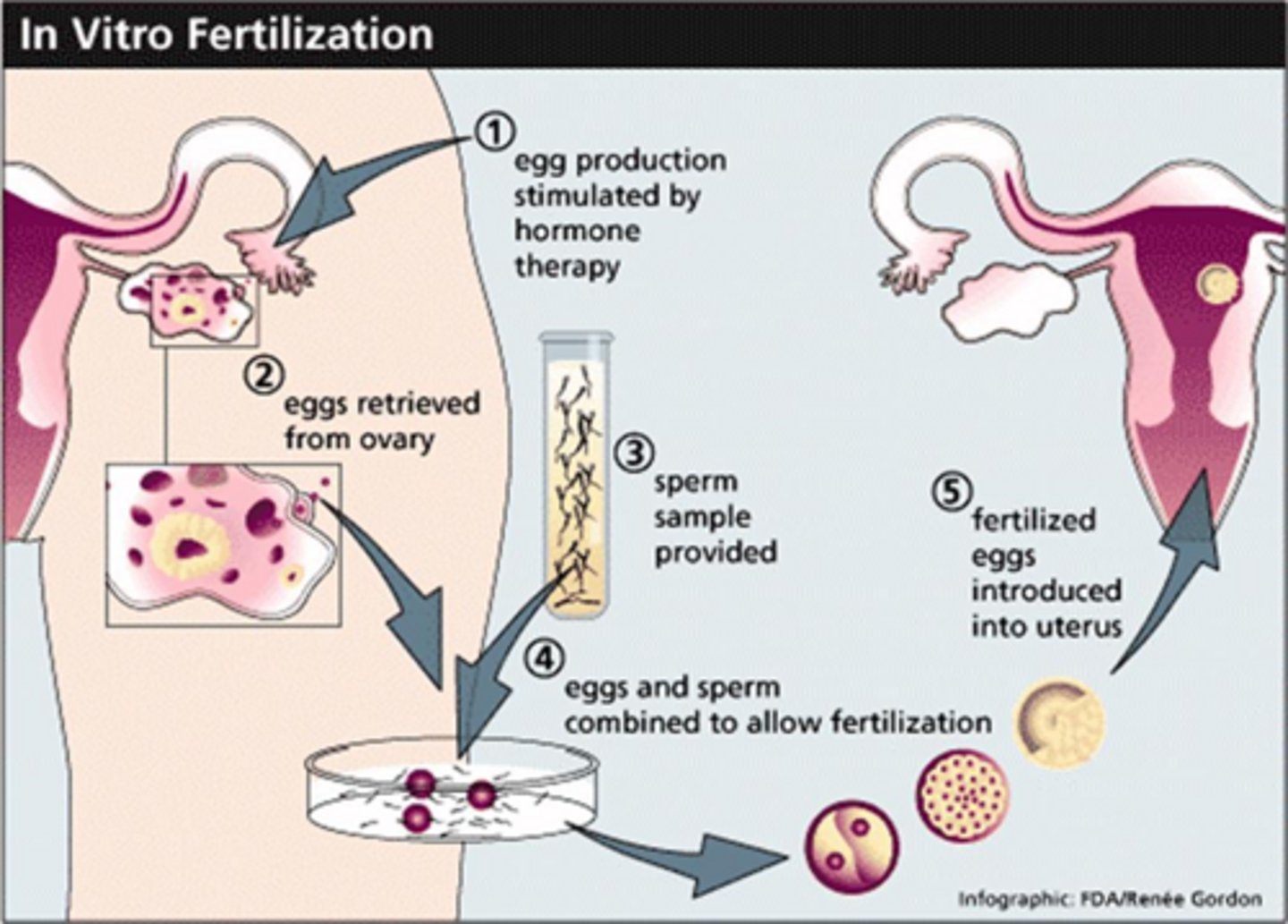
What is the gold standard treatment for severe male factor infertility?
IVF with Intracytoplasmic Sperm Injection (ICSI).
What is the treatment of choice for a woman with blocked fallopian tubes?
In vitro fertilization (IVF).
What is the treatment of choice for a woman with PCOS who is not ovulating?
Ovulation induction with clomiphene citrate or letrozole.
What is the role of daily progesterone in an IVF cycle?
To support the pregnancy, typically continued until 10 weeks gestation.
What are the Tier 1 contraceptive methods?
Long-acting reversible contraceptives (IUD and Nexplanon)
What are the primary mechanisms of Levonorgestrel IUDs (e.g., Mirena)?
Thickening cervical mucus and thinning the endometrial lining.
Which IUD is considered the most effective therapy for heavy menstrual bleeding?
Mirena.
What are the CI for IUD?
Pregnancy, current PID, unexplained vaginal bleeding, uterine cavity distortion, active infection, breast cancer.
What is the primary mechanism of the Copper IUD (Paragard)?
Releases copper ions that act as a spermicide by inhibiting sperm motility and inactivating acrosomal enzymes.
What is the most effective non-hormonal contraceptive option?
The Copper IUD (Paragard).
What are the absolute contraindications for Copper IUD use?
Pregnancy, Wilson's disease, or copper allergy.
Which IUD is safe for women with breast cancer history?
Copper IUD
What is the primary mechanism of the Subdermal Implant (Nexplanon)?
Releases etonorgestrel to suppress ovulation and thicken cervical mucus.
What is the only absolute contraindication for the Nexplanon implant?
Recent breast cancer.
What are the Tier 2 contraceptives?
Combined hormonal contraception (OCPs, patch, ring)
How do combined hormonal contraceptives (pills, patch, ring) prevent pregnancy?
They suppress FSH and LH to inhibit ovulation, thicken cervical mucus, and thin the endometrium.
What are the major cancer-related benefits of combined hormonal contraceptives?
Reduced risk of ovarian cancer (up to 50%) and significantly reduced risk of endometrial cancer.
What are the primary contraindications for combined hormonal contraceptives?
Smokers over age 35, history of VTE/stroke/CAD, migraine with aura, and breast cancer.
What is the mechanism for the mini pill and Depo-Provera?
They thicken cervical mucus and thin the endometrium (Depo also suppresses ovulation)
What are the risks associated with Depo-Provera use?
Bone mineral density loss, significant weight gain, and delayed return to fertility (up to 10 months).
Why is the 'mini pill' recommended for breastfeeding women?
It contains no estrogen, which helps avoid the suppression of prolactin levels.
What are the Tier 3 contraceptives?
Barrier and behavioral methods
Which contraceptive method is the only one that provides protection against STIs?
Condoms.
What are the requirements for Lactational Amenorrhea (LAM) to be an effective contraceptive?
Exclusively breastfeeding, within 6 months postpartum, and remaining amenorrheic.
How long can sperm live in the female reproductive tract?
5 to 7 days.
What is the most effective form of emergency contraception?
Insertion of an IUD (copper or Mirena) within 5 days of unprotected intercourse.
Which emergency contraceptive is most effective for patients with a BMI > 30?
Ulipristal Acetate (Ella). Requires an rx
What must be done if menses does not occur within 3 weeks of taking emergency contraception?
A pregnancy test is mandatory.
What must be confirmed before a patient can rely on a vasectomy for contraception?
A semen analysis (SA) confirming azoospermia at 3 months post-procedure.
What is a significant long-term health benefit of a salpingectomy?
It significantly reduces the risk of future ovarian cancer.
What is the required waiting period for sterilization consent in Florida?
The consent must be signed at least 30 days before the procedure.
Name three risk indicators for regret following permanent sterilization.
Age younger than 25, low parity, and sterilization performed at the time of a C-section.