Spectroscopic Analysis of a Reaction
1/15
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
16 Terms
What is this reaction enabled by?
This reaction is enabled by a carbocation rearrangement to form a more stable carbocation from a (relatively) less stable carbocation, which then readily forms the product.
How are carbocations stabilized?
Inductive effect and hyperconjugation
Carbocations are stabilized by neighboring alkyl groups inductively and via hyperconjugation. The more alkyl groups there are adjacent to a carbocation, the more stable the carbocation is. Thus, stability goes oxonium, tertiary, secondary, and methyl. Methyl carbocations are so unstable that they don’t exist.
Adjacent EDG
Electron-donating groups donate electron density into the empty p orbital of the carbocation and delocalize the positive charge between the carbocation and the neighboring atom. Delocalization of charge is always preferred over having a localized charge.
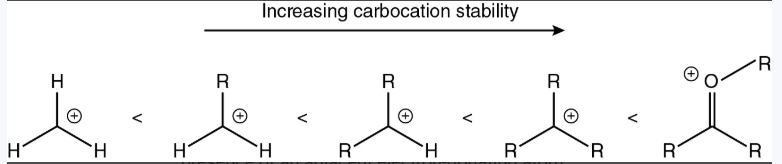
What will happen to form the more stable carbocation?
There will be a rearrangement to form the more stable carbocation.
Rank migratory aptitude
Hydride (H-) > Phenyl (aryl) > 3 alkyl > 2 alkyl > 1 alkyl > methyl
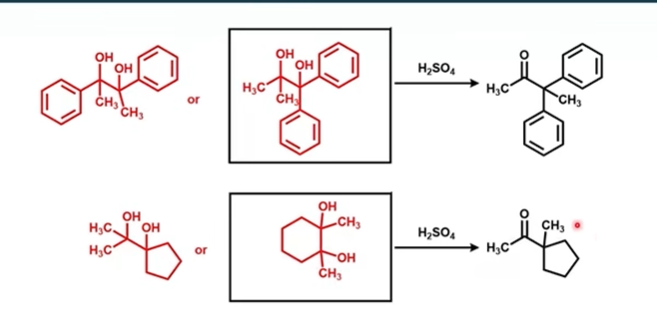
What rearrangement was performed in this reaction?
Pinacol rearrangement, named for 2,3-dimethyl-2,3-butanediol (pinacol), used as a name for the class of 1,2-rearrangements of carbocations formed from vicinal diols to produce carbonyl compounds when treated with aqueous acid.
Pinacol is a _____ vicinal diol
symmetric
Symmetric diol
Both sides are equivalent
Asymmetric diol
The group that migrates is the one that forms the more stable carbocation OR has a higher migratory aptitude
Migratory aptitude
Migratory aptitude is the relative ability of an R-group to move in a rearrangement reaction.
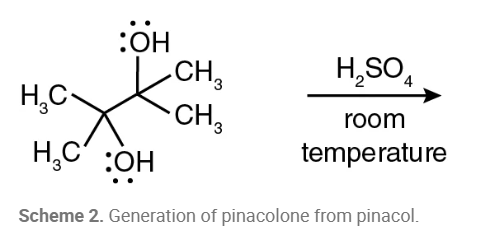
What is the product of this reaction?
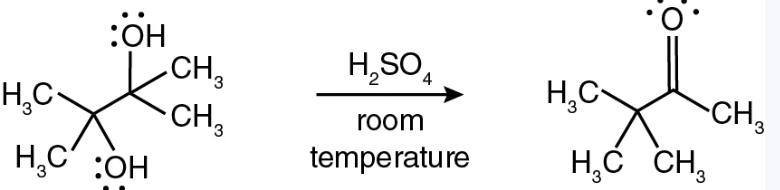
Pinacol rearrangement is an ____ mediated rearrangement? Why?
Pinacol rearrangement is an acid-mediated rearrangement
The reaction is acid-mediated because the acid initiates the entire mechanism by protonating the alcohol, allowing carbocation formation and rearrangement.
We used 3M H2SO4
Why is the pinacolone product purified using distillation?
Pinacolone has a lower boiling point than pinacol, so it evaporates first, and this vapor can condense and turn into a liquid, which is then collected in the condenser
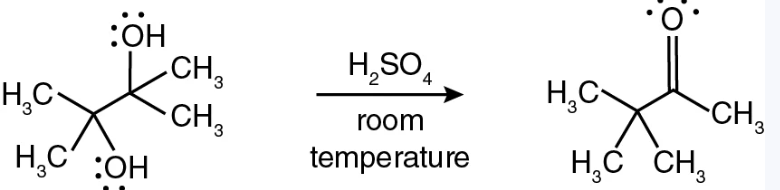
Draw the arrow pushing mechanisms for this reaction
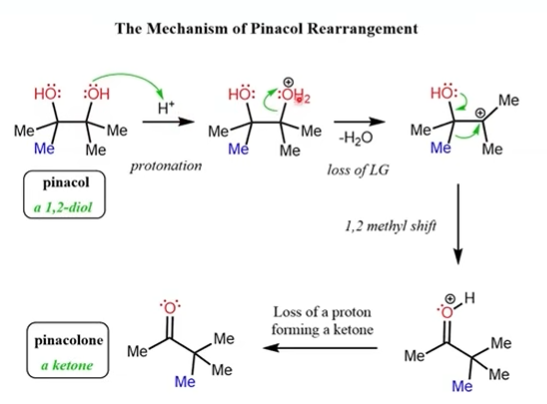
What drying agent was used in this reaction?
Anhydrous magnesium sulphate
What will the IR of this reaction show?
The IR spectrum of this reaction will show an alcohol group converting to a ketone
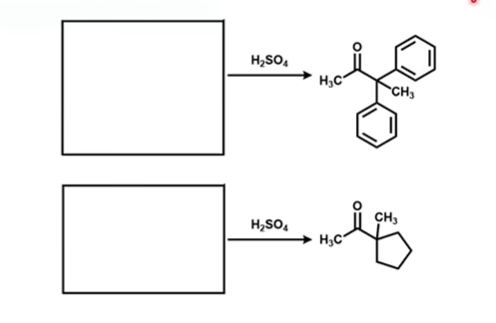
Draw the expected reagents of these reactions: