all organics (most of mechanics)
1/101
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
102 Terms
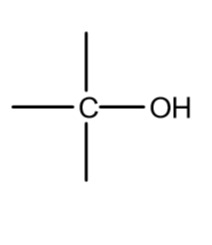
functional group
alcohols
alcohols suffix
-ol
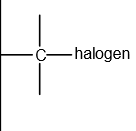
functional group
halogenoalkane
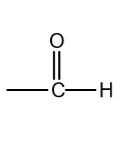
functional group
aldehydes
aldehydes suffix
-al
alcohols prefix
hydroxy
aldehydes prefix
formyl
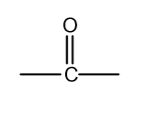
functional group
ketones
ketone suffix
-one
ketone prefix
oxo
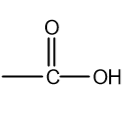
functional group
carboxylic acids
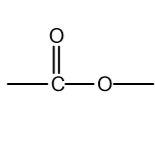
functional group
ester
ester prefix / suffix
-yl -oate
priority order of functional groups:
carboxylic acids, aldehydes, ketones, alcohols, alkenes, halogenoalkanes
define: structural isomerism
molecules with the same molecular formula but a different structural formula
define: chain isomerism
different arrangements of a molecule’s carbon skeleton
define: position isomerism
the differing position of the same functional group in the molecule
define: functional group isomerism
differing positions of atoms give a different functional group
define stereoisomerism
molecules with the same molecular and structural formula but a different spatial arrangement of atoms
alkanes typically used as
fuels and lubricants
are alkanes polar
no
intermolecular forces between alkanes
van der waals
as alkane size increases, IMF strength -
increases
whydo branched chain alkanes have lower melting points than straight chain alkanes with the same number of carbon atoms
they cannot pack as closely together so the van der Waals forces are weaker
alkanes solubility
insoluble
why are alkanes insoluble
water molecules are helf together by hydrogen bonds which are much stronger than the Van der Waals forces between alkanes
what technique is used to convert crude oil into useful products
factrional distillating
the fractional distillating column is cooler
at the top than at the bottom
fractions of fractional distillating column from top to bottom
refinery gas, petrol, naptha, kerosene, diesel, lubricating oil, fuel oil, bitumen
as carbon chain gets longer, the hydrocarbons (4 thing)
become more viscous, harder to ignite, less volatile, have higher boiling points
two types of cracking
thermal and catalytic
what is cracking
the thermal decomposition of alkanes
why do hydrocarbons undergo cracking
to make use of excess larger hydrocarbons and supply demand for shorter ones
temperature conditions for thermal cracking
900 degrees celcius
pressure conditions for thermal cracking
70atm
catalyst for thermal cracking
none
products of thermal cracking
alkenes
temperature conditions for catalytic cracking
450 degrees celcius
catalyst for catalytic cracking
zeolites
products of catalytic cracking
motor fuels (aromatics, cyclic alkanes, branched alkanes)
problem caused by CO2
greenhouse gas
problem caused by CO
toxic
problem caused by C
blackens buildings, can cause respiratory problems
problem caused by SO2
acid rain
problem caused by NOx (NO, NO2)
acid rain
what is the process of removing sulfur from the gases produced by power gas
flue gas desulfurisation
method for flue gas desulfurisation
a slurry of CaO (lime) and H2O is sprayed into the flue gas. The flue gas reacts with the CaO and H2O to form calcium sulfite. The calcium sulfite is further oxidised to form calcium sulfate (gypsum, a saleable product).
write the equation for flue gas desulfurisation with CaO (lime):
CaO(s) + SO2(g) → CaSO3(s)
what can be used instead of CaO (lime) in flue gas desulfurisation?
calcium carbonate (limestone)
write the equation for flue gas desulfurisation with CaCO3 (limestone)
CaCO3(s) + ½ O2(g) + SO2(g) → CaSO4(s) + CO2(g)
what do catalytic converters do
remove CO, NOx, and unburned hydrocarbons from exhaust gases, turning them into CO2, N2 and H2O
what are catalytic converters (i.e. made up of)
a ceramic honeycomb coated with a thin layer of catalyst metals (i.e. platinum) to give a large surface area
write the equation for CO and NO in a catalytic converter
2 CO + 2 NO → 2 CO2 + N2
write the equation for unburned hydrocarbons in a catalytic converter using octane as an example
C8H18 + 25 NO → 8 CO2 + 12.5 N2 + 9 H2O
what do molecules that contain polar bonds do which contributes to the greenhouse effect?
absorb IR radiation to make bonds vibrate
what is the condition for free radical substitution
UV light
3 different reagents for nucelophilic substitution with halogenoalkanes
NaOH, KCN, NH3
what is a nucleophile
an electron pair donor
what are enantiomers
molecules which are non-superimposable mirror images of one another
conditions for nucelophilic substitution NaOH with halogenoalkane
aqueous, warm
conditions for nucelophilic substitution KCN with halogenoalkane
ethanolic, warm
conditions for nucelophilic substitution NH3 with halogenoalkane
excess concentrated ammonia dissolved in ethanol at pressure in a sealed container
reagent for elimination reaction for halogenoalkanes
KOH
conditions for elimination reaction of halogenoalkanes with KOH
hot, ethanolic
what is the beneficial role of ozone in the stratosphere
absorbs UV radiation
OVERALL equation for the decomposition of ozone
2 O3 → 3 O2
are halogenoalkanes soluble
no
why are halogenoalkanes not soluble
the polar C-X bonds are not polar enough to make the halogenoalkanes soluble in water. IMF are permanent dipole-dipole for halogenoalkanes.
boiling point of halogenoalkanes increase with
increased chain length and going down the halogen group
why do increased chain length and larger mr of halogens increase the boiling points of halogenoalkanes
Van der Waal forces
what does the OH- ion act as in the elimination reaction between KOH and halogenoalkanes
a base
why cant the double bond rotate in an alkene
as well as a single carbon bond, there is a p-orbital containing a single electron on each carbon. these two orbitals overlap to form an orbital with a cloud of electron density above and below the single bond. this is called a pi orbital and means the bond cannot rotate.
what does CIP stand for
cahn-ingold-prelog
why are alkenes typically more reactive than alkanes
the electron dense area around the double bond can easily be attacked by electrophiles
what are electrophiles
electron pair acceptors
why are tertiary carbocations more stable than secondary carbocations and secondary carbocations are more stable than primary carbocations?
alkyl groups have a tendency to release electrons. this is known as a positive inductive effect. the positive inductive effect stabilises thepositive charge of the intermediate carbocation.
conditions for electrophilic addition of alkenes with Br2
aqueous
conditions for electrophilic addition of alkenes with HBr
none
conditions for electrophilic addition of alkenes with H2SO4
concentrated H2SO4, cold (typically room temperature)
conditions for electrophilic addition of alkenes with H2O
with strong acid (H2SO4 or H3PO4)
test for double bond
add bromine water to sample, bromine water is decolourised
define polymers
very large molecules built up from small molecules
what is reflux?
heat to boiling point of mixture for a prolonged period of time. Vapour is formed which escapes from the liquid mixture, is changed back into liquid, and returned to the liquid mixture
colour change for acidified potassium dichromate wth alcohols
orange colour turns green (primary and secondary alcohols)
test for carboxylic acids
add NaHCO3, CO2 given off
how is ethanol produced industrially
reacting ethene with steam using a catlyst of phosphoric acid
method for production of ethanol from crude oil
cracking and hydration
method for production of ethanol from carbohydrates
fermentation and distillation
rate of reaction for production of ethanol from ethene
fast
rate of reaction for production of ethanol from carbohydrates
slow
temperature for production of ethanol from ethene
300
temperature for production of ethanol from carbohydrates
35
pressure for production of ethanol from ethene
60-70 atm
pressure for production of ethanol from carbohydrates
1 atm
catalyst for production of ethanol from ethene
phosphoric acid
catlyst for production of ethanol from carbohydrates
enzymes in yeast
conditions for production of ethanol from carbohydrates
anaerobic, aqueous
type of process for production of ethanol from carbohydrates
batch
type if production for production of ethanol from ethene
continuous
purity of ethanol for production of ethanol from carbohydrates
aqueous solution produced requiring fractional distillation