3.3.15 nmr
1/27
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
28 Terms
what is nuclear magnetic resonance (nmr)?
a very powerful tool for identifying unknown compounds
what are the units of chemical shift (δ)?
ppm
what does nmr give information about?
about the position of ¹³C or ¹H atoms in a molecule
give the 3 steps of how nmr is carried out
sample must be dissolved in a solvent
a standard must be added + the peak is set to 0ppm chemical shift on the x-axis — all the other peaks’ shifts are measured relative to the TMS peak
then carry out either a carbon (¹³C) NMR or proton (¹H) NMR
what must the solvent not have any of for ¹H NMR? outline why
it must not have any ¹H atoms present
as they would produce a big signal + interfere with the spectrum of the compound you were analysing
what solvent is used if:
a. sample is polar?
b. sample is non-polar?
a. use CDCl₃ not CHCl₃
b. use CCl₄
explain why we use D (deuterium) instead of H in the solvent?
the nucleus of H: 1 proton — odd number of nucleons so is magnetic - is NMR active - produces a signal - interferes with the spectrum of the compound you are analysing
the nucleus of D: 1 proton + 1 neutron — even number of nucleons so is not magnetic - is not NMR active - does not produce a signal - does not interfere with the spectrum of the compound you are analysing
what is the standard that is added? + give its structure
tetramethylsilane (TMS)
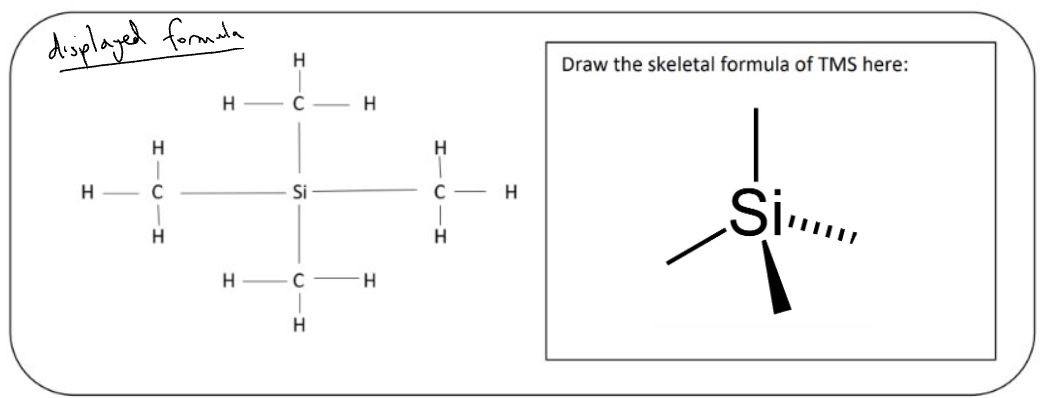
outline 3 reasons why tetramethylsilane is added to all sample analysed

carbon-13 NMR is…...than H¹ NMR
less complex
what determines the shift with C-13 NMR?
the environment that each C atom is in will determine the shift
eg if there are 2 different carbon environments (like in ethanol) how many peaks will there be in the C-13 NMR?
2 peaks
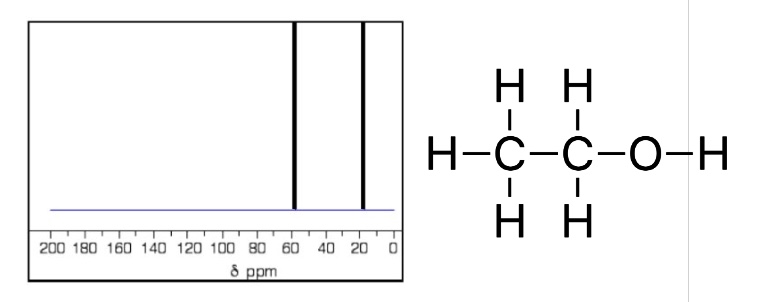
what is the number of peaks in a C-13 NMR always equal to?
to the number of different carbon environments in the compound
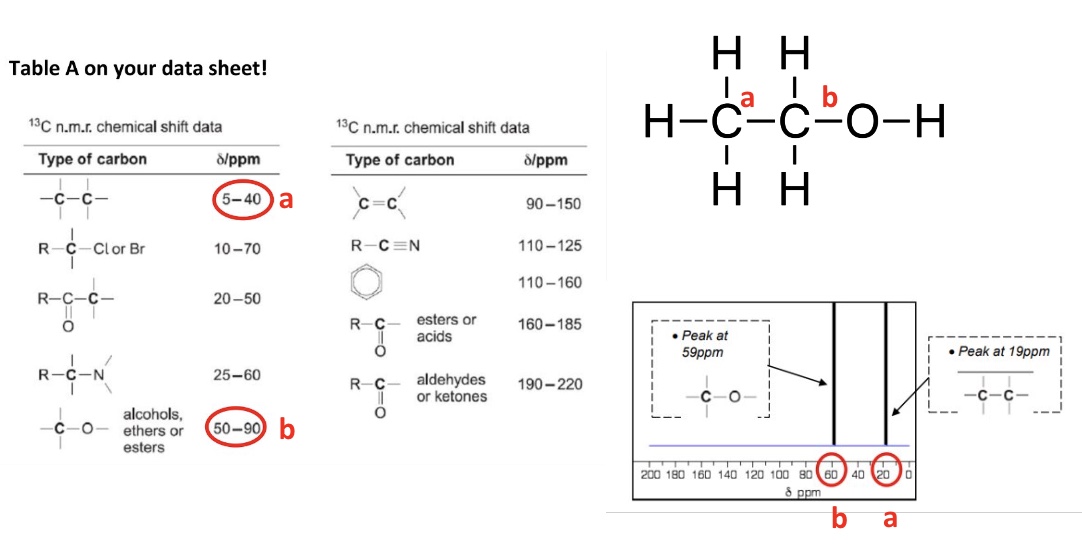
what determines the shift of each peak in C-13 NMR?
the type of carbon
example in image
however when will the number of peaks not equal the number of carbons in a compound?
if there is any symmetry in the molecule
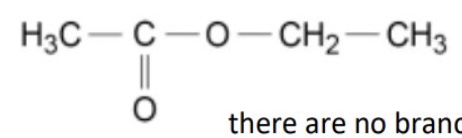
eg how many environments + so peaks for this molecule?
there are 4 environments so 4 peaks
there are no branches, there is no symmetry so every carbon has a unique environment
example of symmetry in molecule
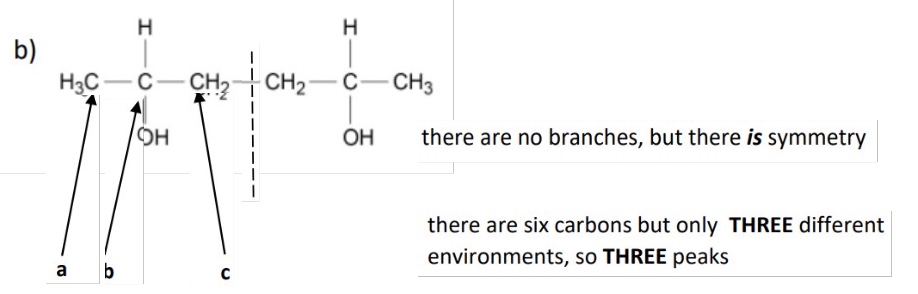
outline what will be produced if there are ever a number of of methyls on the same carbon
they will be in the same environment due to symmetry on the tetrahedral C
and so will give rise to 1 peak
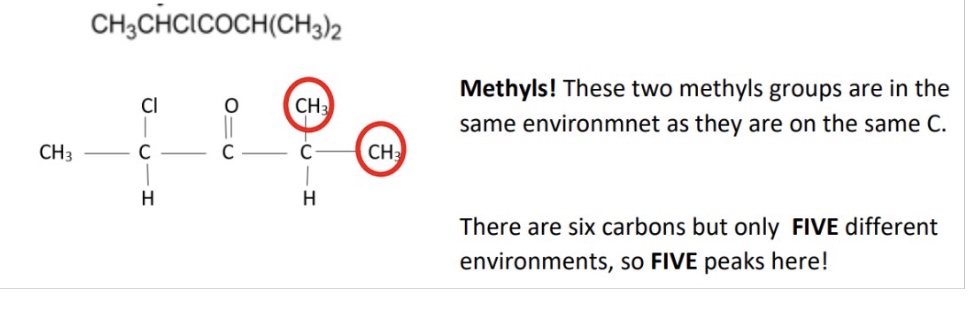

A - 3
B - 2
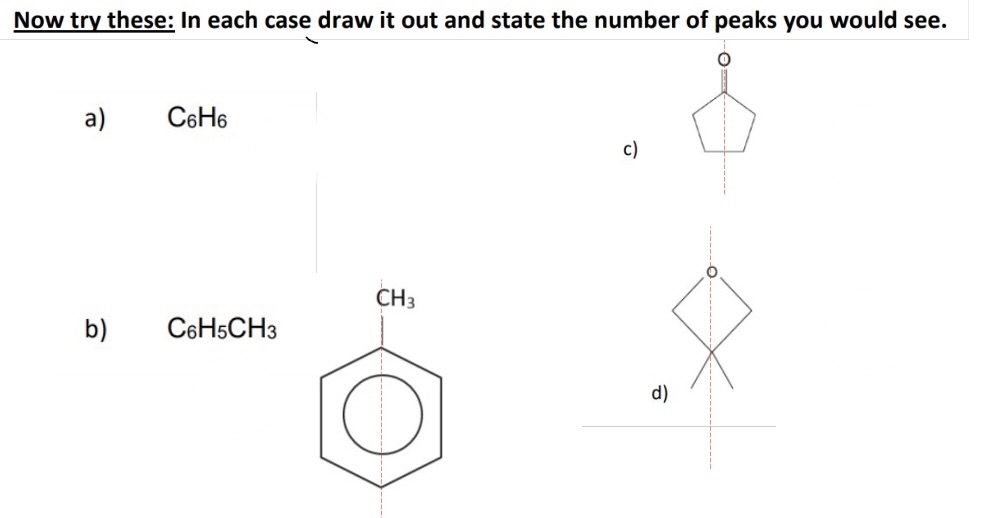
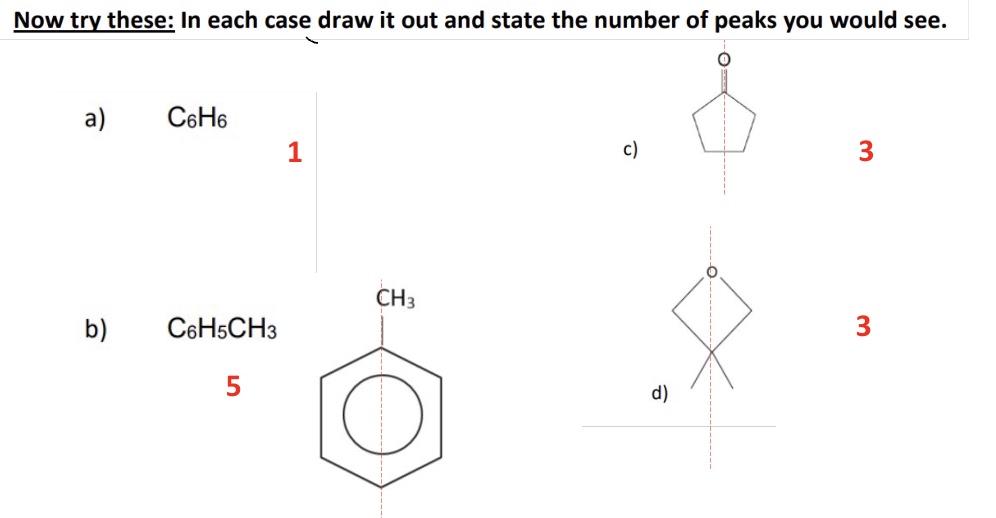
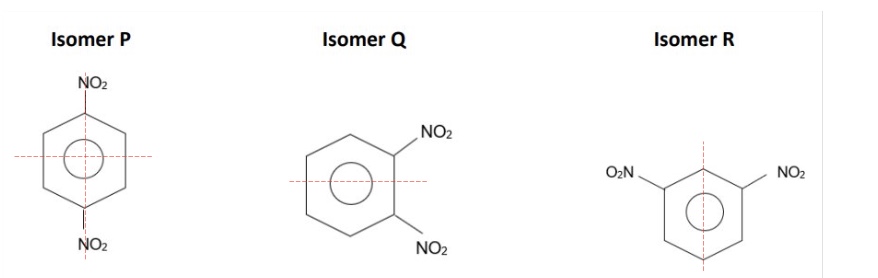
give the number of C-13 NMR peaks for each isomer
P - 2
Q - 3
R - 4
in some exam questions, what will you have to deduce from a molecular formula + given number of C-13 NMR peaks
the structure of a compound


in other types of questions, what would you be required to suggest?
a chemical shift value for a given type of carbon atom
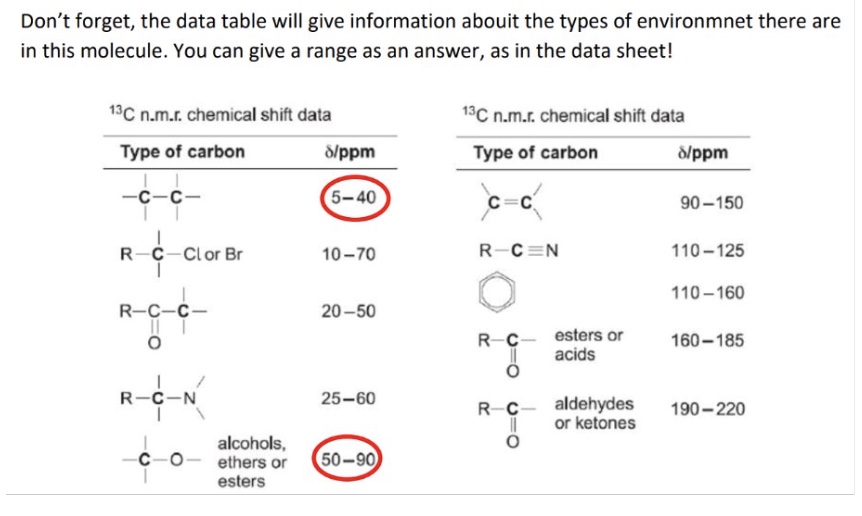
eg - giving chemical shift values
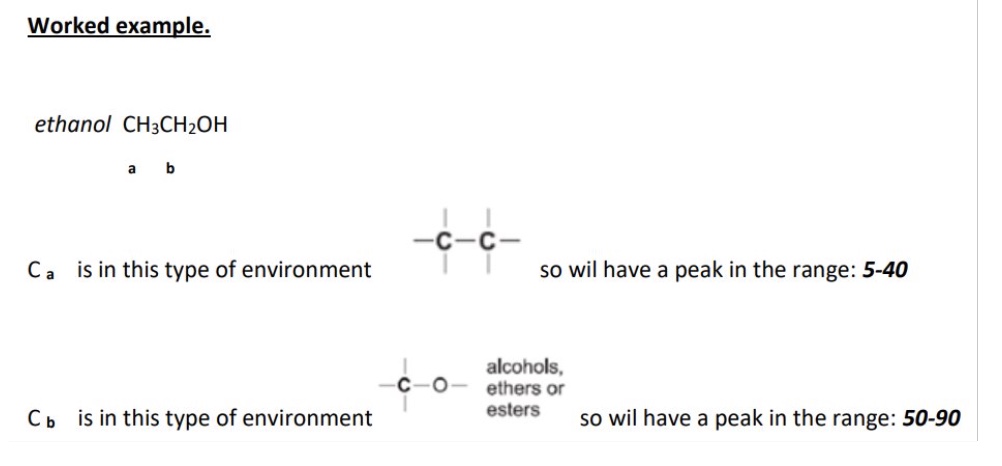
give the type of environment the labelled C is in + the range it will have a peak in
benzene environment
peak in the range 110-160ppm

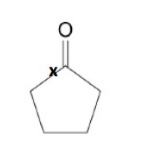
give the type of environment the labelled C is in + the range it will have a peak in
ketone environment
peak in the range 190-220ppm

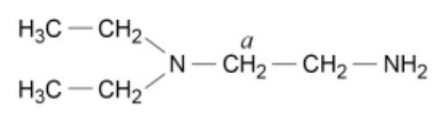
give the type of environment the labelled C is in + the range it will have a peak in
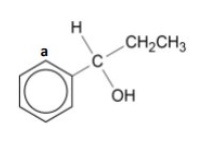
peak in the range 25-60ppm
