AQA A level Chem 2.3 Group 7 the Halogens
1/24
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
25 Terms
Chlorine is used to treat water even though it is toxic to humans. Give one reason why water is treated with chlorine. Explain why chlorine is added to water even though it is toxic. Give an equation for the reaction of chlorine with cold water. (3)
Chlorine sterilise water/kill bacteria/microorganisms (1)
Explanation : chlorine is only used in small quantities (1)
Equation : Cl2 + H2O ⇌ HCl + HClO (1)
Chlorine reacts with cold, aqueous sodium hydroxide in the manufacture of bleach. Give an equation for this reaction. (1)
Cl2 + 2 NaOH → NaCl + NaClO + H2O (1)
Which pair of solutions, when mixed, reacts to form a dark brown solution?
A NaF(aq) + Cl2(aq)
B NaCl(aq) + Br2(aq)
C NaBr(aq) + Cl2(aq)
D NaI(aq) + Br2(aq) (1)
D (1)
Some solid sodium halides are reacted with concentrated sulfuric acid.
Which solid sodium halide does not produce a sulfur-containing gas as one of
the products?
A NaCl
B NaBr
C NaI
D NaAt (1)
A (1)
Give an equation for the reaction of solid sodium bromide with concentrated sulfuric acid to form bromine. State one observation made during this reaction. (2)
2H2SO4 + 2NaBr → Na2SO4 + SO2 + Br2 + 2H2O (1)
orange/brown fumes solution (1)
A student completes an experiment to determine the percentage by mass of
sodium chloride in a mixture of sodium chloride and sodium iodide.
The student uses this method.
• 600 mg of the mixture are dissolved in water to form a solution.
• An excess of aqueous silver nitrate is added to the solution. This forms a
precipitate containing silver chloride and silver iodide.
• Excess dilute ammonia solution is then added to the precipitate. The silver
chloride dissolves.
• The silver iodide is filtered off from the solution, and is then washed and
dried.
The mass of the silver iodide obtained is 315 mg
Silver nitrate is added to the solution. Suggest why an excess is used. (1)
To ensure that all the halide ions (chloride and iodide) are removed from the solution / to ensure that all the halide ions precipitate out of solution (1)
This question is about sodium halides.
State what is observed when silver nitrate solution is added to sodium
fluoride solution. (1)
colourless (1)
State one observation when solid sodium bromide reacts with concentrated sulfuric acid. Give an equation for the reaction. State the role of the bromide ions in the reaction (3)
2 NaBr + 2 H2SO4 → Na2SO4 + SO2 + Br2 + 2 H2O (1)
brown/orange gas (1)
oxidising agent (1)
What is the best oxidising agent?
A F2
B F–
C I2
D I– (1)
A (1)
In the halogens group, oxidising strength decreases down the group:
F₂ > Cl₂ > Br₂ > I₂
The best oxidising agent is the substance that most readily gains electrons (is reduced).
Which statement is correct about reactions involving halide ions?
A Sodium chloride forms chlorine when added to
concentrated sulfuric acid.
B Sodium chloride forms chlorine when added to
bromine.
C Sodium bromide forms bromine when added to
concentrated sulfuric acid.
D Sodium bromide forms bromine when added to iodine (1)
C (1)
Solution Y contains two different negative ions.
To a sample of solution Y in a test tube a student adds
• silver nitrate solution
• then an excess of dilute nitric acid
• finally an excess of concentrated ammonia solution.
The observations after each addition are recorded in the table.
Reagent added to solution Y | Observation |
|---|---|
silver nitrate solution | cream precipitate containing compound D and compound E |
excess dilute nitric acid | cream precipitate D and bubbles of gas F |
excess concentrated ammonia solution | colourless solution containing complex ion G |
Give the formulas of D, E and F.
Give an ionic equation to show the formation of E.
Give an equation to show the conversion of D into G. (6)
D - AgBr (1)
E-Ag2CO3 (1)
F- CO2 (1)
2Ag+ + CO32- → Ag2CO3 (1) m
AgBr + 2 NH3 → Ag(NH3)2 + + Br–(1)
Separate unlabelled solid samples of three anhydrous sodium compounds are provided for a student to identify.
These compounds are known to be sodium carbonate, sodium fluoride and sodium chloride but it is not known which sample is which.
Outline a logical sequence of test-tube reactions that the student could carry out to identify each of these compounds.
Include the observations the student would expect to make.
Give equations, including state symbols, for any reactions that would take place. (6)
Add HCI to all three samples
Add water/make into a solution
Add AgNO3
NaCl gives white ppt with AgNO3
Na2CO3 will fizz with acid
NaF shows no (visible) change / no ppt
Na2CO3 + 2HNO3 → 2NaNO3 + CO2 + H2O
AgNO3 + NaCl → AgCl + NaNO3(6)
A student was given a 50.0 g sample of solid silver chloride contaminated with
solid silver carbonate.
The student suggested the following method to obtain the maximum amount of
pure dry silver chloride from the sample:
1. Tip the solid into a boiling tube.
2. Add dilute nitric acid.
3. Allow the remaining solid to settle.
4. Decant off the liquid.
5. Leave the sample to dry on a shelf.
Identify any faults or omissions in the method suggested by the student.
Suggest improvements to the method, using commonly available laboratory
equipment.
The following chemicals are also available:
distilled water, dilute solutions of NaOH, NH3, HCl, H2SO4 (6)
Stage 1 Apparatus
• Boiling tube likely to be too small for 50.0 g solid plus acid
• Use a (large / medium / 250 ml) beaker instead
Stage 2 AgCl Prep
• HNO3 will form soluble AgNO3 so max AgCl
not obtained
• Use HCl instead
• Method doesn’t say how much acid should be
used
Stage 3 Making pure dry solid
• Decanting means solid wet / contaminated
with HNO3 / acid or decanting means some
solid lost
• Instead use filter and then wash with distilled
water (6)
Bromine (Br2), strontium chloride (SrCl2) and iodine monochloride (ICl) all have similar Mr values.
Suggest, with reasons, the order of melting points for these three
substances. (6)
SrCl2> ICl > Br> (1)
SrCl2 strong ionic bonds / (strong electrostatic attraction between opposite ions) (1)
Lattice so many strong bonds to overcome (1)
ICl has dipole-dipole forces between molecules – weaker than ionic bonds (1)
Br2 has van der Waals forces between molecules – much weaker (1)
Suggest a disadvantage of treating water in chlorine (1)
Some people suffer eye irritation / Some people find the taste unpleasant (1)
Bromine reacts with phosphorus to form phosphorus tribromide. Write an equation for this reaction (1)
6Br2 + P4 ⟶ 4PBr3 (1)
Sodium bromide reacts with concentrated sulfuric acid in a different way from sodium chloride.
Write an equation for this reaction of sodium bromide and explain why bromide ions react differently from chloride ions. (3)
2NaBr + 2H₂SO₄ → Na₂SO4 + Br₂ + SO₂ + 2H₂O (1)
- bromide ions are bigger than chloride ions (1)
therefore Br⁻ ions are more easily oxidised/lose an electron more readily than Cl⁻ (1)
A colourless solution contains a mixture of sodium chloride and sodium bromide. Using aqueous silver nitrate and any other reagents of your choice, develop a procedure to prepare a pure sample of silver bromide from this mixture. Explain each step in the procedure and illustrate your explanations with equations, where appropriate. (6)
Stage 1: formation of precipitates
• Add silver nitrate
• to form precipitates of AgCl and AgBr
• AgNO3 + NaCl → AgCl + NaNO3
• AgNO3 + NaBr → AgBr + NaNO3
Stage 2: selective dissolving of AgCl
• Add excess of dilute ammonia to the mixture of
precipitates
• the silver chloride precipitate dissolves
• AgCl + 2NH3 → Ag(NH3)2++ Cl−
Stage 3: separation and purification of AgBr
• Filter off the remaining silver bromide precipitate
• Wash to remove soluble compounds
• Dry to remove water (6)
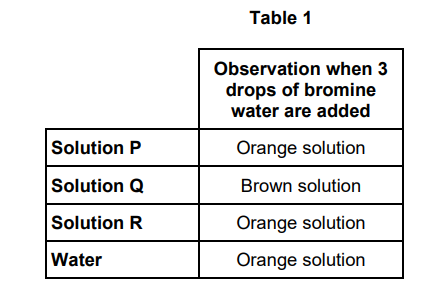
A student does a series of reactions with aqueous solutions of some potassium halides (P, Q and R) of equal concentration. Each solution contains a different halide ion (chloride, bromide or iodide).
The student adds 3 drops of bromine water to 3 drops of each aqueous solution of potassium halide. The student also adds 3 drops of the bromine water to 3 drops of water.
Table 1 shows the student’s observations
Identify the halide ion present in Q.
Give the ionic equation for the reaction that occurs when bromine water is
added to Q. (2)
Iodide ion / I- (1)
Br2 + 2 I- → I2 + 2 Br (1)
Explain why the electronegativity of the halogens decreases down the group (2)
Larger atoms /more electron shells (1)
weaker attraction between nucleus and bonding pair of electrons (1)
Concentrated sulfuric acid reacts with solid sodium chloride and with solid
sodium bromide.
State one similarity in, and one difference between, these reactions.(2)
similarity : form hydrogen halides (1)
difference : bromide undergoes a redox reaction (1)
In acidic solution, IO3– ions oxidise iodide ions to iodine. IO3– + 5 I– + 6 H+ → 3 I2 + 3 H2O Give a half-equation for the oxidation of iodide ions to iodine. Deduce the half-equation to show the reduction process in this reaction. (2)
Oxidation half equation : 2I- → I2 + 2 e– (1)
Reduction half equation : 2 IO3 – + 12 H+ + 10 e– → I2 + 6 H2O (1)
When iodide ions are oxidised using concentrated sulfuric acid, sulfur
dioxide, a yellow solid and a foul-smelling gas are all formed.
Give an equation to show the reaction between iodide ions and
concentrated sulfuric acid to form the yellow solid.
Identify the foul-smelling gas. (2)
6 I– + 6 H+ + H2SO4 → S + 3 I2 + 4 H2O (1)
Foul smelling gas – H2S / hydrogen sulphide (1)
The student does an additional experiment to show that solution D contains a mixture of halide ions. One of the halide ions is chloride.
Method:
Step 1 Add an excess of AgNO3(aq) to 10.0 cm3 of solution D.
Step 2 Filter, wash, dry and weigh the precipitate.
Step 3 Add an excess of dilute ammonia to the dry precipitate.
Step 4 Filter, wash, dry and weigh the solid that remains.
Explain how the masses recorded during this experiment can be used to
show that solution D contains a mixture of halide ions. (2)
The second mass is smaller / the mass after step 4 is smaller than the mass after step 2 (1)
AgCl dissolves in dilute ammonia / some ppt dissolves as AgCl is soluble in dilute ammonia (1)
Solid sodium iodide reacts with concentrated sulfuric acid to form hydrogen sulfide.
Give a half-equation to show the oxidation of iodide ions. Give a half-equation to show the reduction of concentrated sulfuric acid to hydrogen sulfide. Use your half-equations to deduce an overall equation for this reaction. (3)
2 I- → I2 + 2 e-(1)
H2SO4 + 8 H+ + 8 e- → H2S + 4 H2O (1)
H2SO4 + 8 H+ + 8 I- → 4 I2 + H2S + 4 H2O (1)