PPFB 14 - Modified release oral drug Delivery
1/28
Earn XP
Description and Tags
● Introduction to Modified-Release Oral Drug Delivery ● Sites of Action & Biopharmaceutical Considerations ● Designing a Modified-Release Formulation ● Types of sustained Release Systems
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
29 Terms
What is the main purpose and application of modified-release drugs ?
deliver drugs at specific rates, times or body sites to improve therapy
maintain steady drug levels, reduce dosing frequencies and side effects
Applications - Targeting specific areas of the gastrointestinal tract (e.g., colon). Once-daily dosing for better patient compliance.
What are the different types of modified release ?
Extended / Sustained / Controlled Release
Delayed Release (e.g., enteric coating).
Gastro-resistant Release
What are the patients benefits?
What impact does sr have on healthcare professionals ?
more choice for treatment - immediate vs released
product life extension
healthcare cost savings
Downside:
Higher Development Costs (More expensive)
What are the sites of action and the biopharmaceutical impact
● GI Tract: Release → Dissolution → Absorption
● pH: Stomach acidic, Small intestine ~7, Colon variable variability with age, disease, ethnicity, etc.
● Transit Time: Stomach 1–2 h, SI 3–4 h, Colon 1–72 h Timing with food (empty stomach vs with food)
● Fluid: Less liquid further down; fats slow release) May not dissolve well in the colon (< 10 ml fluid)
What factors do you have to consider when designing modified release formulations?
Factor 1 - single vs multiple unit dosage forms
Factor 2 - Matrix vs Coated Formulation
Why do we consider factor 1 ?
Single-unit dosage forms (e.g., tablets)
Easy to manufacture
Risk: can get trapped in the stomach (especially with
food).
Multiple-unit dosage forms (e.g., pellets in capsules)
More consistent gastric emptying.
Lower risk of dose dumping.
Harder and more costly to manufacture.
Why do we consider Factor 2 ?
Matrix formulation:
Drug mixed with release-modifying ingredients throughout the tablet/capsule. The entire dosage form controls drug release. (can split)
Coated formulation:
Drug core surrounded by a release-controlling coat; drug released through/dissolves via the coating. (should not split)
What is the drug release control mechanism?
Two main processes :
dissolution of the drug
diffusion of dissolved drug
What are the 4 steps in modified release systems?
1. Hydration/swelling or dissolution of part of the device.
2. Diffusion of water into the device.
3. Dissolution of the drug inside.
4. Diffusion of dissolved drug out of the device.
What is a matrix former?
A matrix former is an excipient (or mixture of excipients) used to control the release rate of a drug from a tablet. It helps either:
Speed up drug release (faster dissolution), or
Slow down drug release (sustained or controlled release)
How does a matrix former work?
How It Works:
The drug is embedded in the matrix material.
The matrix controls how quickly the drug is released when it comes into contact with fluids in the body.
What are the types of matrix formers?
Water-soluble (e.g., polyethylene glycol, PEG) → Used when faster release is needed.
Insoluble or gel-forming (e.g., lipids or polymers like HPMC → Used to prolong the release of the drug over time.
Key Point:
Matrix formers are often polymers or lipids, and they can
make up a large portion of the tablet.
What are the different types of release profiles
Four possible release patterns:
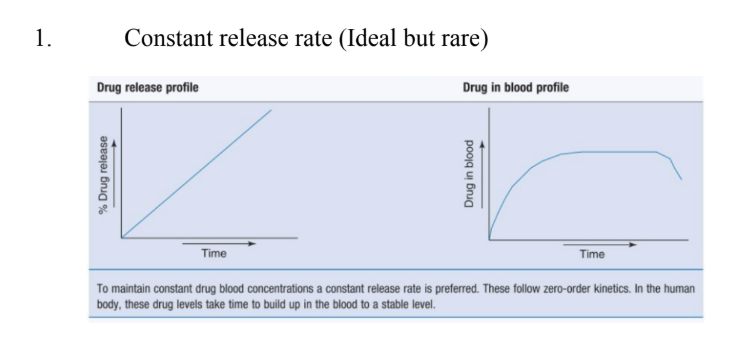
1. Constant release rate.
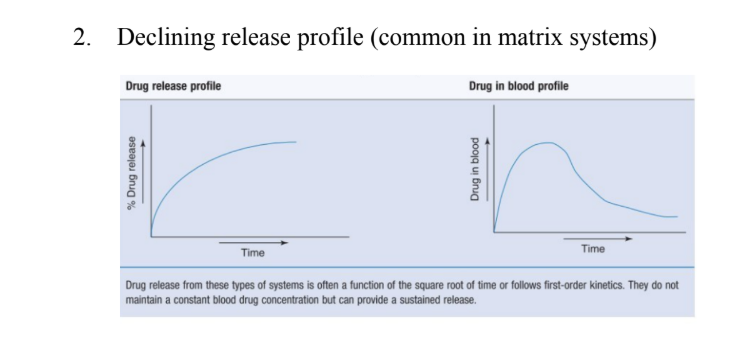
2. Declining release profile.
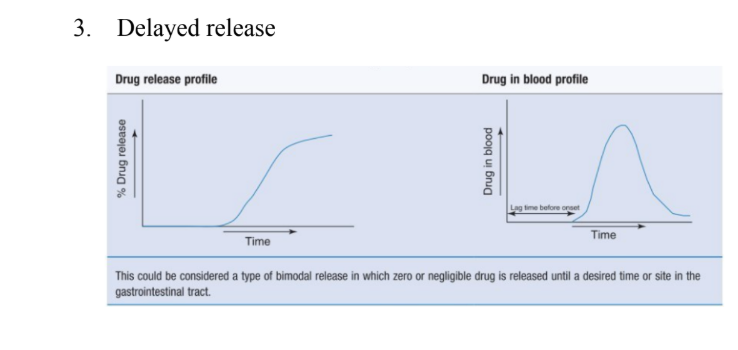
3. Delayed release.
4. Bimodal release.
Describe the constant release profile.
Describe the declining release profile?
Describe the delayed release profile
Describe the bimodal release profile
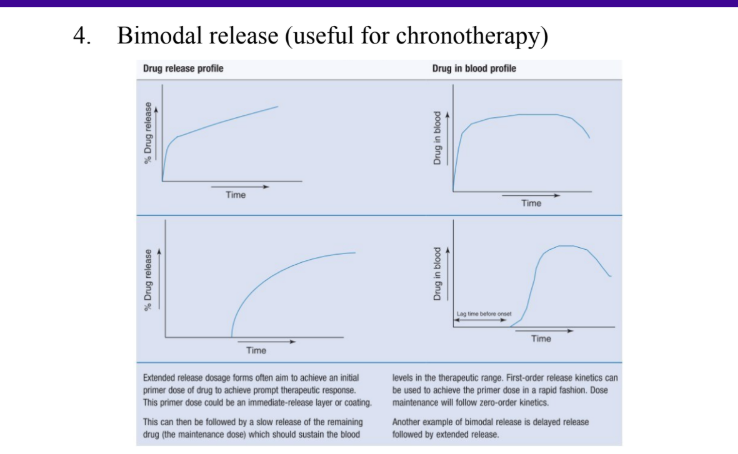
How do you choose the right drug for extended release ?
Choosing the right Drug (Key Factors)
Solubility in aqueous media – Affects dissolution rate.
Intestinal permeability – Determines absorption efficiency.
The rate-limiting step may be:
-Release from the dosage form
-Dissolution of the drug
-Absorption through the GI mucosa
Biopharmaceutics Classification System
Type I: High solubility, high permeability → Best suited for extended release.
Type II: High solubility, low permeability → Absorption-limited, less suitable.
Type III: Low solubility, high permeability → Dissolution-limited, may suit ER
if dissolution is controlled.
Type IV: Low solubility, low permeability → Poor candidates.
What are some other considerations made for extended release?
Half-life: Ideal t1⁄2 = 4–6 h.
• Long half-life → may not need ER (pseudo-sustained effect with immediate release formulations.
• Very short half-life → may require high doses to
maintain blood levels.
Dose & tablet size:
Potency matters – high dose drugs (>1000 mg ER tablets)
can be too large, affecting patient acceptability (paediatrics, geriatrics).
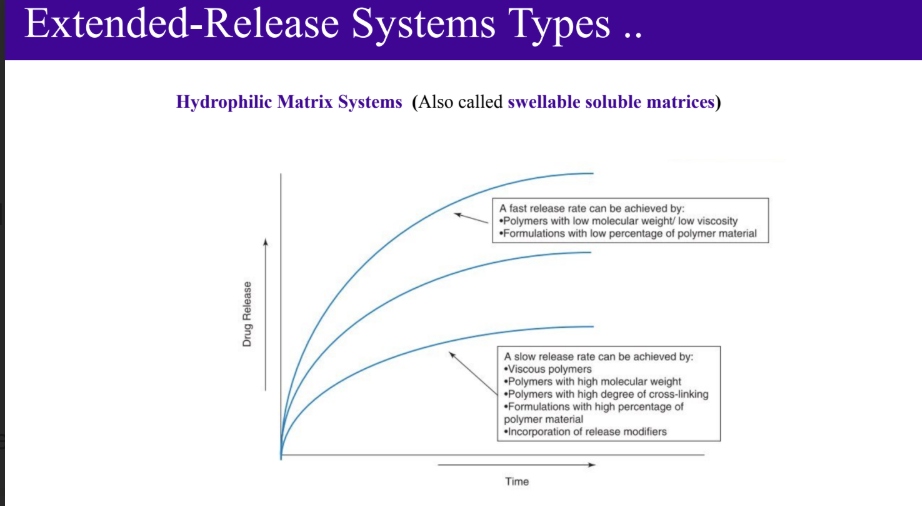
What is the hydrophobic extended release system?
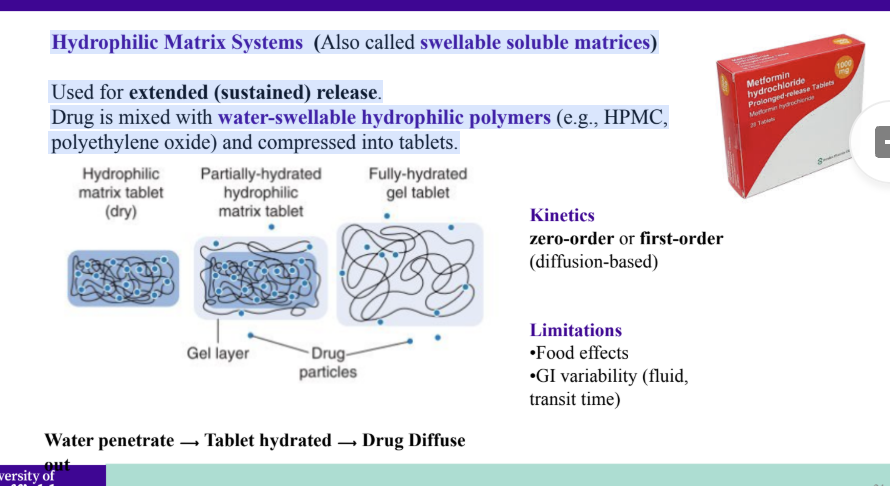
Hydrophilic Matrix Systems (Also called swellable soluble matrices)
Used for extended (sustained) release.
The drug is mixed with water-swellable hydrophilic polymers (e.g., HPMC, polyethene oxide) and compressed into tablets.
What is the limitation of hydrophilic matrix systems?
Limitations
•Food effects
•GI variability (fluid, transit time)
What is an insoluble polymer extended-release system?
Insoluble polymer matrix
Drug embedded in inert, water-insoluble polymer (like a sponge).
Stays intact through the GI tract, unlike hydrophilic matrices.

What is the membrane-controlled extended-release system?

What is the osmotic pressure extended release system?

What are the delayed release system types ?
What does the gastro- resistant delayed release system consist of?

What does the colonic drug delivery delayed release system consist of ?

summary
