Ch. 29- Nuclear Physics
1/9
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
10 Terms
terminology
-atomic number
-isotops
-atomic mass
-The periodic table has elements sorted by number of protons (𝑍), a.k.a. the atomic number. (*determine identity of element)
-Atoms with the same number of protons and different numbers of neutrons (𝑁) are called isotopes of that element
-The atomic mass number (𝐴) is the total number of nucleons in the nucleus of an atom. A is approximately the number of g/mol for the atom or it’s molar mass Mmol: 𝐴 = 𝑁 + 𝑍. (*table=average of isotopes)
ex: He6=Mmol is 6g/mol
-The standard notation to identify different isotopes is: 𝑍/𝐴 𝑋, where 𝑋 is the chemical symbol from the periodic table.
-Note that a deficit of electrons will give the atom a net charge (*but not in this class?)
The Strong Force
-Binds the nucleus together and only acts in the nucleus
-Only effects nucleons. A nucleon is a particle found in a nucleus (proton or neutron)
-Short range: 3.0 fm or less. 1 fm = 1 femto-meter = 10-15 m
-Strongest of the 4 fundamental forces in the universe
-Only attractive! (*doesn’t repel)
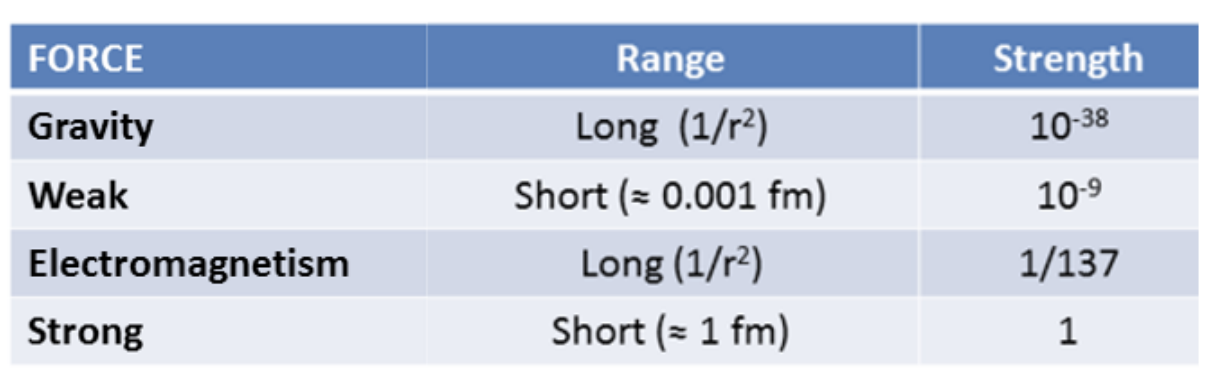
size of nuclei
-where 𝑟0 = 1.2 fm and 1 fm = 1 femto-meter = 10-15 m
-most of an atom is empty space (why a photon would fly right through); nucelus small in comparison but very dense
-nucleus more dense than anything but a black hole, why need a lot of energy to hold the nucelus together; protons and nutreons packed tightly together
true or false: The nuclear density is roughly constant ≈ 2.3 × 1017 kg/m3
true! bc protons and neutrons roughly same mass, so A cancels out and just left w/ constants
neutrinos
-Italian for “tiny neutral one”
-Theorized to exist in 1930 to conserve momentum in some radioactive decays
-Experimentally discovered 1956.
-Symbol: 𝝂 𝟎 the lower-case Greek letter “nu”
-The anti-neutrino has the Symbol: 𝝂bar 𝟎
-Neutrinos have:
a very small mass (𝑚𝜈 ≈ 1x10-37 kg)
zero charge
travel at about 99% the speed of light
are NOT affected by the Strong Force
-They are hard to detect, since neutrinos travel very far (through the Earth and you for example) without interacting with any matter; harmless.

particles vs anti-particles
-same mass, opposite charge, opposite spin
-particles: neutron, proton +, electron -, photon
-anti-particles: neutron bar, proton -, electron +/”positroin”, photon bar
The Weak Nuclear Force
-transforms particles in the nucleus to make the nucleus more stable
-charge is convserved
-2 types of beta decay
Neutron decay: can occur outside the nucleus; neutron turns into protron
Proton decay: can only occur in a nucleus; proton turns into neutron

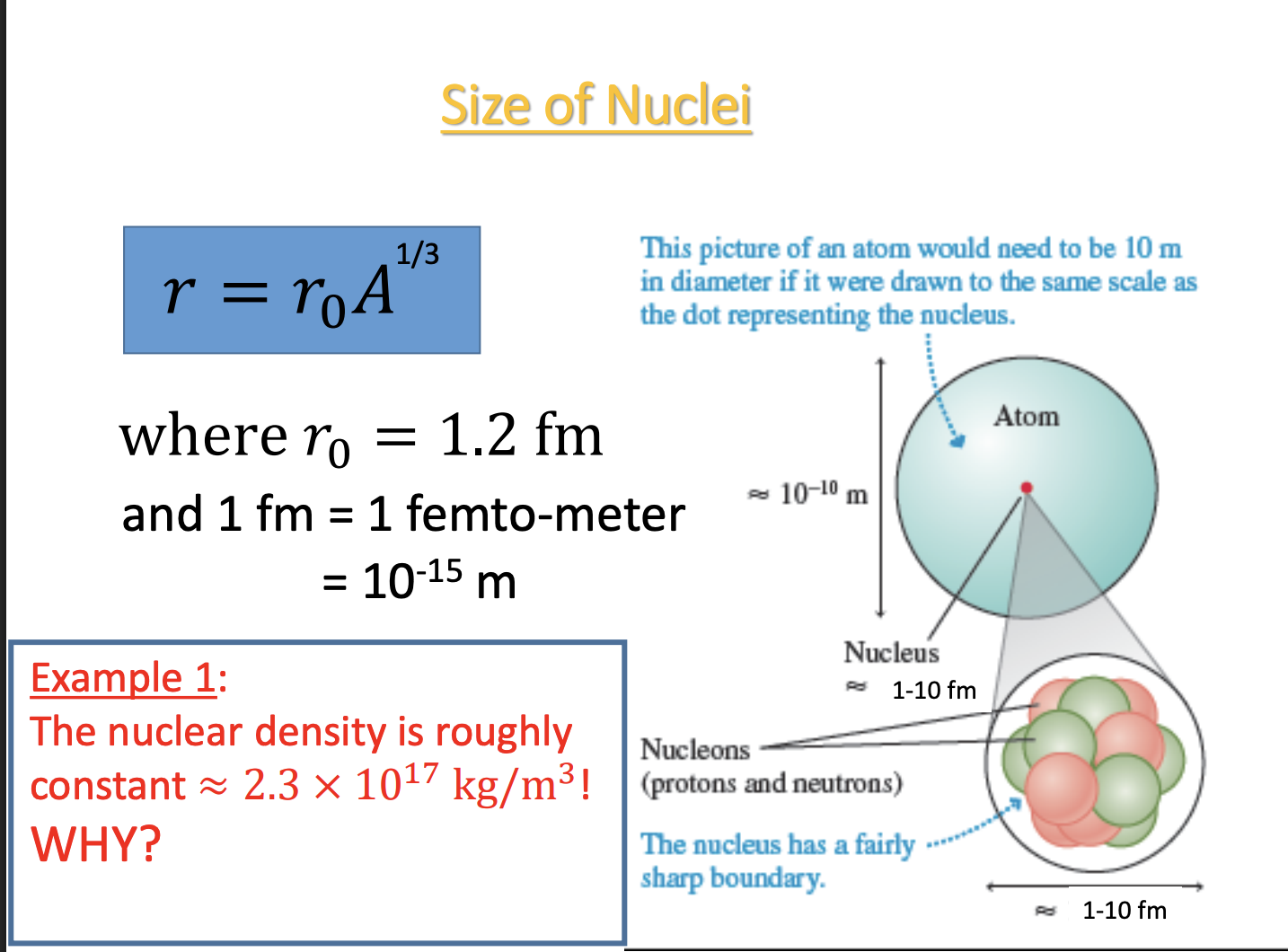
PET
-A use for positrons (anti-electrons, e+) : Positron emission tomography (PET)
-injected w/ short half-life radioactive material and edmits positrons → positrons hit electrons and send out photons (in opposite directions) → detector detects photons and their energy
-particle + anti-particle = annihilate (and edmit photons can detect; e- + e- → 2 photons)
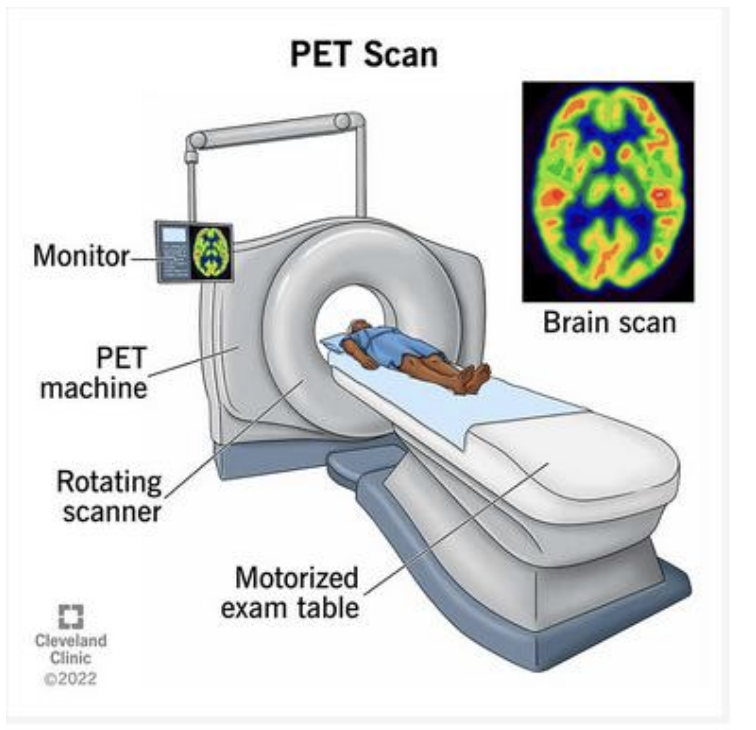
Nuclear Stability for Z < 10
-If N and Z are equal; the nucleus is generally stable and is safe to ignore Coulomb repulsion (electric force) (*repulsion their but not significant/strong enough; stable isotope)
-For larger Z (above 10) Coulomb repulsion is not negligible
-ex picture: Particles in a box model for 12C (*stable bc same # of protrons and neutrons; 13C radioactive bc extra neutron)
-The Pauli exclusion principle permits two protons and two neutrons per level
*most things are radioactive
Nuclear Stability for Z > 10
-What is the effect of taking into account electrostatic repulsion on our diagrams?
-Answer: all energy levels for protons (including the ground state) are raised relative to the neutron levels
*repulsion of proton significant; highest energy in proton box; raise 1st proton level to neutron level; want same number of particles in highest level box