Cell bio exam 4
1/74
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
75 Terms
Tyrosine kinase receptors , SARS - CoV2 and spike proteins
TYROSINE KINASE RECEPTORS
EGFR receptors - type of RTK Mutated in many forms of cancer
Deletion of Exon19 occurs in ~10-20% of lung cancers
Makes EGFR receptors more active
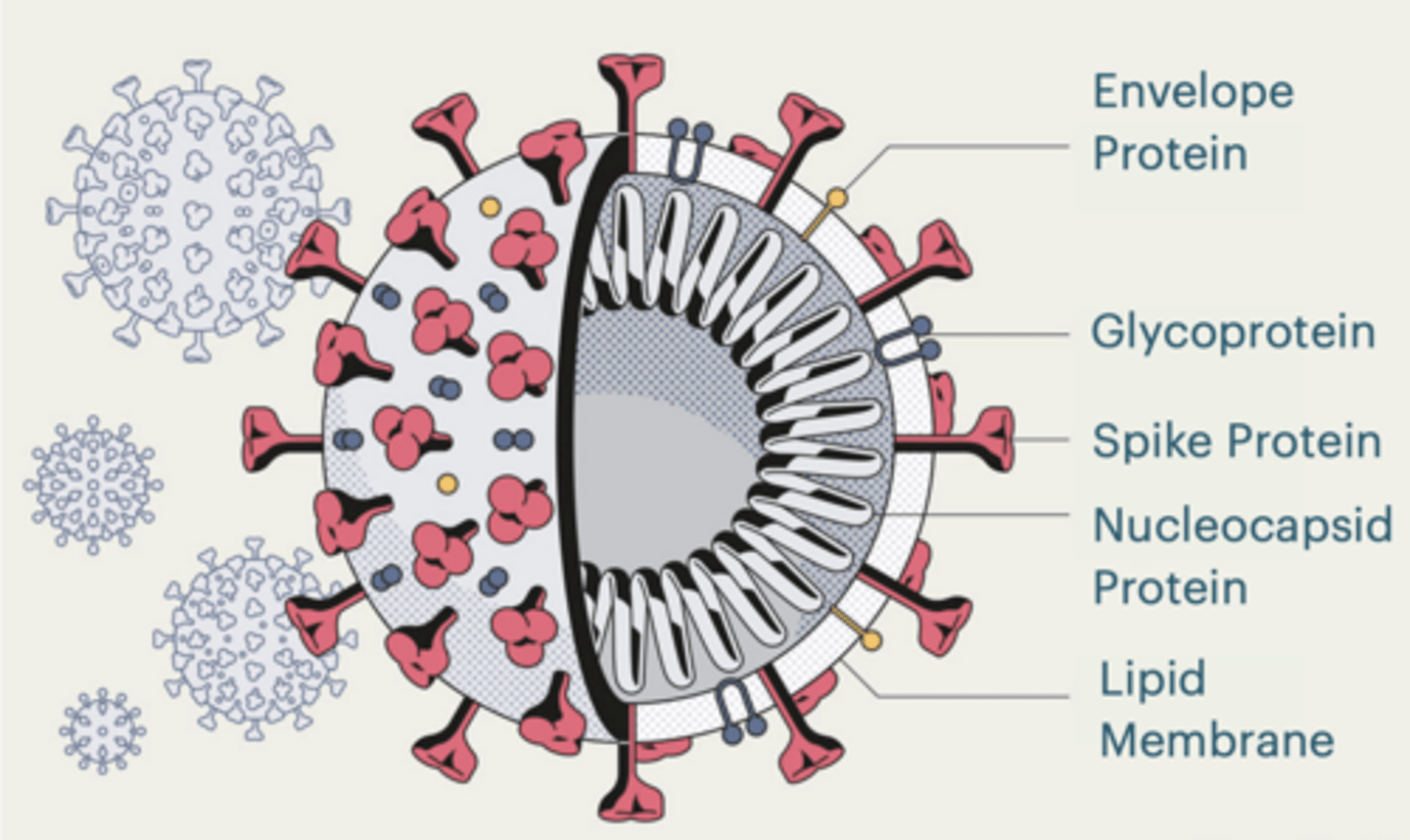
SARS-CoV-2
Corona virus
Lipid membrane surrounding virus
spike proteins - That bind host receptor (ACE2)
RNA genome
Nucleocapsid protein protects RNA
Causes the disease COVID-19
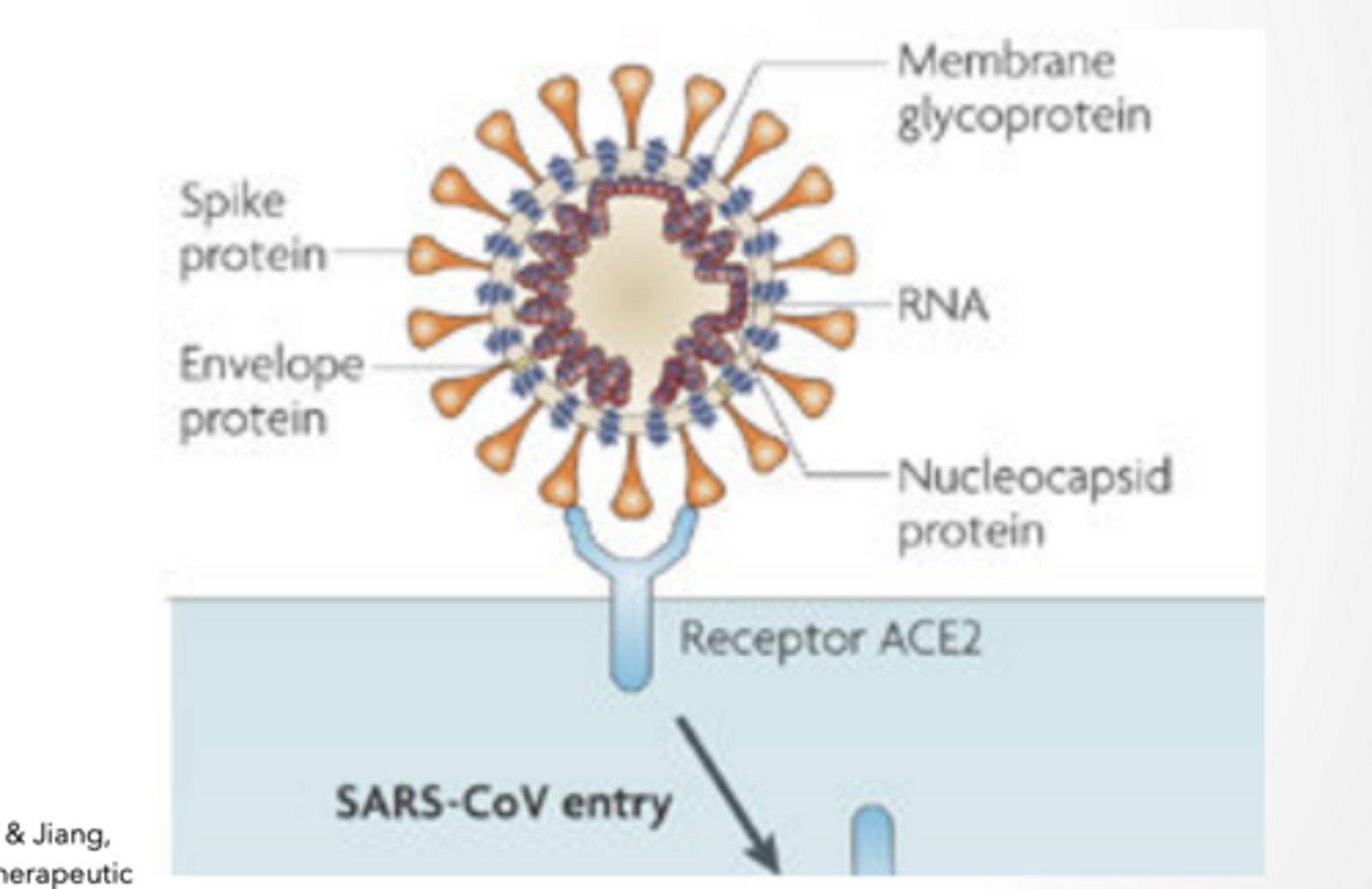
SPIKE PROTEINS
binds ACE2
changes shape and fuses w cell membrane
THIS IS THE ENTRY MECHANISM
Feet of Sars life cycle
feet
Attachment and Entry and processing
ATTACHEMENT
Spike protein Binds to ACE-2 receptor
Angiotensin converting enzyme 2
Found in heart, lungs, and testes most prevalently
This is protein-protein interaction → very specific
Cells must have:
ACE2 (receptor)
AND enzymes like TMPRSS2 or CTSL
More enzymes = easier infection/entry
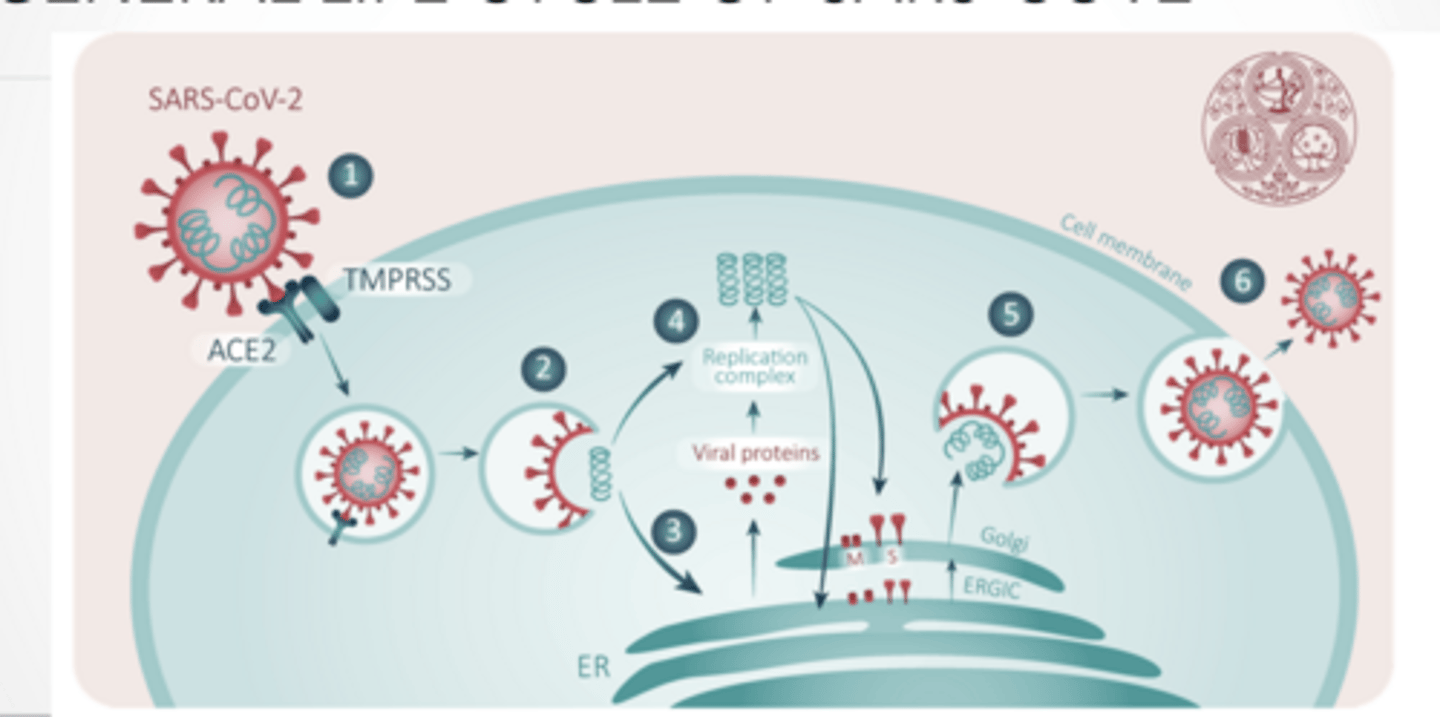
ENTRY AND PROCESSING
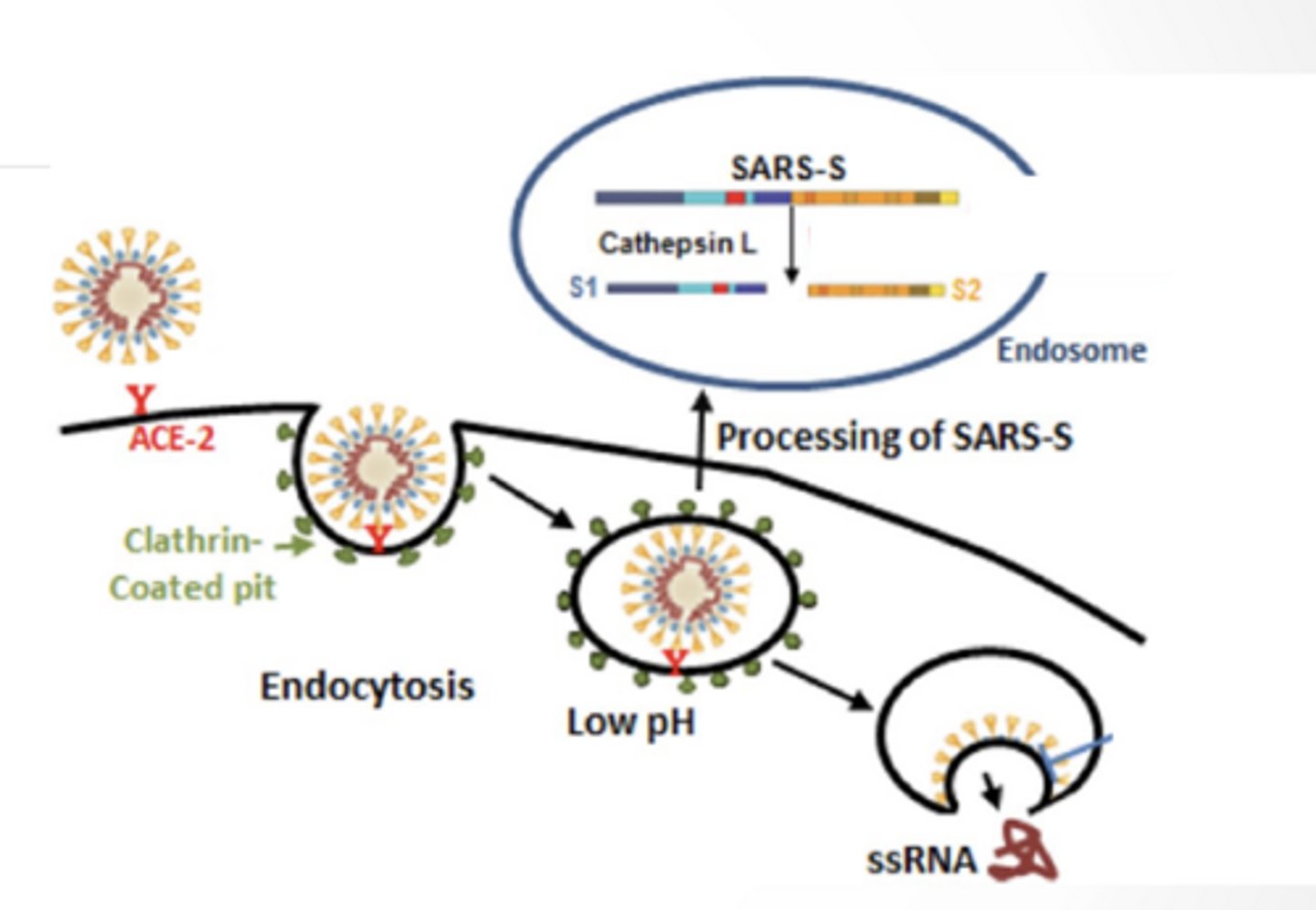
ENDOCYTOSIS
Enters through receptor mediated endocytosis
Low pH in lysosome/ endosome free RNA genome from virus
Viral membrane protein facilitate release of genome into cell
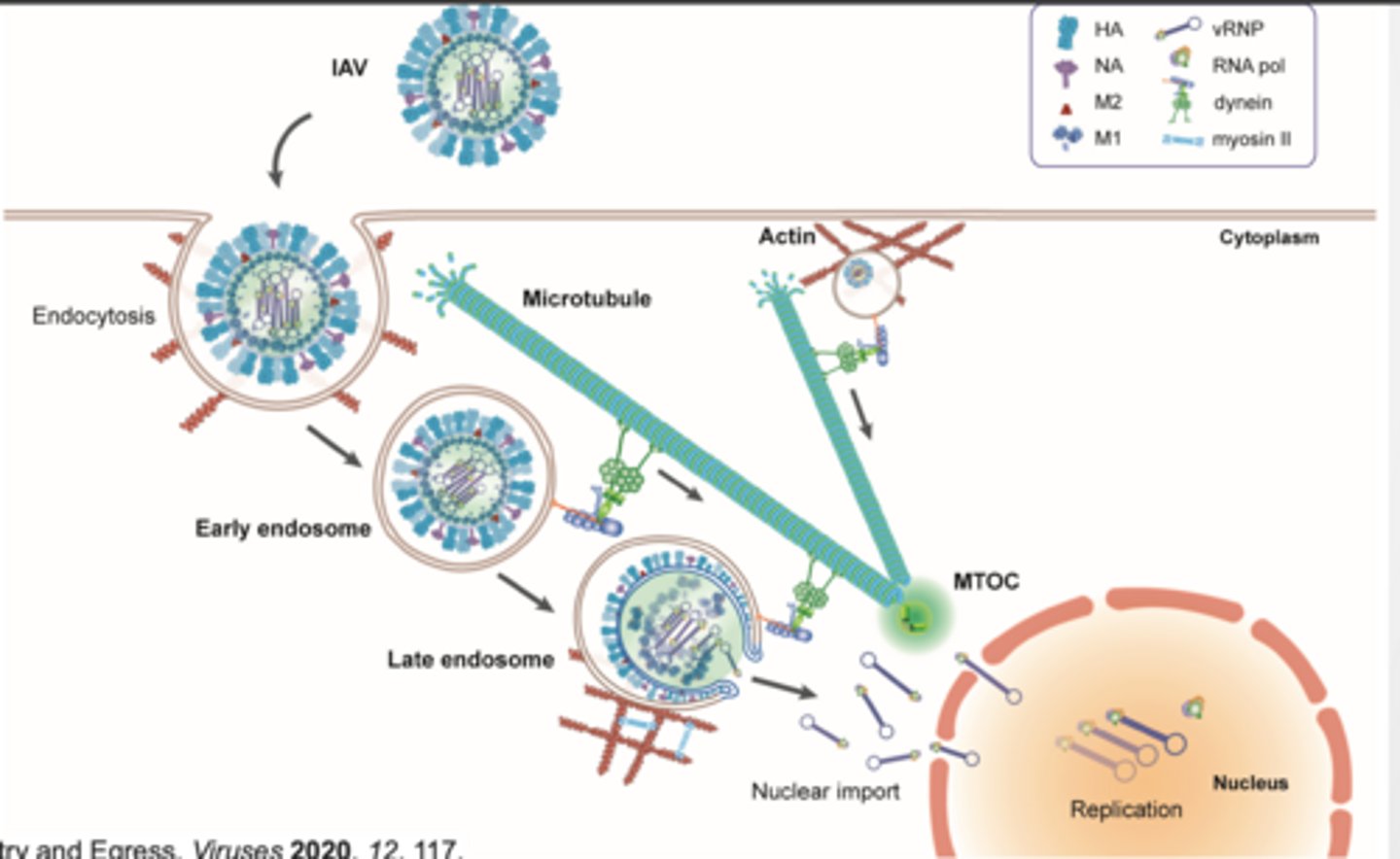
TRANSPORT ALONG MICROTUBULES
Flu viral entry is similar to COVID
Endosome travel along microtubules (dynein) Actin plays a role in endocytosis
endocytosis feet
feet
more feet
feet
Replication and packeging
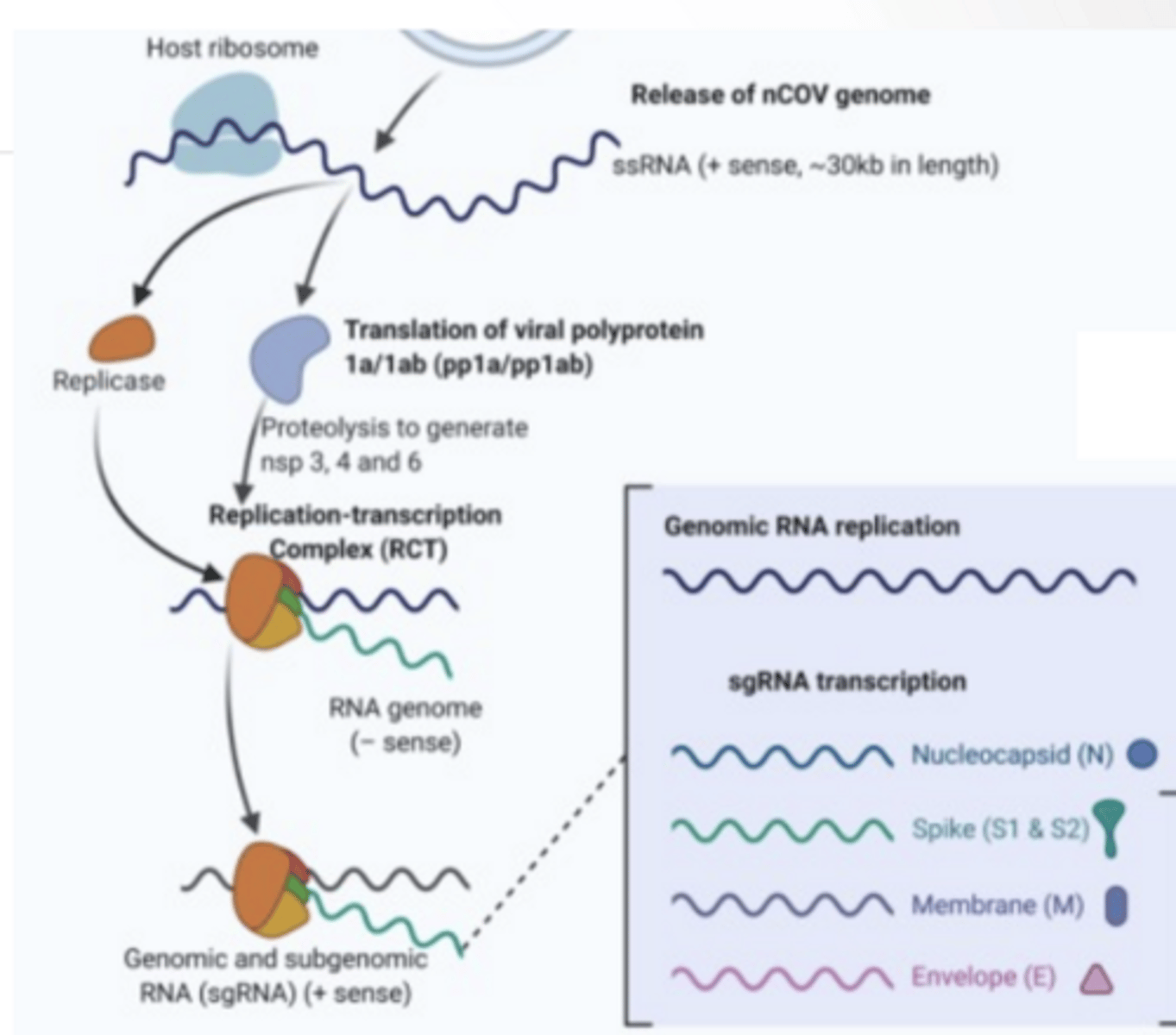
VIRAL REPLICATION
Host ribosome translate viral RNA
Replication complex
RNA dependent RNA polymerase Replicated genome
Proteases
Cleave proteins to functional viral units
VIRUS STOPS CELL FROM MAKING ITS OWN PROTEINS
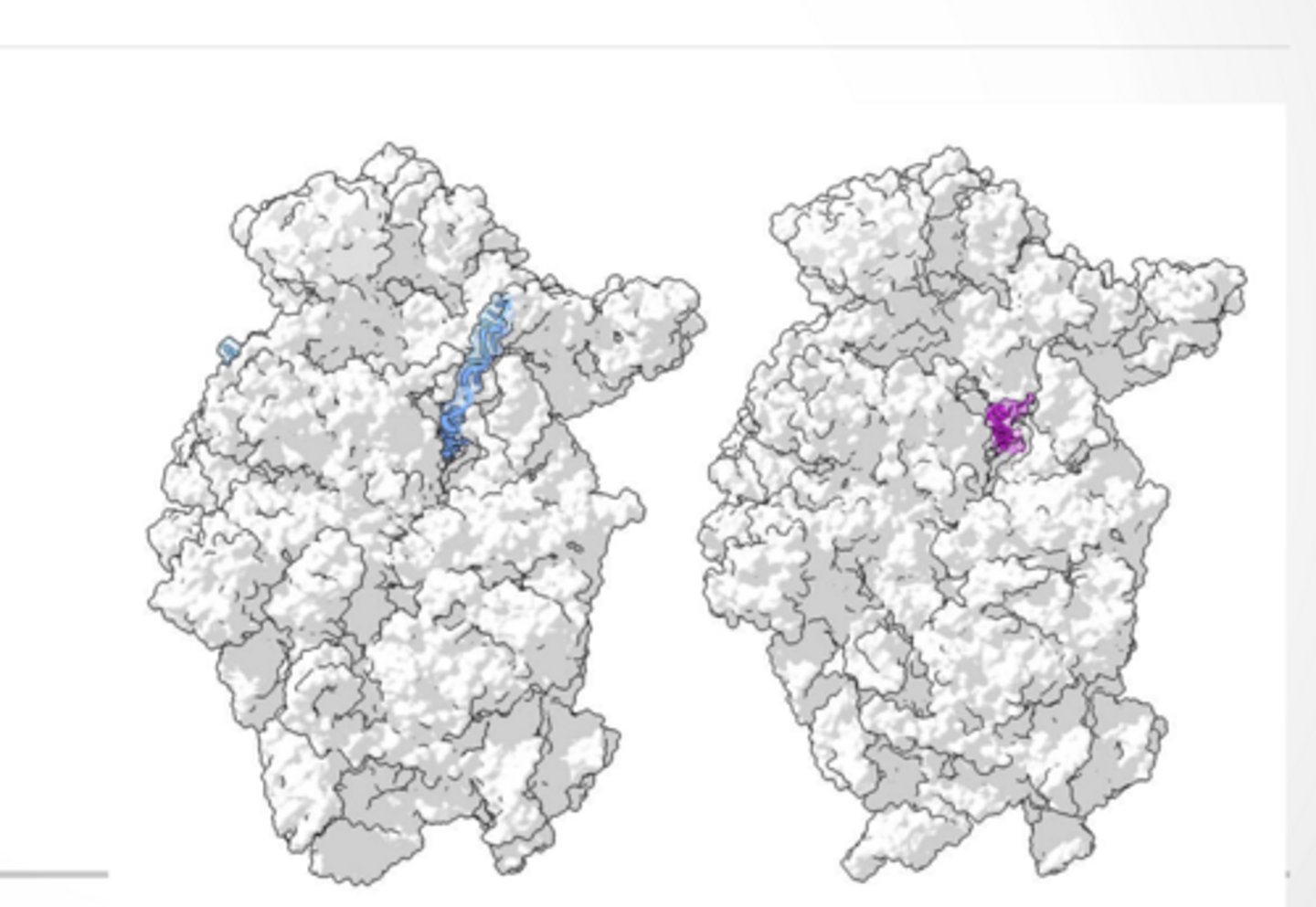
Nps1 - viral protein that shuts down host protein production.
Possible that virus is better at attaching to ribosome so that when Nps1 falls out the virus can take over
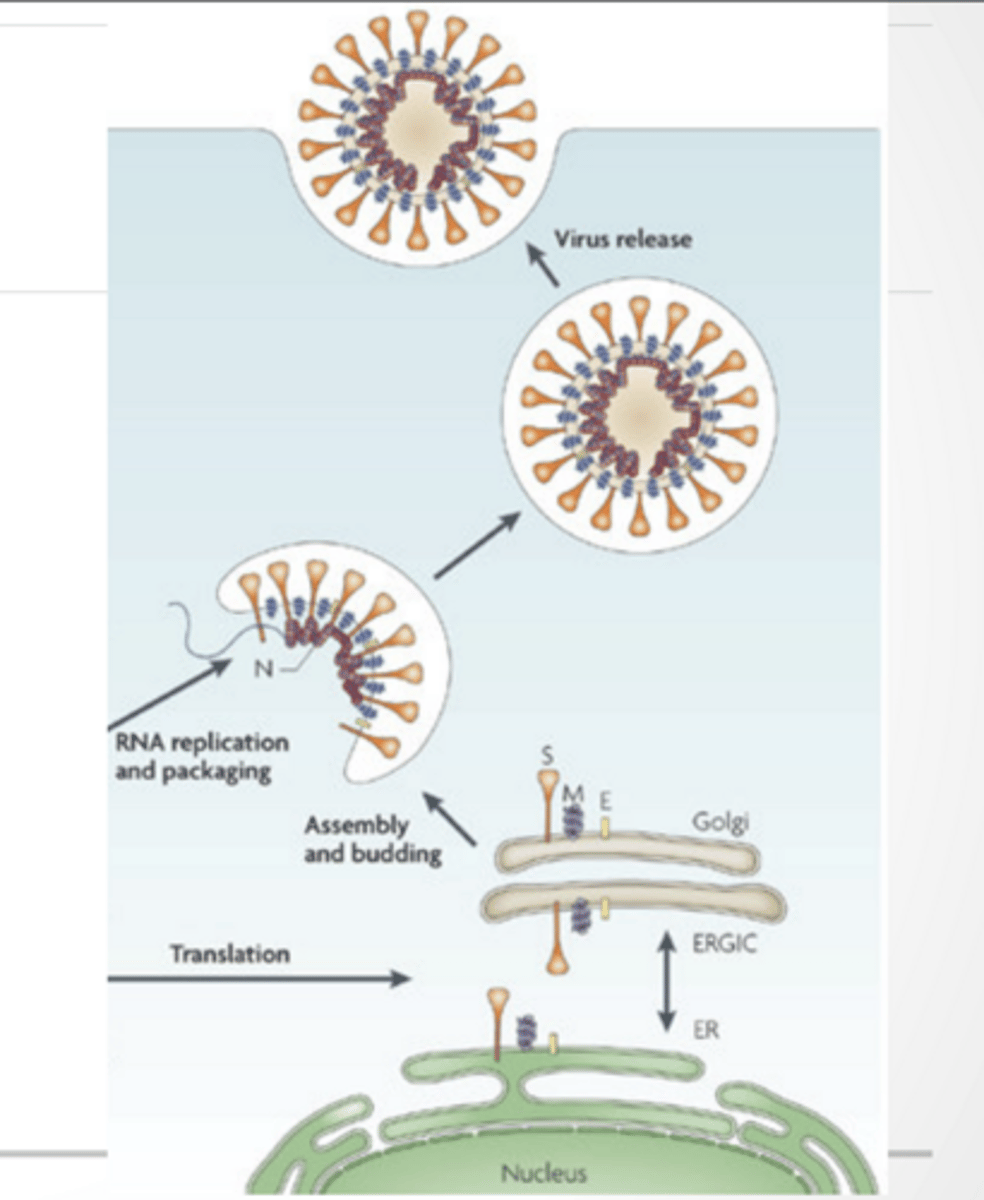
PACKEGING
Virus makes droplets that house RNA
Virus escapes through a Nsp3 protein
Nsp3 helps: protect viral RNA from immune detection
RNA is replicated Proteins are translated into the ER and move through the Golgi Vesicles from Golgi envelope viral genome Exocytosis occurs
Feet of Nps1
Nps1 interacting with ribosomes Pink -Nps1
RNA -blue
Feet of packaging
feeet
Export and cell death
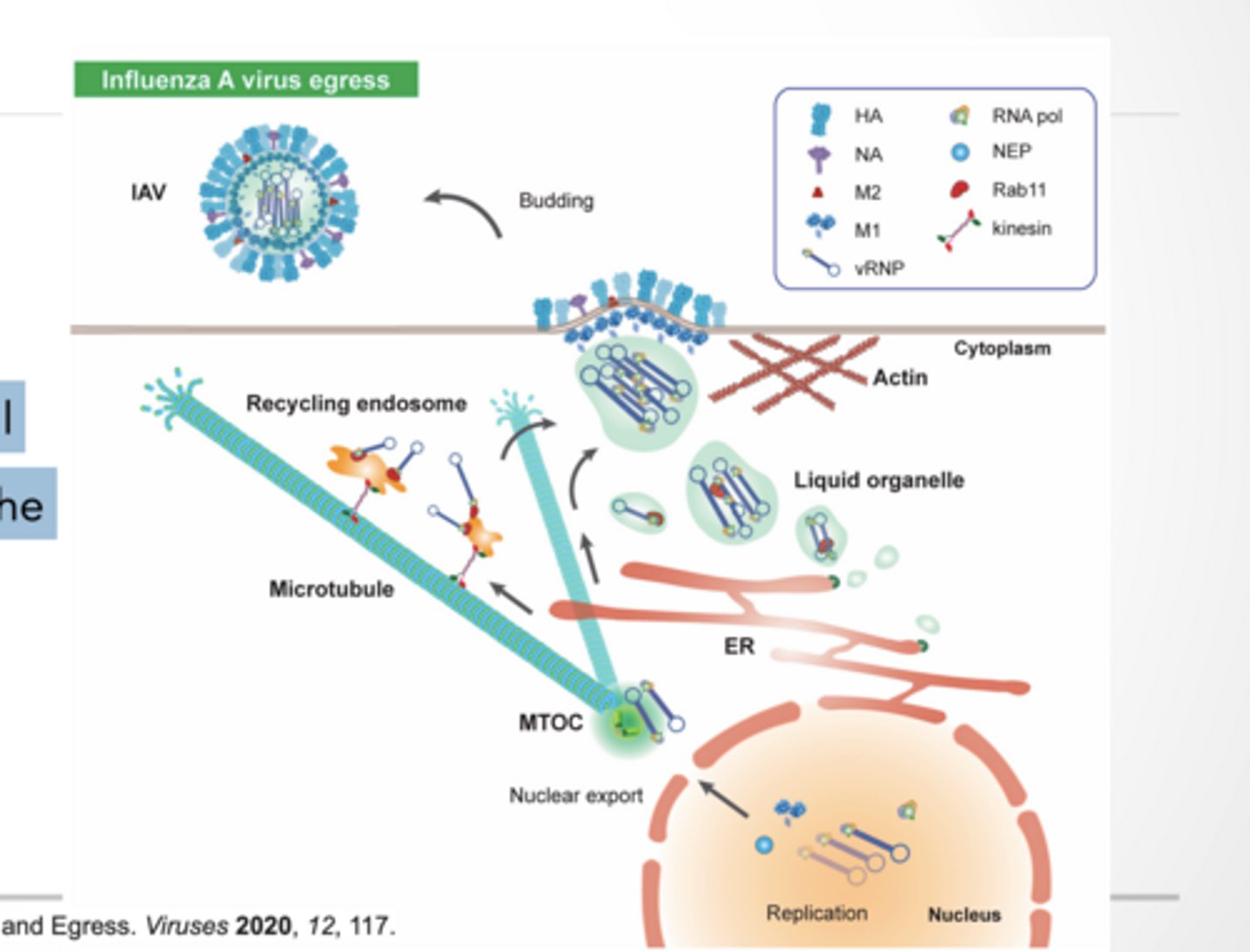
EXPORT
EXOCYTOSIS Influenza is model for this as well
Particles are transported along the microtubules and spit out in vesicles.
Actin plays a role in exocytosis
HOW DO VIRUSES KILL CELLS
Some virus induce cellular degradation as their escape mechanism
Corona virus can induce apoptosis
Some types Due to co-opting of cellular machinery
Specific protein may induce cellular response
Apoptosis (programmed death → controlled)
Necrosis (cell bursts → messy inflammation)
Overusing cell machinery until it collapses SARS-CoV-2 can trigger apoptosis or stress responses, contributing to tissue damage.
Where to target a drug
Where to target a drug (general)
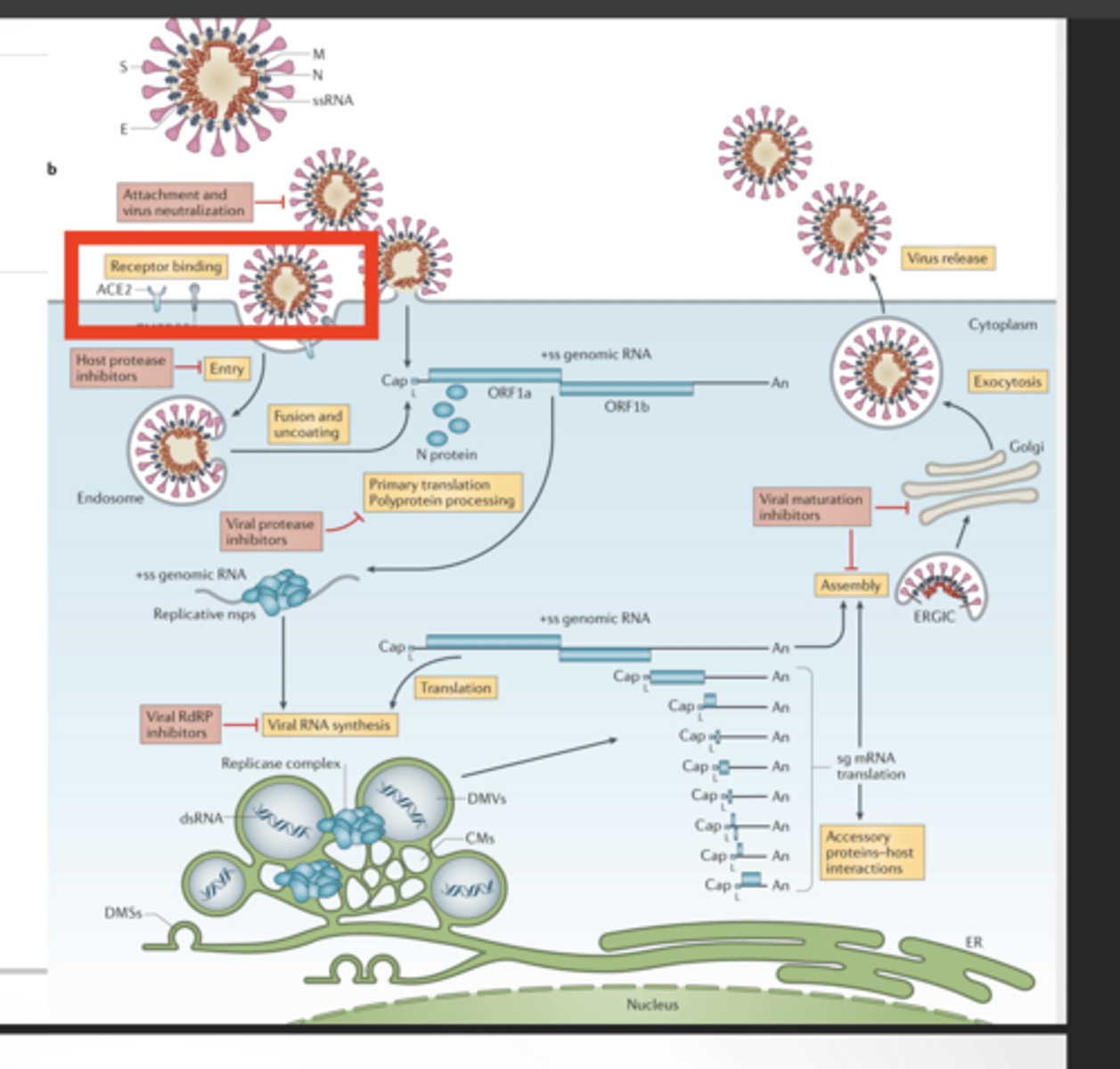
You can block the virus at multiple steps:
Entry (ACE2 binding)
Drugs here stop the virus from even getting inside:
Block spike protein
Block ACE2 interaction
Block TMPRSS2 (host protease)
Protease activity (cutting viral proteins)
RNA replication (RdRp)
Virus makes one long protein → needs proteases to cut it
nsps (like Nsp3) form replication machinery
RdRp copies RNA
Drugs here:
Paxlovid → protease inhibitor
Molnupiravir → RdRp inhibitor
Assembly/release
Why target the spike protein and mRNA vaccines
TARGET THE SPIKE PROTEIN
Spike is glycosylated
Antibodies have a hard time
Convalescent plasma seems to be effective for acute treatment
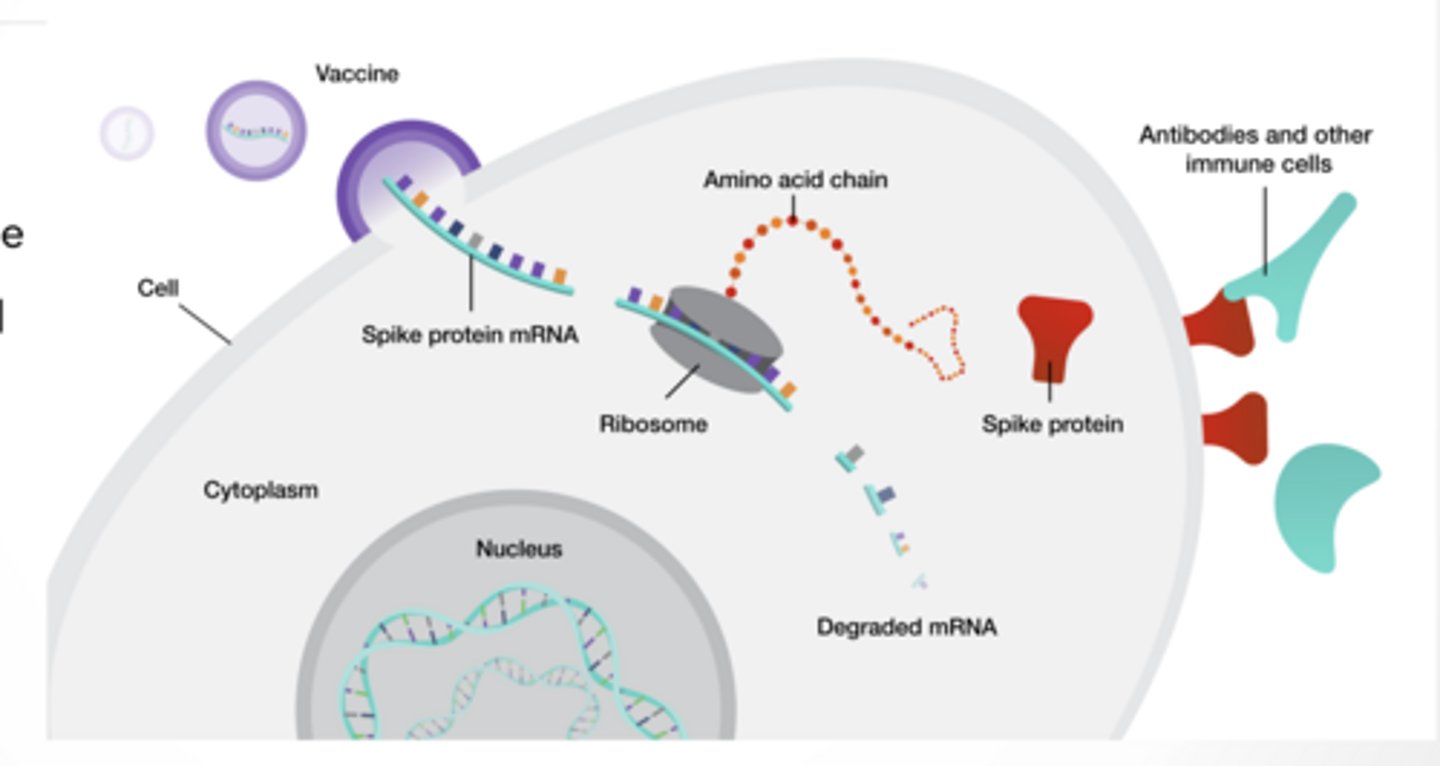
mRNA vaccines
Body makes the spike protein but nothing else
Natural glycosylation occurs so self made antibodies are better
All vaccines do the SAME goal ----> show immune system spike
mRNA vaccines – how they work
mRNA of spike protein is injected and protein is made
mRNA is degraded just like all your normal mRNA
Immune system is presented with a foreign protein made by mRNA
Vaccines
Vaccines:
Reduce infection risk
MASSIVELY reduce severe disease/death
Variant names
Alpha/beta – first wave
Delta – second wave
Omicron – 3rd wave
Virus evolves by:
Mutations
Recombination (mixing variants)
Mutations mostly occur in:
RBD (receptor binding domain) → affects ACE2 binding
NTD → affects antibody recognition
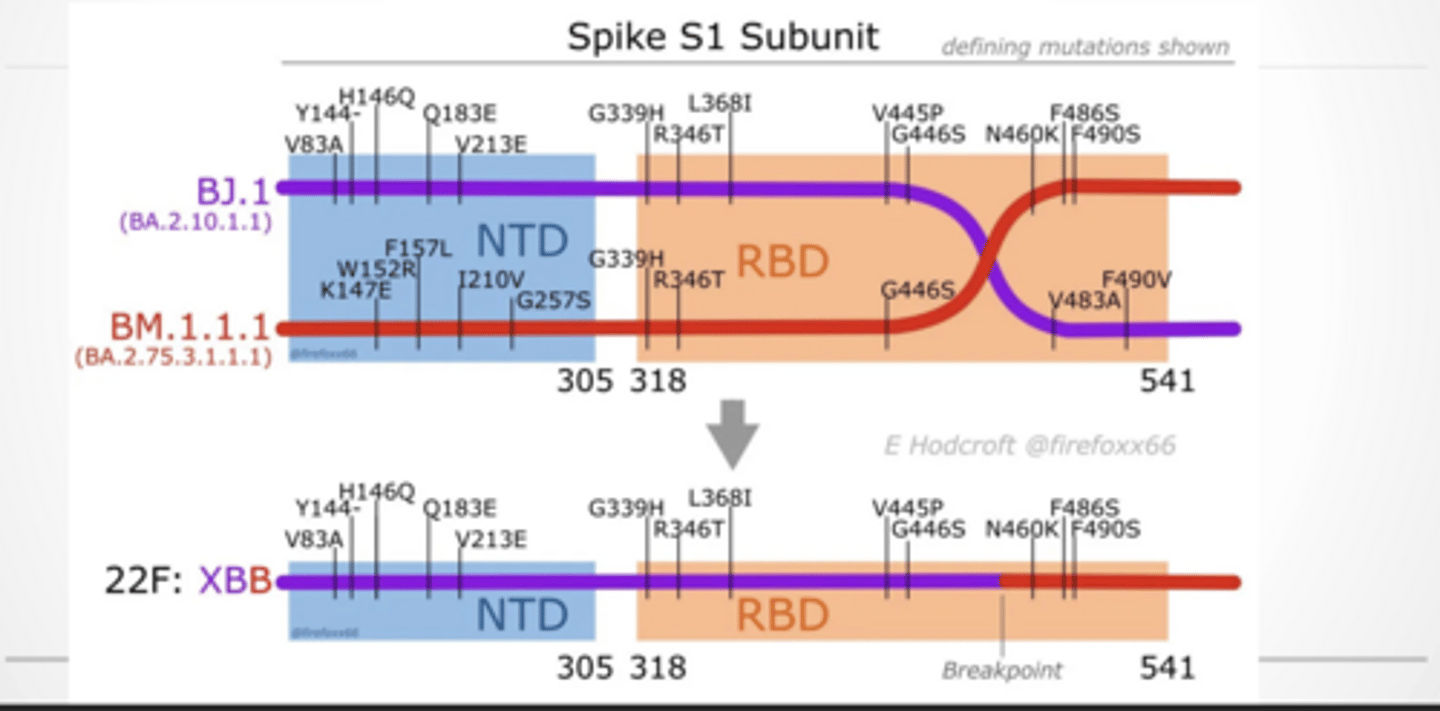
Spike has two major parts:
S1 → binds ACE2
S2 → fusion
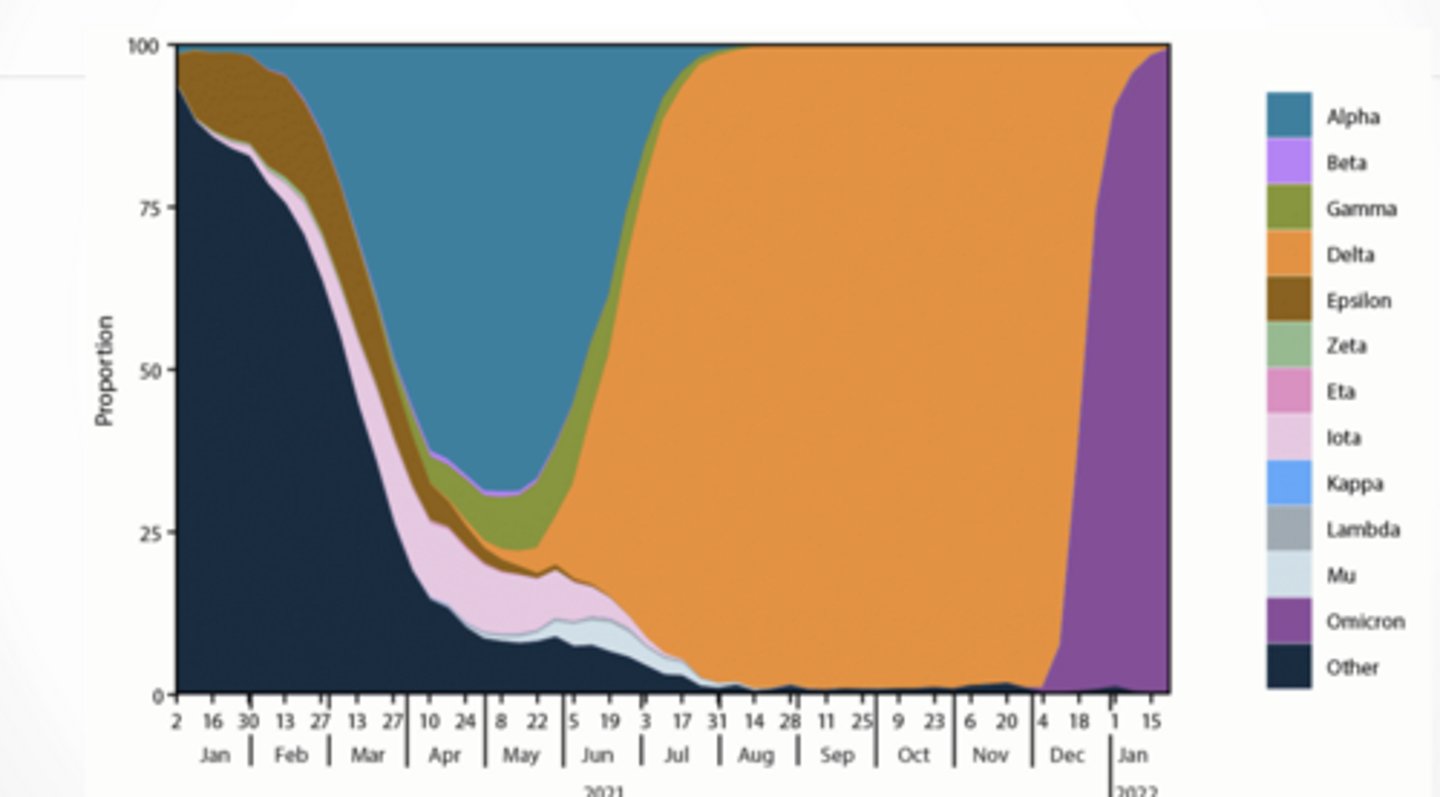
Variant prevalence
Variant prevalence over time
Early → Alpha/Beta
Mid → Delta takes over
Late → Omicron completely dominates
Variants don’t stop evolving.
You’ll see new lineages (XBB, BA, etc.)
Each has slight mutations → affects:
transmissibility
immune escape
Long COVID = symptoms lasting months after infection
long-term damage/response
The more reinfection that occurs the higher the rate of long covid
Long covid feet
feet
DIFFERENCES IN ION CONCENTRATION CREATE MEMBRANE POTENTIALS
DIFFERENCES IN ION CONCENTRATION CREATE MEMBRANE POTENTIALS
Small amount of ion movement can cause large changes in membrane potential (Vm)
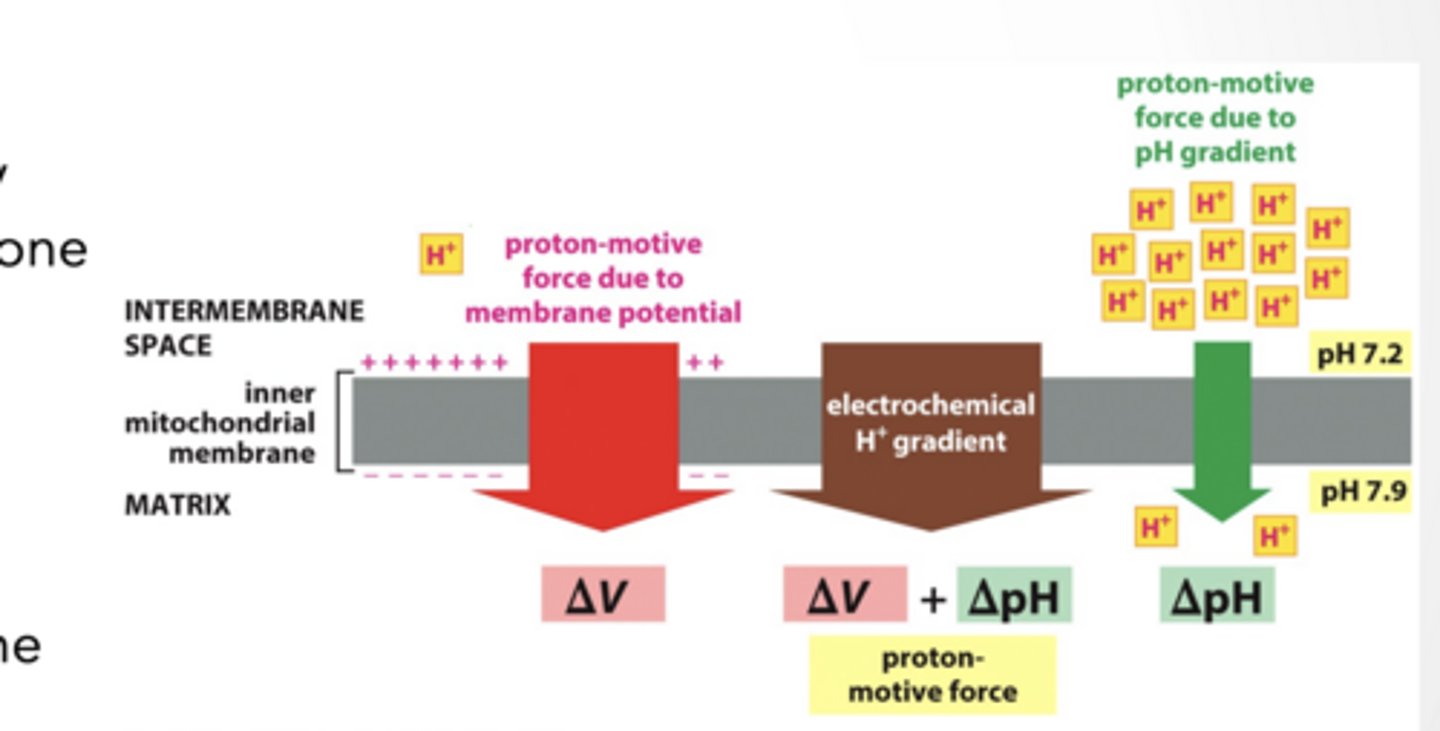
Membrane potential = the voltage (electrical difference) across a cell membrane
ions involved are VERY close (<1nm) to the membrane
Electrochemical gradient is created by protons being more concentrated on one of the membrane
2 Parts (concentration AND electrical gradient)
Both pushing the same directions
Both caused by the movement of one ion
Membrane potential gradients
MEMBRANE POTENTIAL IS BALANCE OF VOLTAGE AND CONCENTRATION GRADIENTS
Membrane potential is measured in reference to the extracellular space
Membrane potentials may not have both forces going the same way
concentration and voltage gradients could be going opposite.
Inside cell:
K⁺ HIGH
Na⁺ LOW
Ca²⁺ VERY LOW
Outside cell:
Na⁺ HIGH
Cl⁻ HIGH
Ca²⁺ HIGH
These gradients power:
action potentials
signaling
GENERATING MEMBRANE POTENTIALS
GENERATING MEMBRANE POTENTIALS
NON-SELECTIVE CHANNEL: + ONLY
Channel opens and ions flow
Keep track of relative charge either side of the membrane(grey) and concentration (red)
Net Change:
Out: +3 charge, 3 ions
In: -3 charge, 3 ions
Final result is a balanced membrane
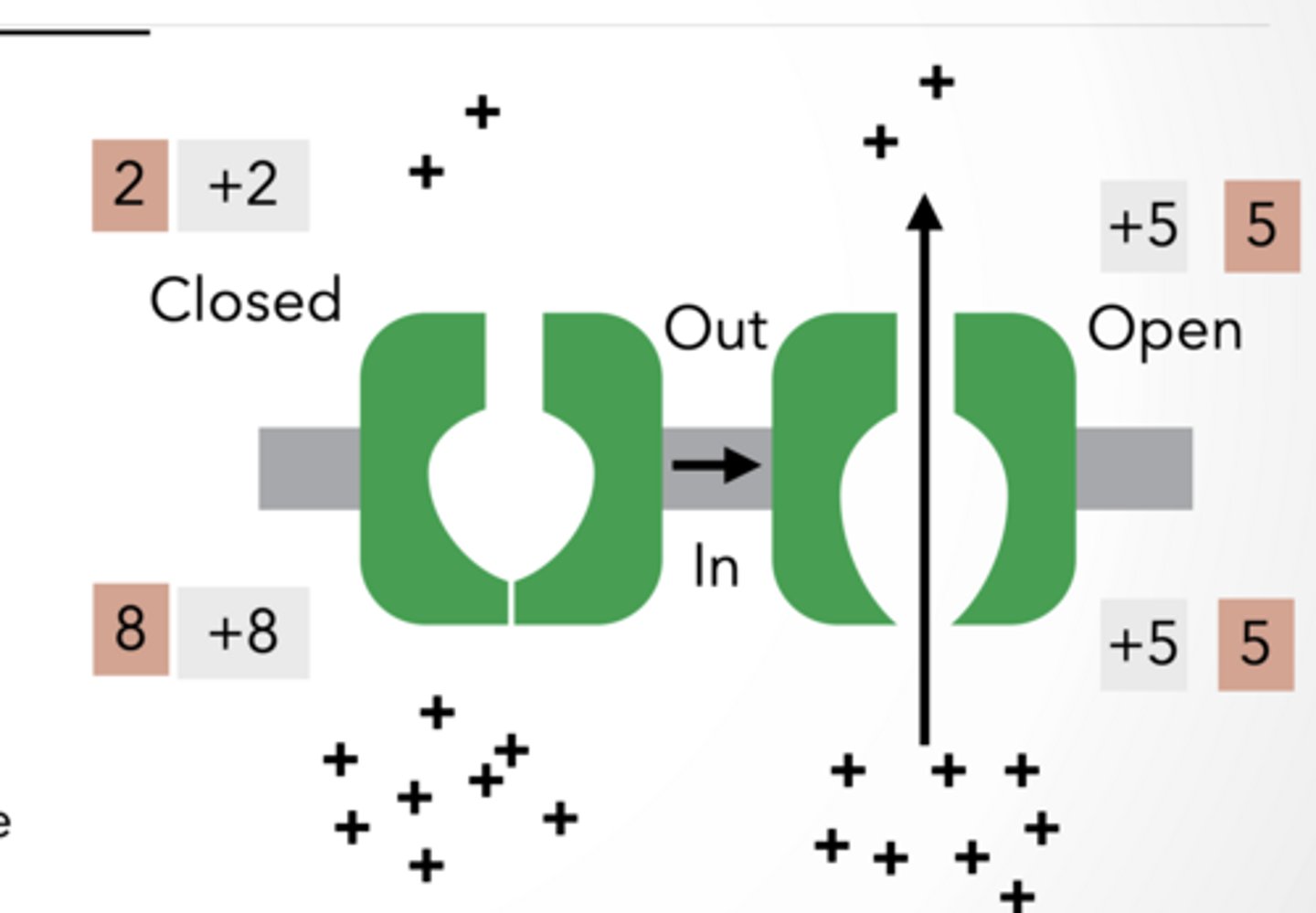
NON- SELECTIVE CHANNEL: + AND -
Both + and − ions move, so charges cancel out. Even though ions move, there’s still no electrical difference, so no membrane potential is generated.
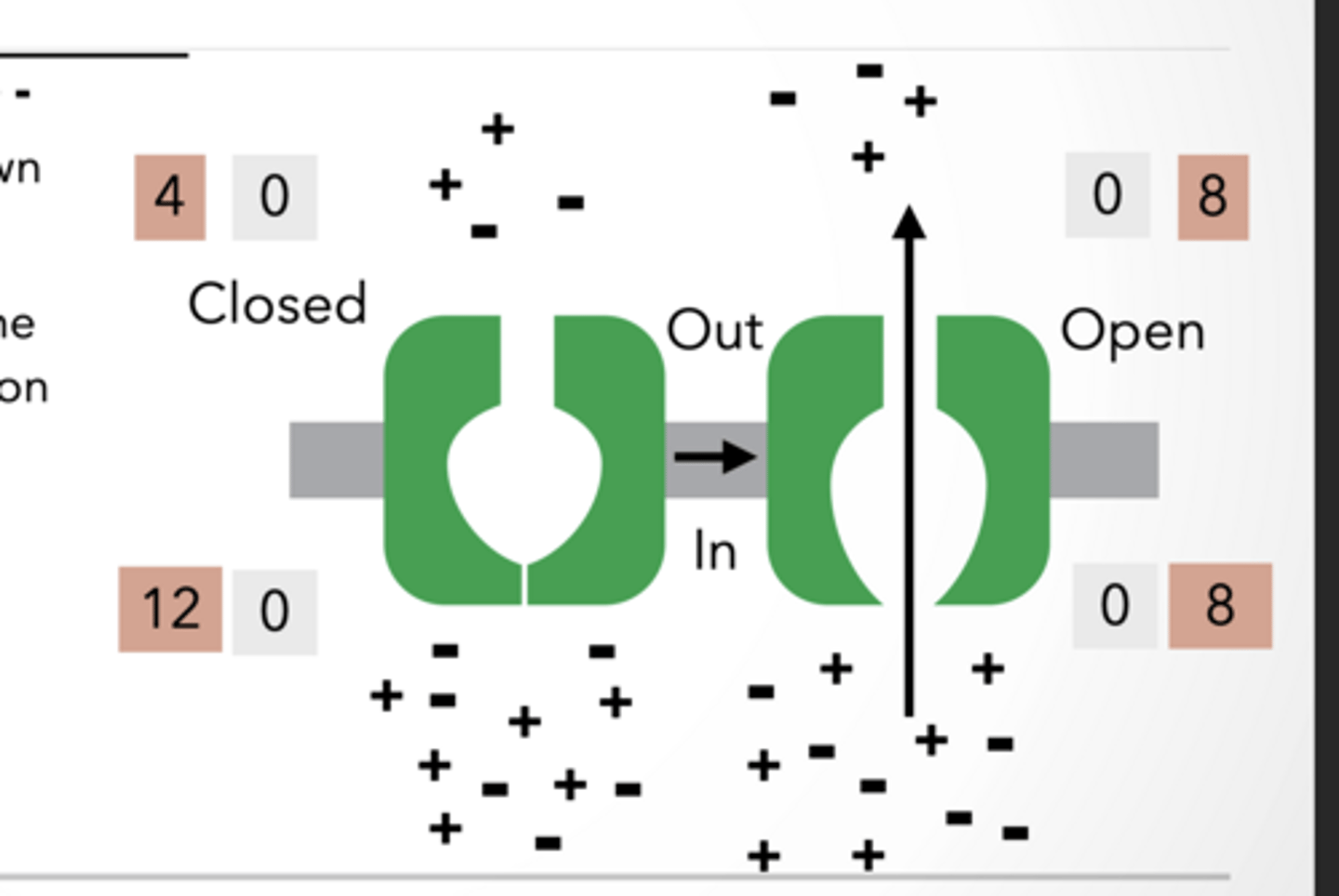
SELECTIVE CHANNEL: + AND -
Only + ions flow down concentration gradient
Only positive ions move → charges become uneven across membrane
This creates a voltage difference = membrane potential
Final result is a voltage gradient
Non selective + - feet
feet
selective feet
feet
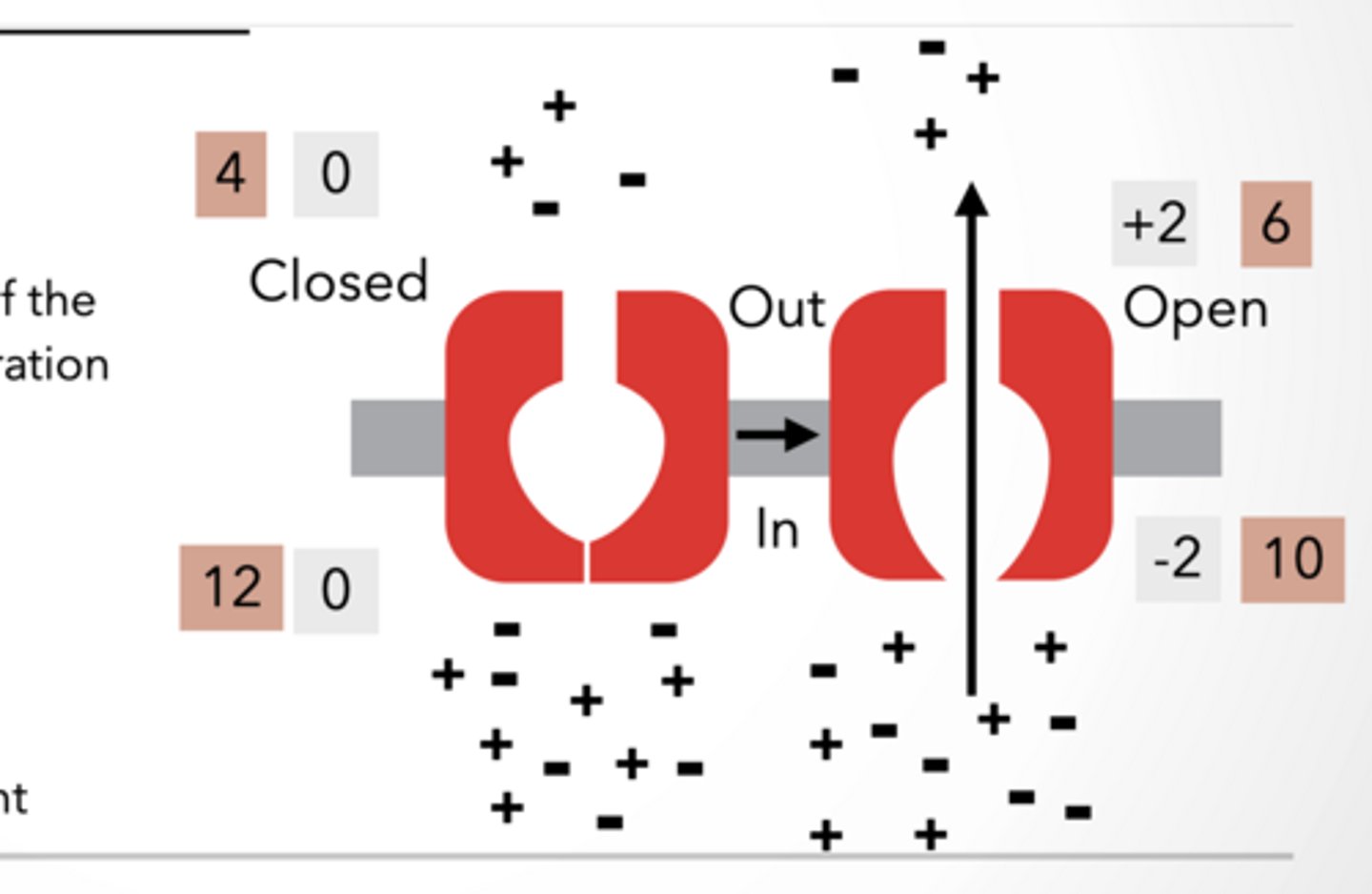
Anatomy of neuron resting potential and ion channel
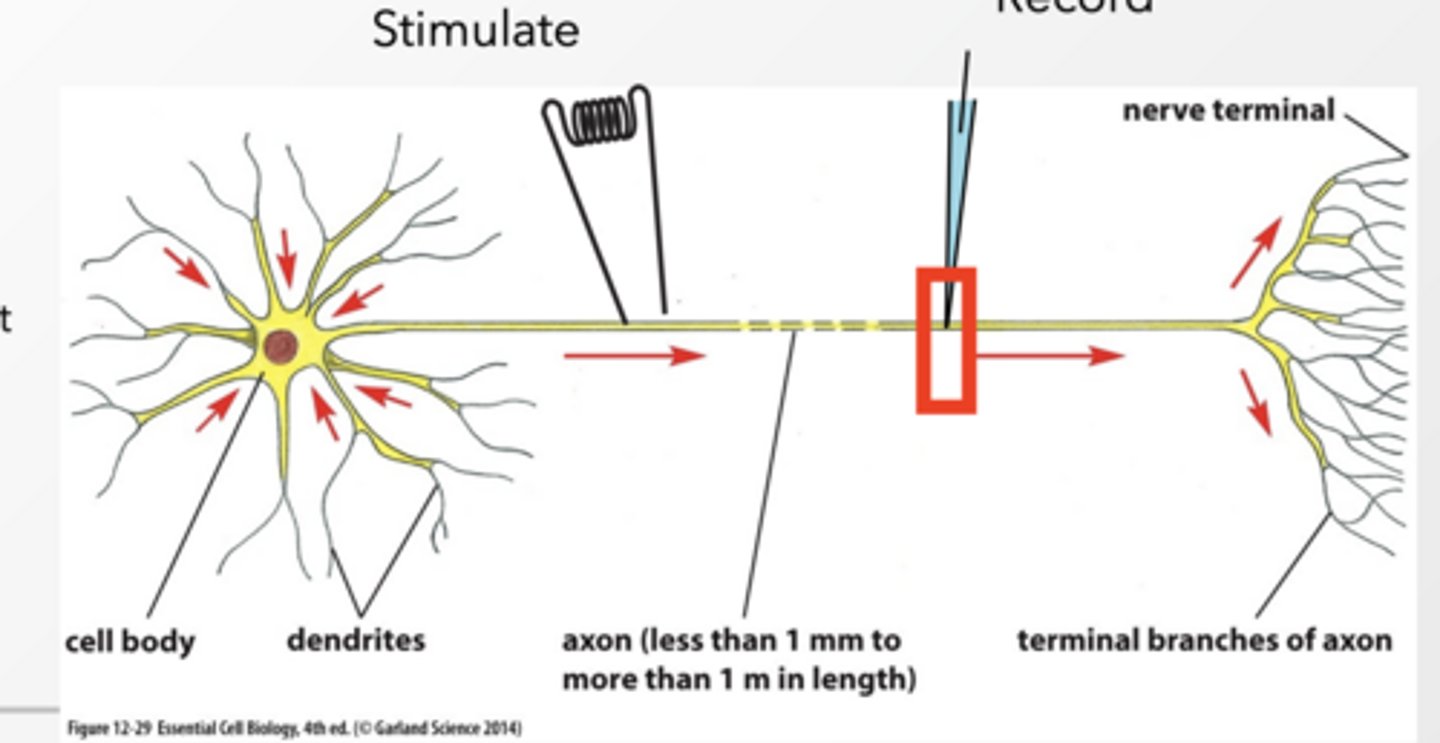
Anatomy of a neuron
Dendrites = receive signals
Cell body = integrates signals
Axon = sends signal (action potential)
Terminal = communicates with next cell
Voltage can be recorded in a neuron
We place an electrode in a neuron and measure membrane potential over time → this gives us graphs like action potentials.
Resting potential – voltage difference when the cell is not transmitting a signal
AP is the voltage change in the cell that is transmitted along the axon
Ion channels have 3 states:
Closed → nothing happens
Open → ions flow → voltage changes
Inactivated → temporarily shut off
Depolarization = more positive inside
Anatomy feet
feet
What do neurons do? Synapse
What do neurons do?
Detect and response to stimuli
Carry signal from one place to another
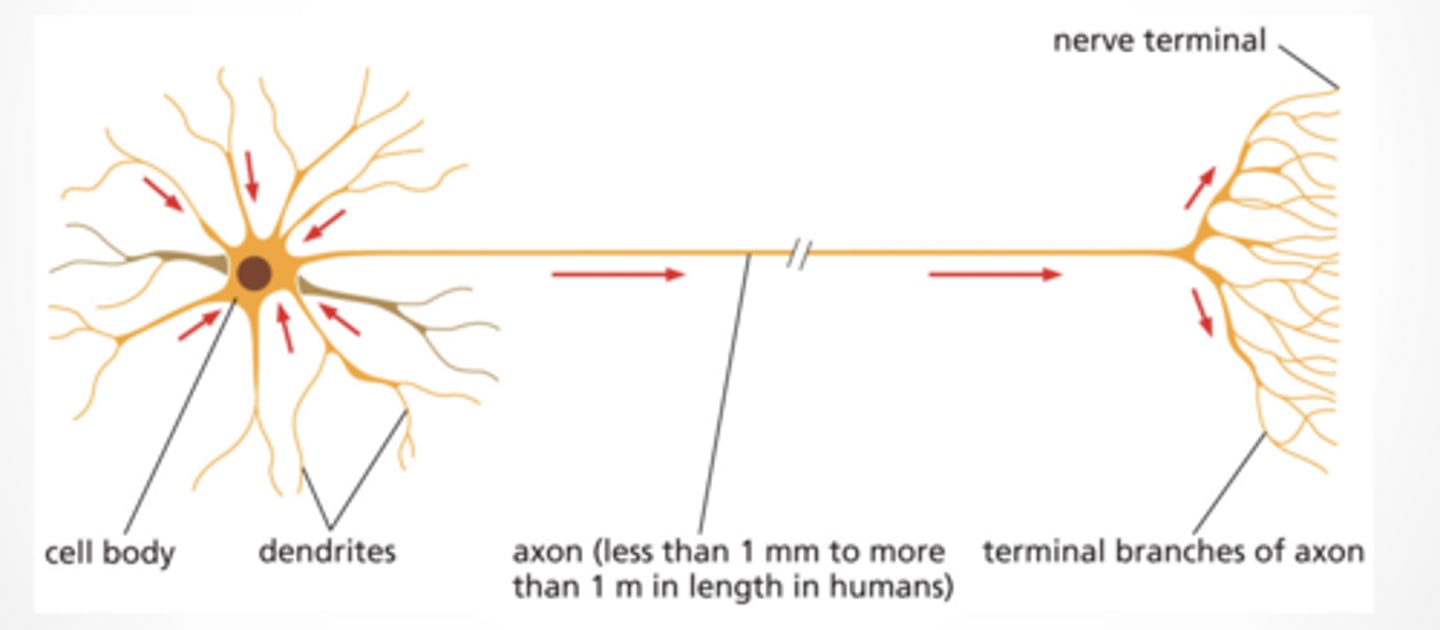
NEURONS COMMUNICATE WITH EACH OTHER THROUGH SYNAPSES
Pre neuron → axon → synapse → post neuron
Electrical signal travels down axon
Gets converted to chemical signal at synapse
Then back to electrical in next neuron
SYNAPSE IS CONNECTION FROM NEURON TO ANOTHER CELL
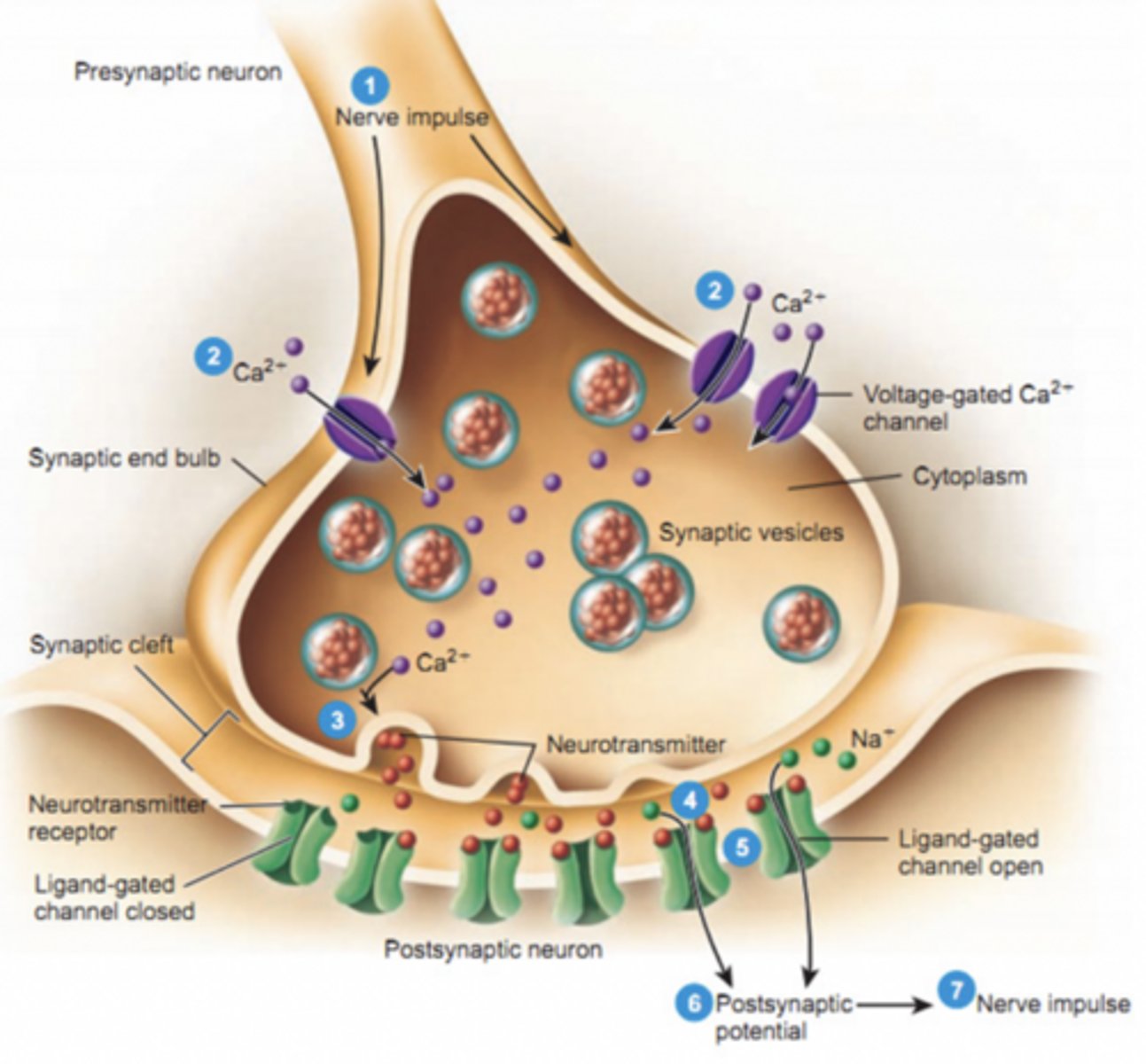
Vesicles containing neurotransmitter gather at the synapse
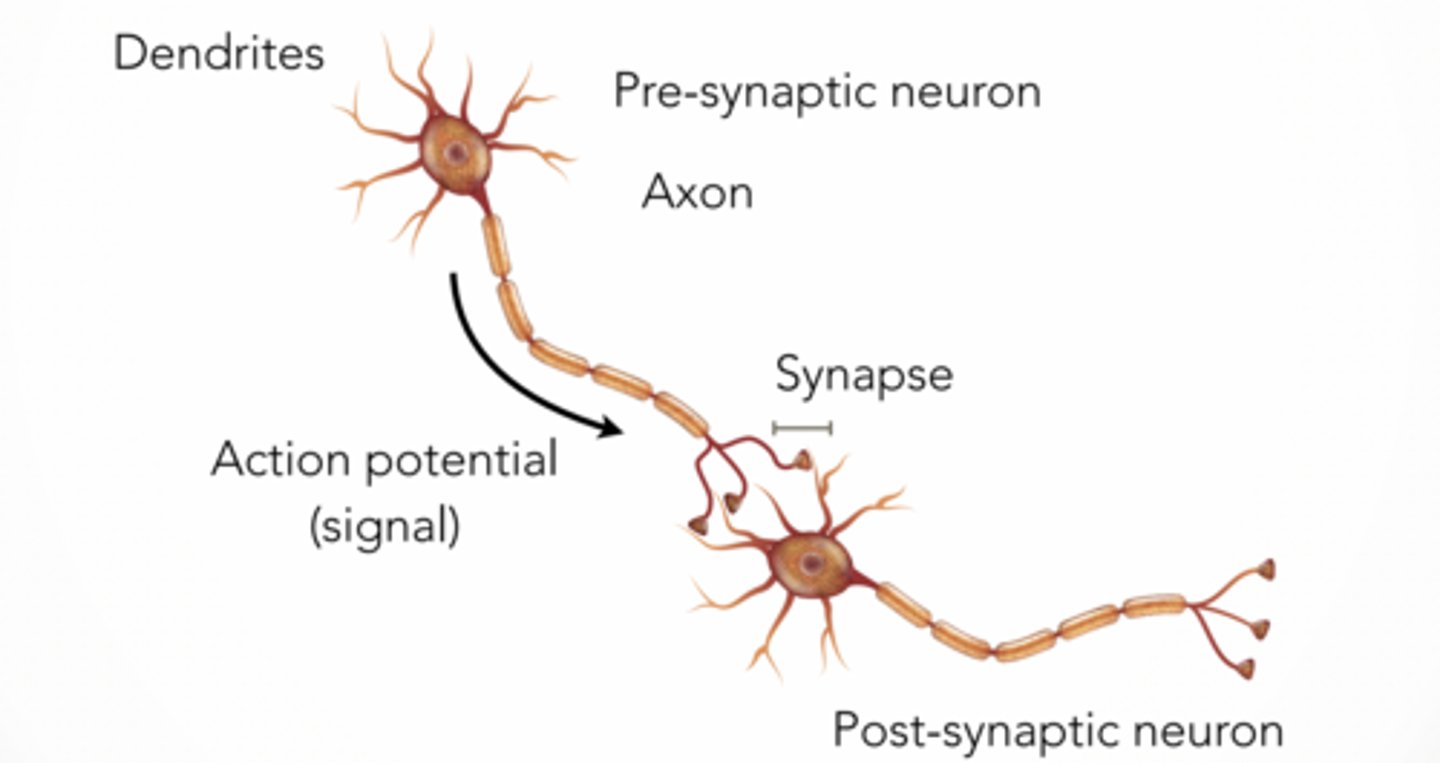
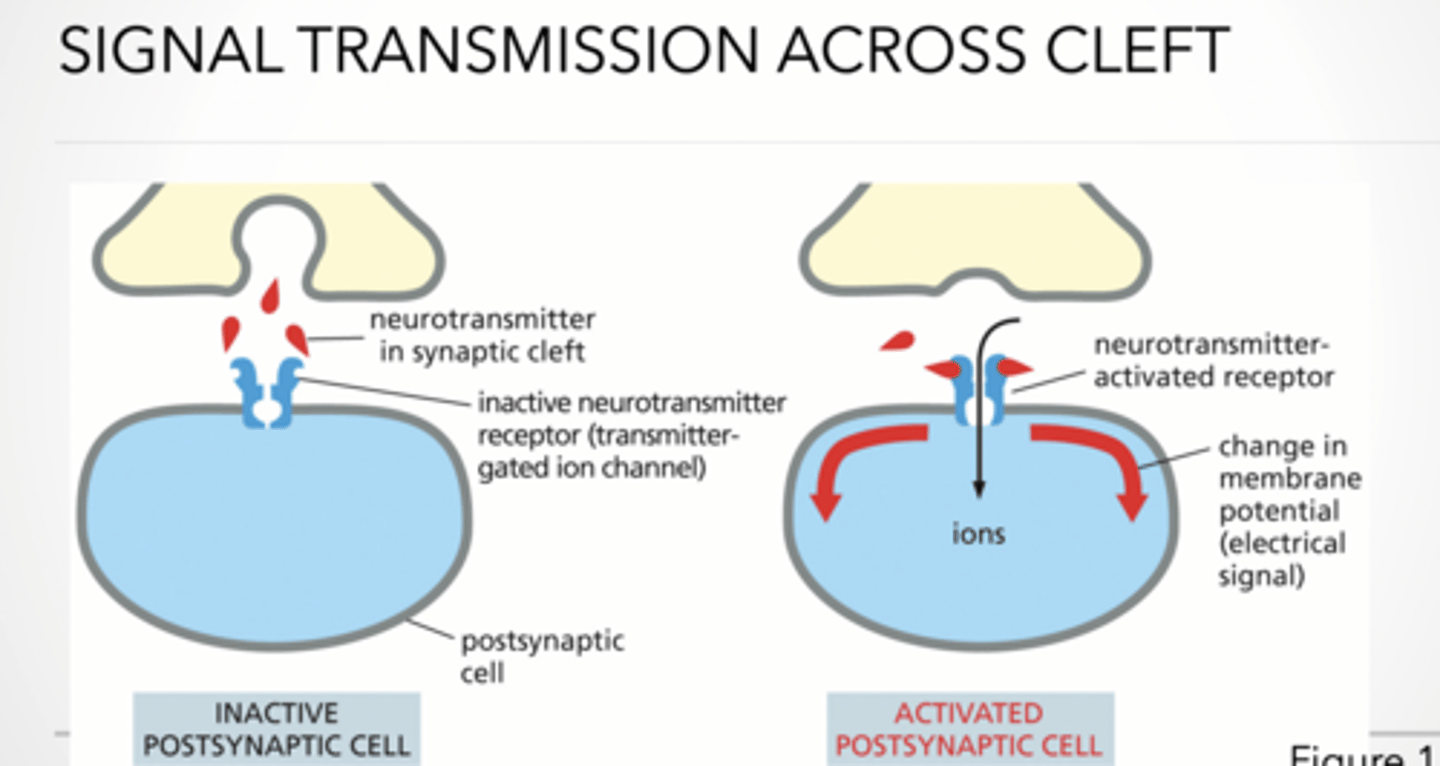
Signal transmission across cleft (rest vs activated)
Resting nerve terminal
voltage-gated Ca²⁺ channel CLOSED
Activated nerve terminal
voltage-gated Ca²⁺ channel OPEN
arriving action potential (electrical signal)
fused synaptic vesicle
released neurotransmitter (chemical signal)
Ca²⁺ = trigger for release
Feet of nerve terminal
feet
Activating a postsynaptic cell
Neurotransmitter binds receptor
activating neurotransmitter receptor
this
Opens ion channels
This causes ions to flow → Changes membrane potential → New signal generated
This is how one neuron activates the next
DOPAMINE
Dopamine released → binds receptors → signal
Transporter pulls dopamine back in (recycling)
👉This is how drugs like cocaine work (block reuptake)
Feet of this
feet
Communication between neurons
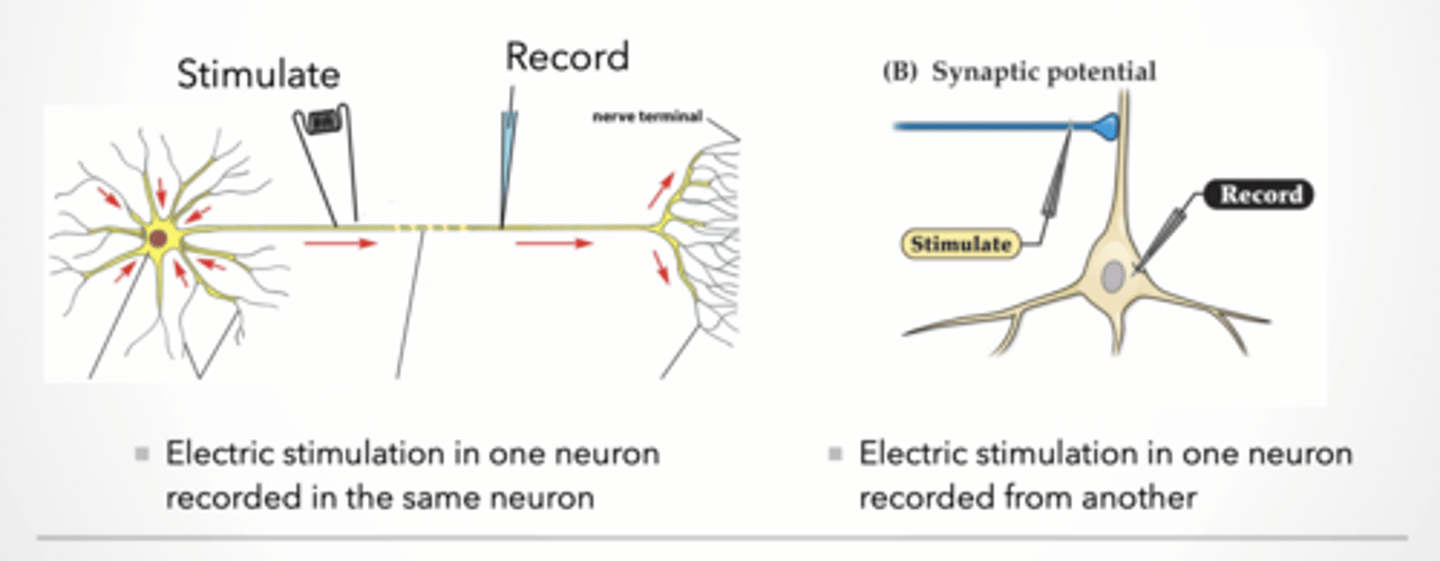
Recording communication between neurons
Electric stimulation in one neuron recorded in the same neuron
Electric stimulation in one neuron recorded from another
Explanation: Two setups:
Same neuron → measures action potential
Different neuron → measures synaptic transmission
Synaptic plasticity – cellular basis for memory
Incoming signal stays the same
Even if input is the same, output can change.
This is learning:
Stronger response = memory forming
Weaker response = forgetting
Terms:
Plasticity – ability of a neuron to change its response to a signal
LTP – long term potentiation – increased response to the same input signal
LTD – long term depression – decreased response to the same signal
Caveats:
Synaptic plasticity is not the same thing as remembering and forgetting
Memories are likely made of groups of neurons cooperating with each other
The word “RED” is not stored in one neuron
Case study intro
Dr. Marshall Westwood ate a meal of pufferfish.
felt numbness everywhere
Started throwing up
As Dr. Westwood was rushed to the hospital, he had: - trouble breathing; - signs of paralysis in his upper body and arms.
THE PHYSICIANS
• kept his airway open.
• administered drugs to bring his heart back to a normal rhythm.
• put a mixture of charcoal into his stomach to help absorb any chemicals that might still be there
he learned that he had probably been the victim of a pufferfish poisoning.
Resting potential in neurons
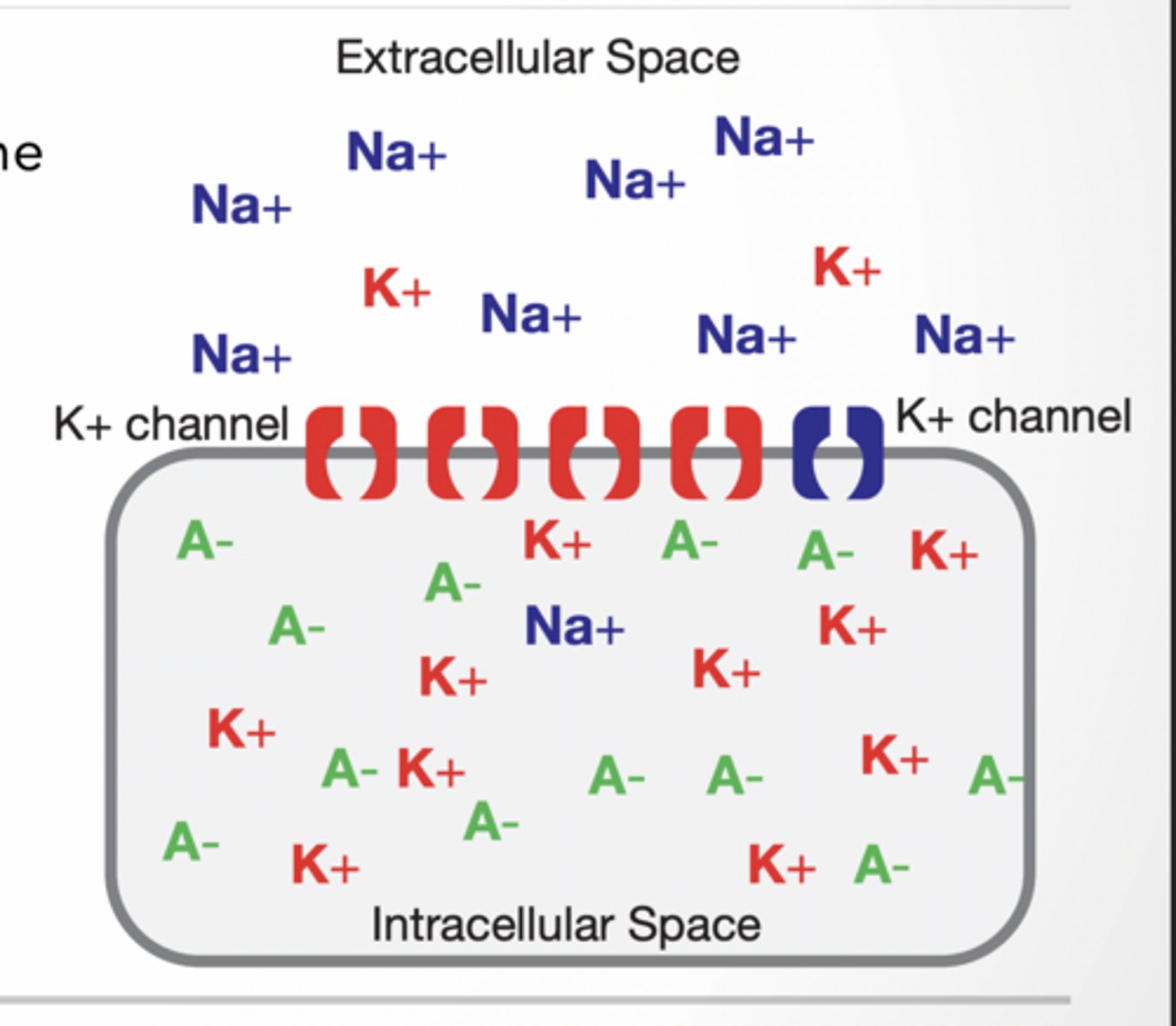
REMEMBER All cells have an electrical potential difference across their plasma membrane. - Called a MEMBRANE POTENTIAL
• The cell's inside is negative relative to the outside.
• Sodium (Na+) potassium (K+) and large anions (A-) are important in maintaining the membrane potential.
Diffusion =high to low (Na+ diffuses into cell bc conc. is high outside)
Nerst equation (finally math ) RESTING POTENTIAL OF ONE ION
V = (RT/zF)*ln (Co/Ci) Co/Ci = concentration outside / inside
RESTING POTENTIAL IN NEURONS
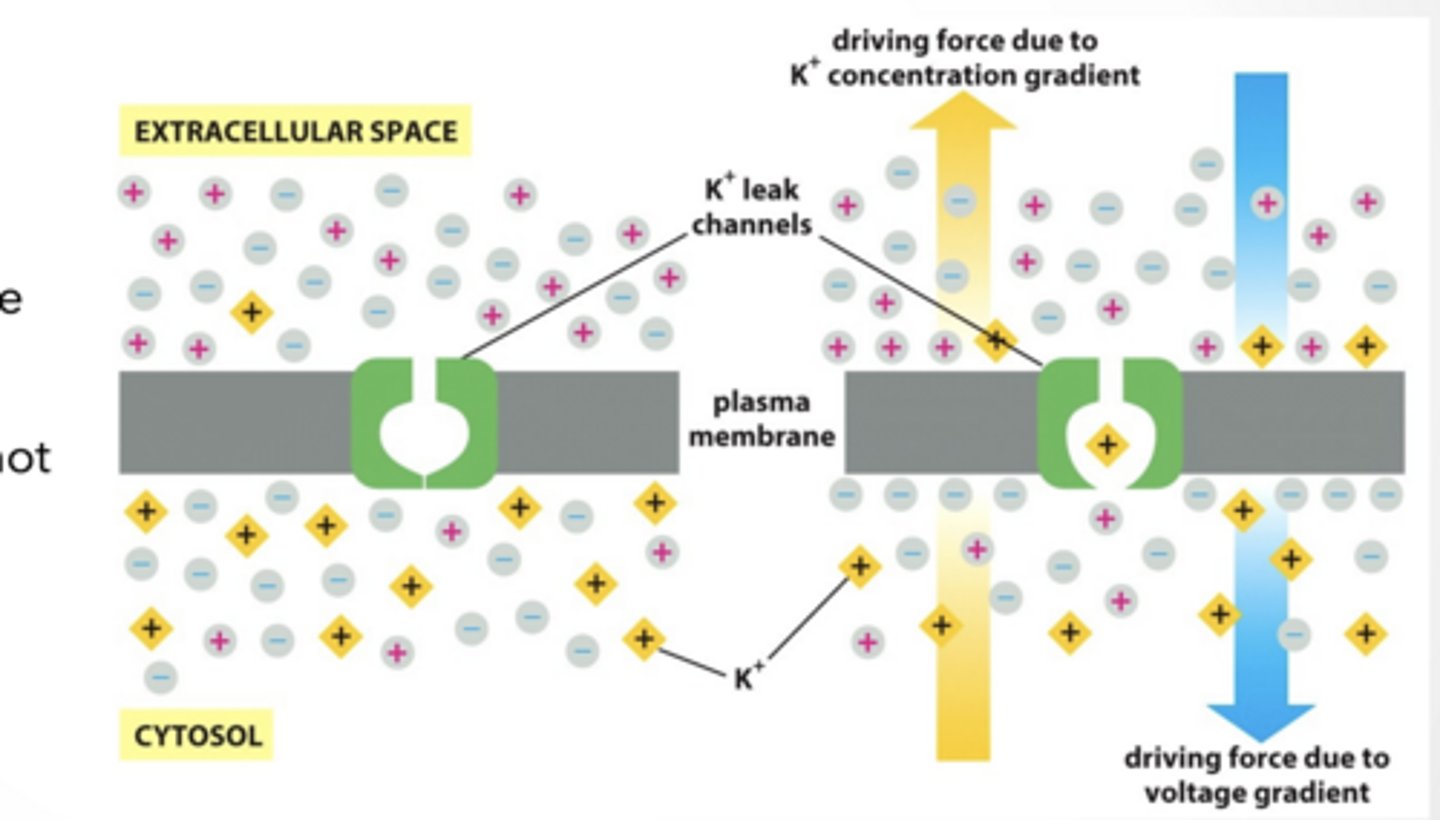
Diffusion of K+ (and less Na+) leads to a separation of charges across the membrane, and the resting potential. - Remember: MANY K+ and very few Na+ channels, membrane permeability is 100x for K+ than Na+. - Movement of K+ increases the positive charge outside the membrane relative to the inside.
Thus an ELECTRICAL gradient is formed that can also influence ion flow.
Mainting concentration gradient
RESTING POTENTIAL IS WHEN ION FLOW IS IN BALANCE WITH DRIVING VOLTAGE GRADIENT
How does the cell maintain the K+ driving force (aka concentration) if K+ is always leaking out
ACTIVE TRANSPORT
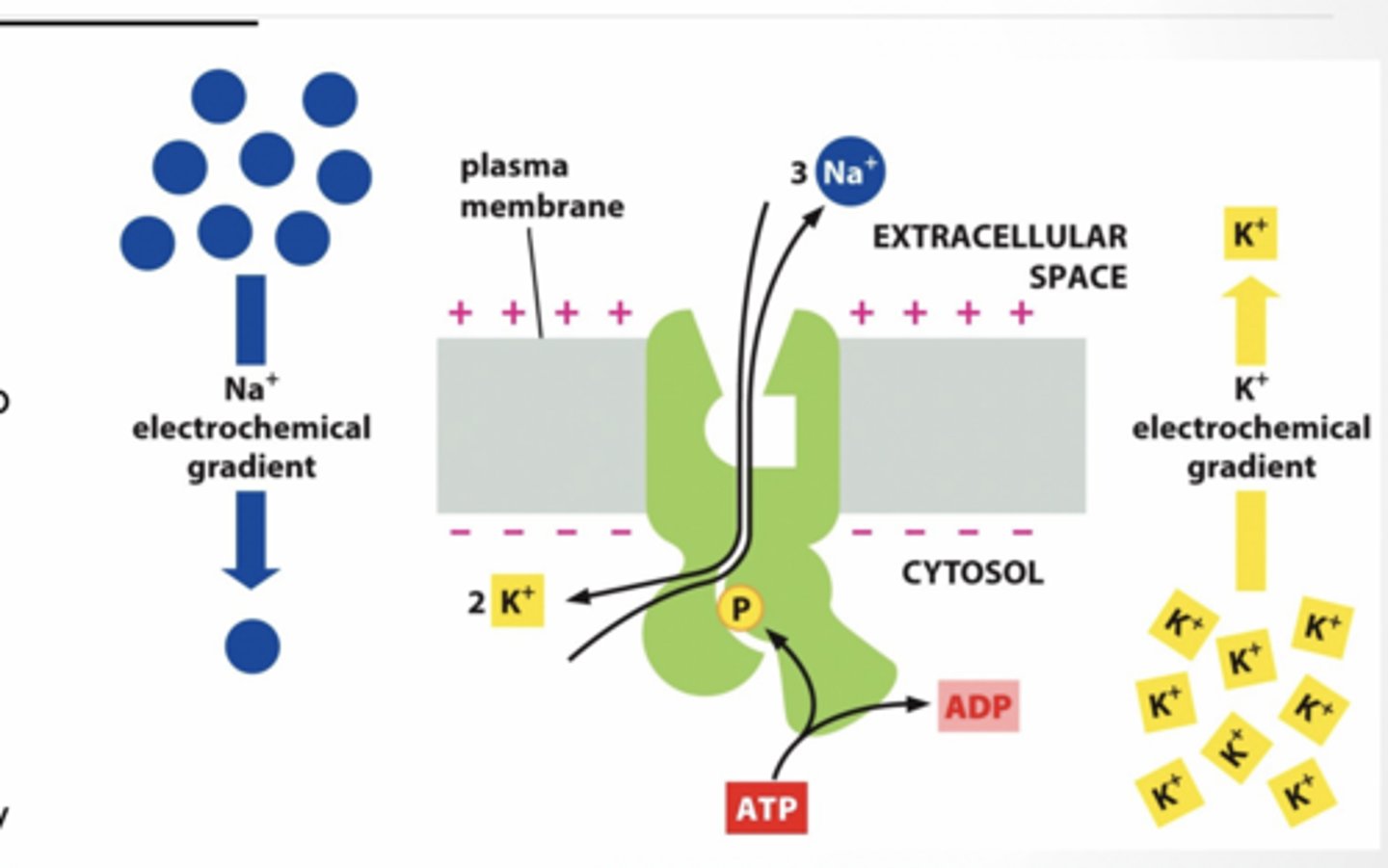
3 Na+ Out of cell 2 K+ In to cell
10-30 fold difference in concentrations from outside to inside
Relative voltage gradient established is: “+” outside and “-“ inside ATP is hydrolyzed for energy
ACTION POTENTIALS: HOW NEURONS COMMUNICATE Neurons use changes in membrane potential for fast communication- called an ACTION POTENTIAL.
• Neurons have voltage gated ion channels, that open or close in response to changes in membrane potential.
• Membrane potential may change in response to stimuli that open or close those channels
We record the wave of ions moving in an axon at a single point
Action potential
At Resting Potential, the inside of the neuron is negative relative to the outside and is thus polarized. (-70Mv).
Na+/K+ pump and K+leak channels maintain this potential
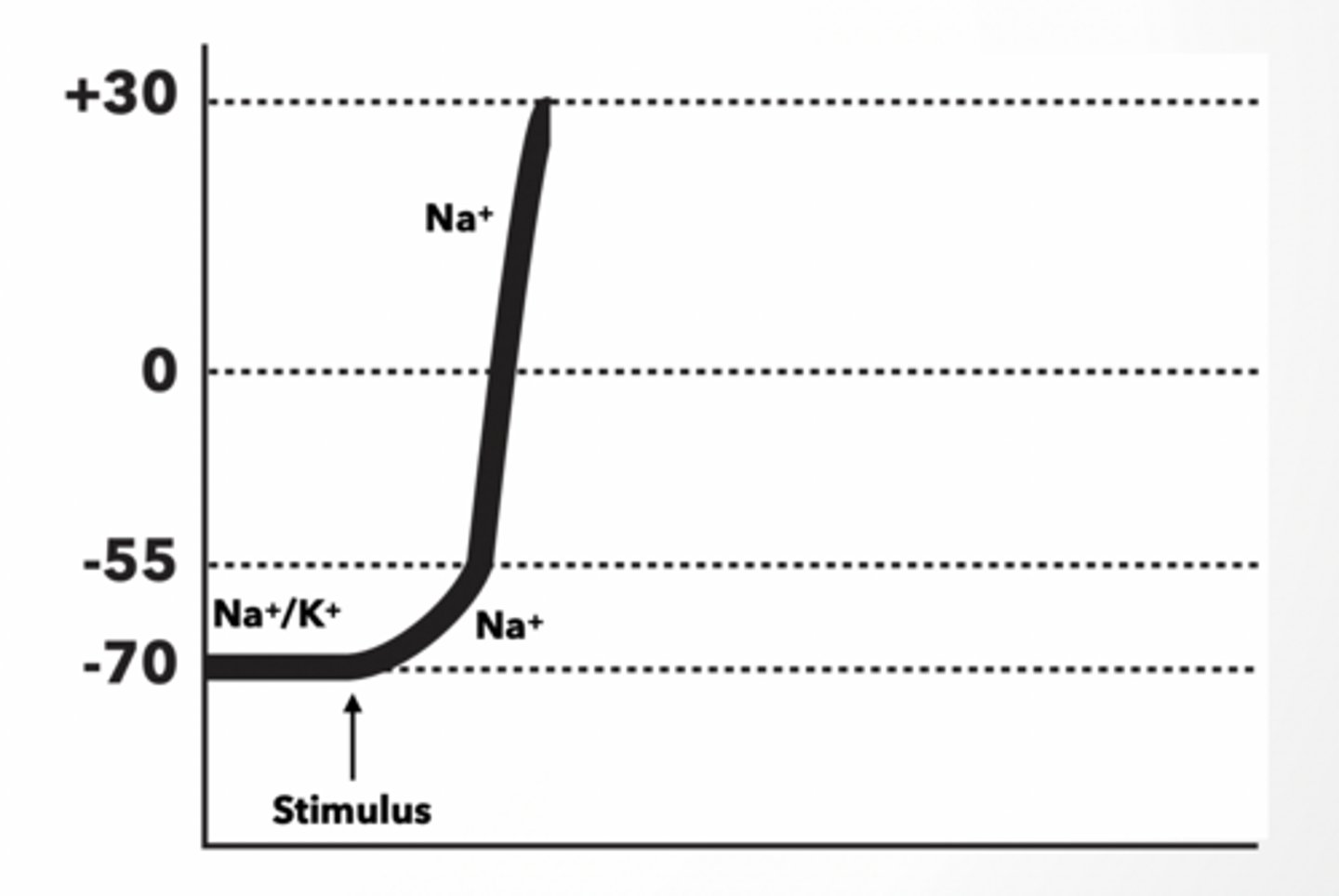
WHEN A NEURON IS STIMULATED, THE AREA OF THE MEMBRANE AT THE POINT OF STIMULATION BECOMES MORE PERMEABLE TO NA+.
If a cell starts at resting potential (-70mv), and then is stimulated:
The membrane voltage will become >-70mV because Na+ will move INTO the cell.
For an action potential (nerve firing) to occur, the cell membrane potential must reach a threshold value of ... ~ -55mV
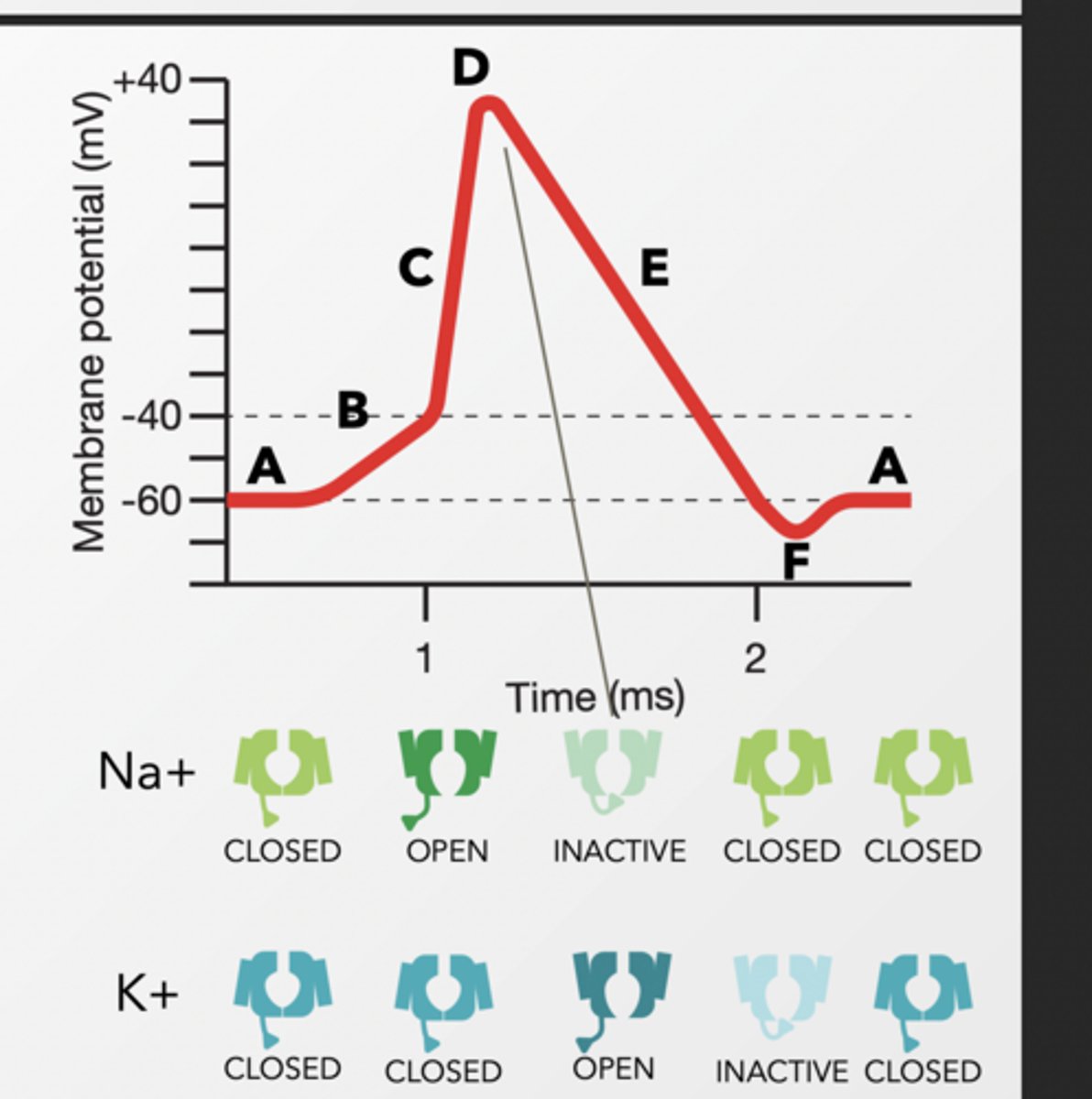
• When this threshold is reached, the voltage-gated Na+ channels open, allowing more Na+ to diffuse rapidly into the cell. - DEPOLARIZATION
REESTABLISHING RESTING POTENTIAL AFTER AN AP
AT THE PEAK OF THE ACTION POTENTIAL, NA+ VOLTAGE-GATED CHANNELS CLOSE, AND K+ VOLTAGE-GATED CHANNELS OPEN IN RESPONSE TO POSITIVE MEMBRANE POTENTIAL. TO RETURN THE CELL TO ITS NEGATIVE RESTING POTENTIAL QUICKLY:
K+ will diffuse along its concentration gradient OUT of the cell
Returns back to resting
back to the case study
Tetrodotoxin in fish is a neurotoxin - it affects nerve cells (neurons).
• Specifically, tetrodotoxin blocks voltage-gated sodium ion channels.
This can lead to Paralysis bc now there is no action potential so theres no signal to muscles
Then after recovering he touched a bird
Dr. Westwood got same symptoms but they quickly faded
It was a pitohui
he isolated the toxic compound that he was active in the feathers of the pitohui. It was a homobatrachotoxin
Batrachotoxin and homobatrachotoxin
Batrachotoxin and homobatrachotoxin are both known to act on voltage-sensitive sodium channels in excitable tissues.
THEY Prevent Na+ ion channels from closing so after depolarizing , they cant repolarize.
Na/k channels
Na⁺ channels open when voltage threshhold is met and inactivate at peak.
K+ channels do the opposite
GPCR FAST EFFECTOR RESPONSE
G proteins can effect ion channels to have immediate effect Beta/gamma interacts with K+ channel and allow K+ to flow across membrane Gi G-protein
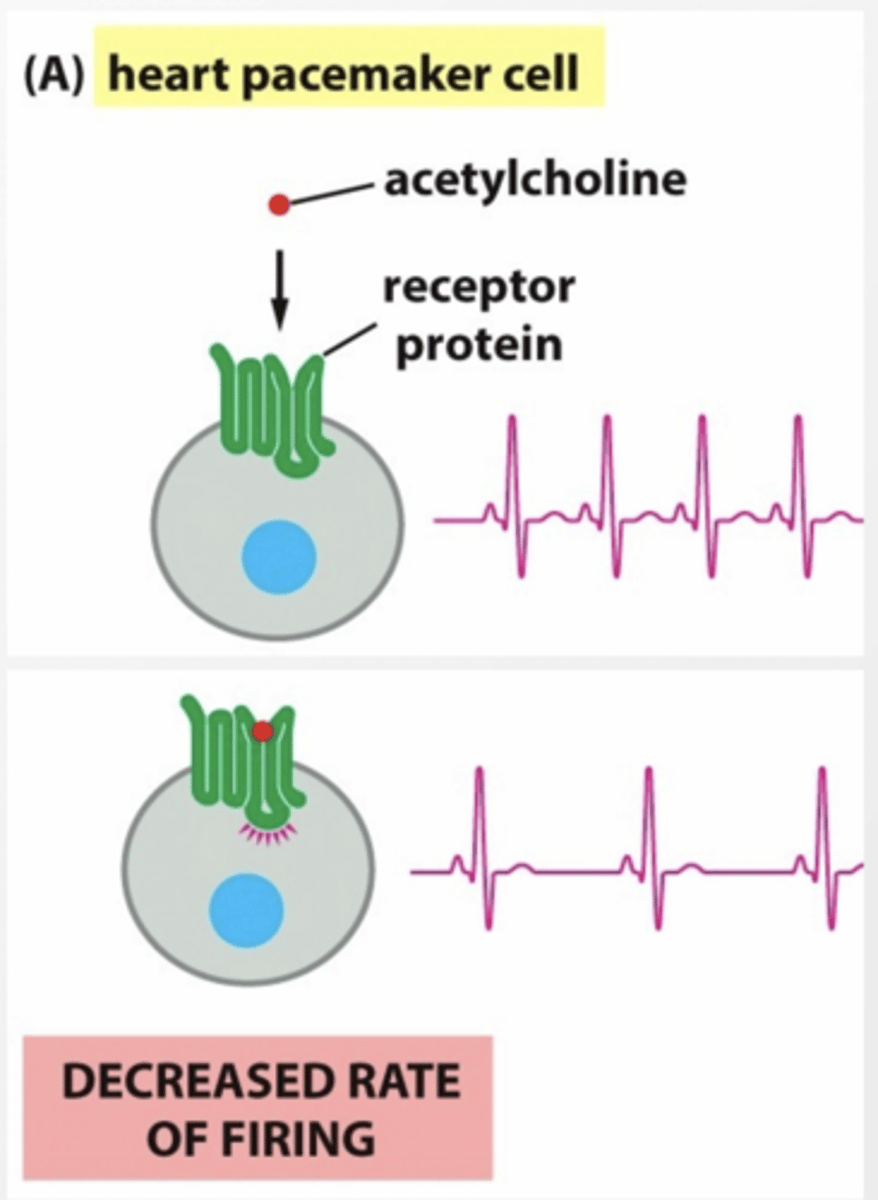
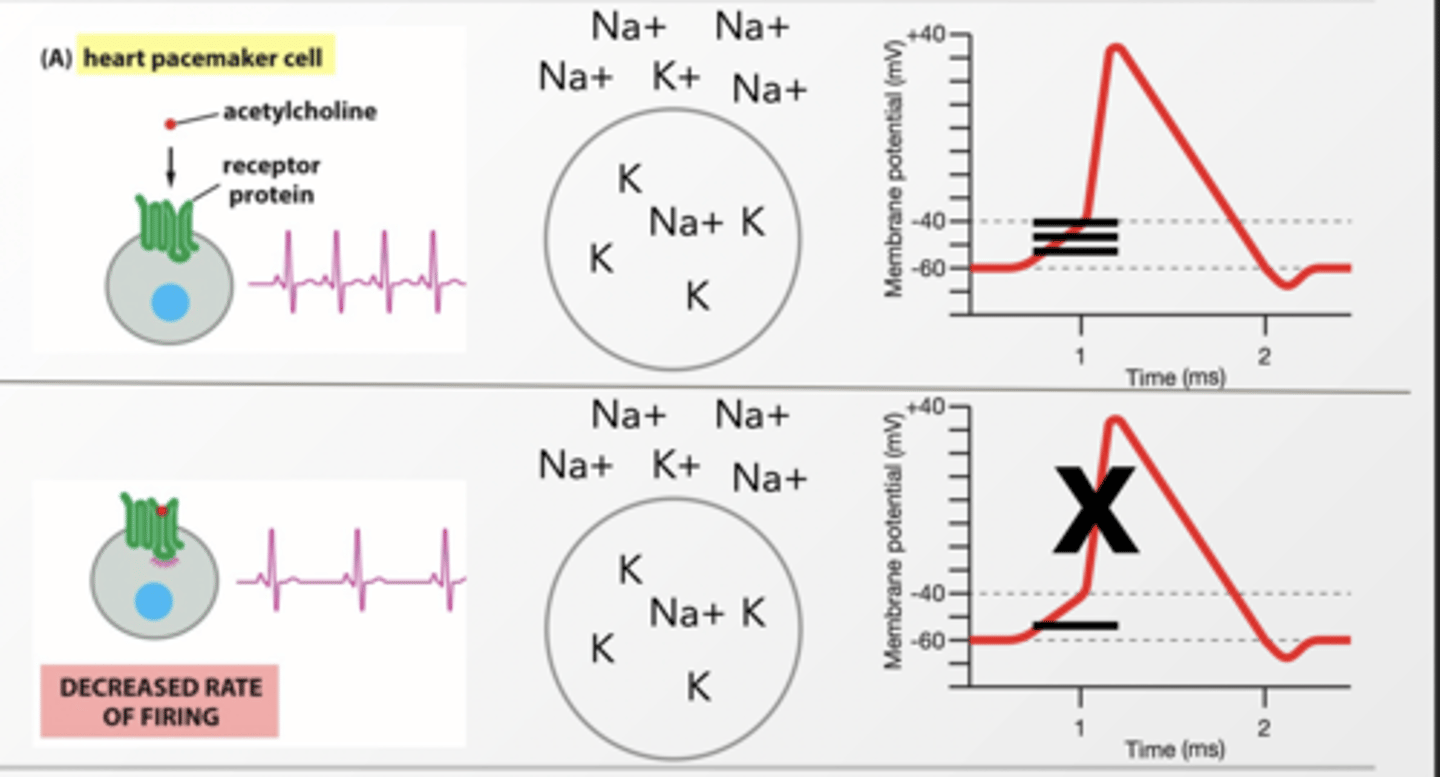
Heart pacemakers cells have a mechanism that causes action potentials to fire on a regular basis by leaking in + ions slowly.
What happens to regular pace of action potentials when K+ channels are activate by Gi linked GPCR?
K+ leaks out of the cell canceling the movement of Na+, lowering the membrane potential, and slowing the rate of action potential firing because it is harder to reach threshold
DECREASED RATE OF FIRING
Pacemaker feet
feet
better one feet
feet
Mice shhh
CHANNELRHODOPSIN
Scientists put a light-activated ion channel into neurons
Shine blue light → channel opens → Na⁺ goes IN
That causes depolarization → neuron fires
ACTIVATING NEURONS ALTERS BEHAVIOR
Parts of Neuron
Neuron parts are specialized:
Each uses different structural proteins
Different areas of the cell have different cytoskeletal elements
Soma = cell body
Nucleus • Mitochondria • Ribosomes • Etc
Cytoskeleton
Actin ,Microtubules, Neurofilaments
Neurofilaments relate to diseases like Alzheimer’s
Axon hillock
Initial segment for the axon
Cytoskeletal elements compress into axon
Free of most organelles
Due to compression of cytoskeleton limiting the space available
Axons
Only 1 axon present in each neuron
Has MTs and neurofilaments
Tau is the spacer for microtubules - determines spacing
Transport of mitochondria and other elements
No ER or Golgi
Contd
Dendrites
Receive information from other neurons
• Synapses
• More widely spaced cytoskeletal elements that allow for diffusion
• MAP2 - •
Relatively fewer neurofilaments • Thousands of connections
Material transport
Material must be transported around the cell - axons make this more difficult
• Small molecules diffuse down the axon
• Peptides must be transported down the axon
Motor proteins
Synthesis occurs in the soma
• Material is transported along Mts
• Anterograde - towards the synapse - kinesin
• Retrograde - away from the synapse - dynein
Signaling for protein synthesis
Signaling for protein synthesis
Activated PKA can be transported to the nucleus •
PKA phosphorylates transcription factor proteins
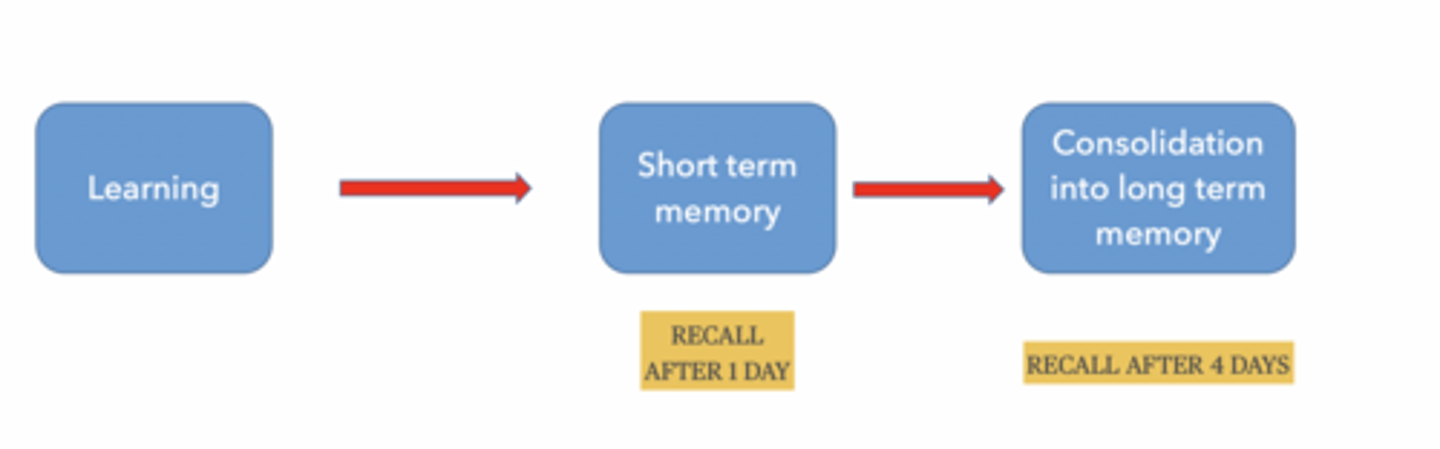
DOES MEMORY REQUIRE PROTEIN SYNTHESIS
Short-term memory → NO
Long-term memory → YES
Memory happens in stages:
Learn something
Short-term memory (temporary)
Consolidation → long-term memory
Behavioral testing for memory
Train an animal to have a response to a certain set of conditions.
Limitations in model animals
Response repertoire
Size and repeatability of response
Fear conditioning
Strong repeatable responses
Variable strength of memory
Present and measurable with non-verbal behavior in many model systems
Zebrafish - twitching
Mouse - freezing and jumping
Fish experiments
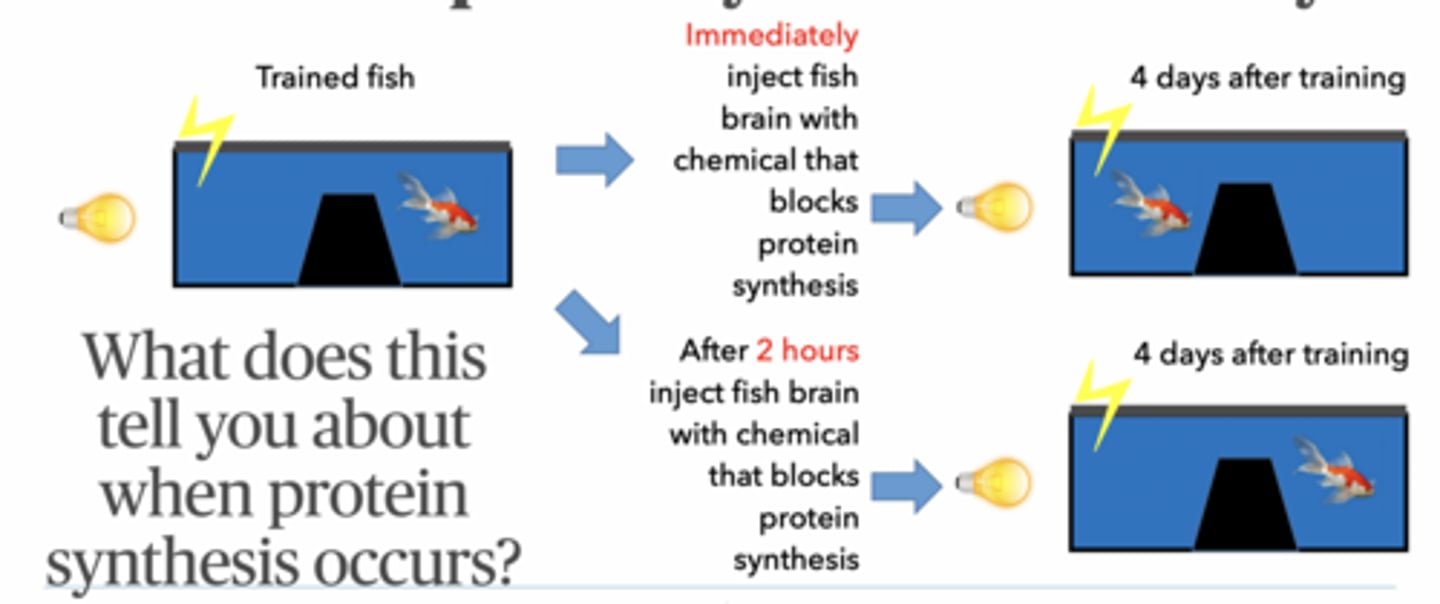
Design a test that would determine if protein synthesis is required for different memory types
Block protein synthesis
Test memory later
Memory works at 1 day → short-term ok
Memory fails at 4 days → long-term needs proteins
Classic conditioning:
Light → shock
Fish learns: light = danger
Test memory after time
If fish moves away when light turns on: → memory exists
Short-term memory:
No protein synthesis
lasts hours-day
Long-term memory:
Requires protein synthesis
requires gene transcription
lasts days+
Autophagy vs proteasome
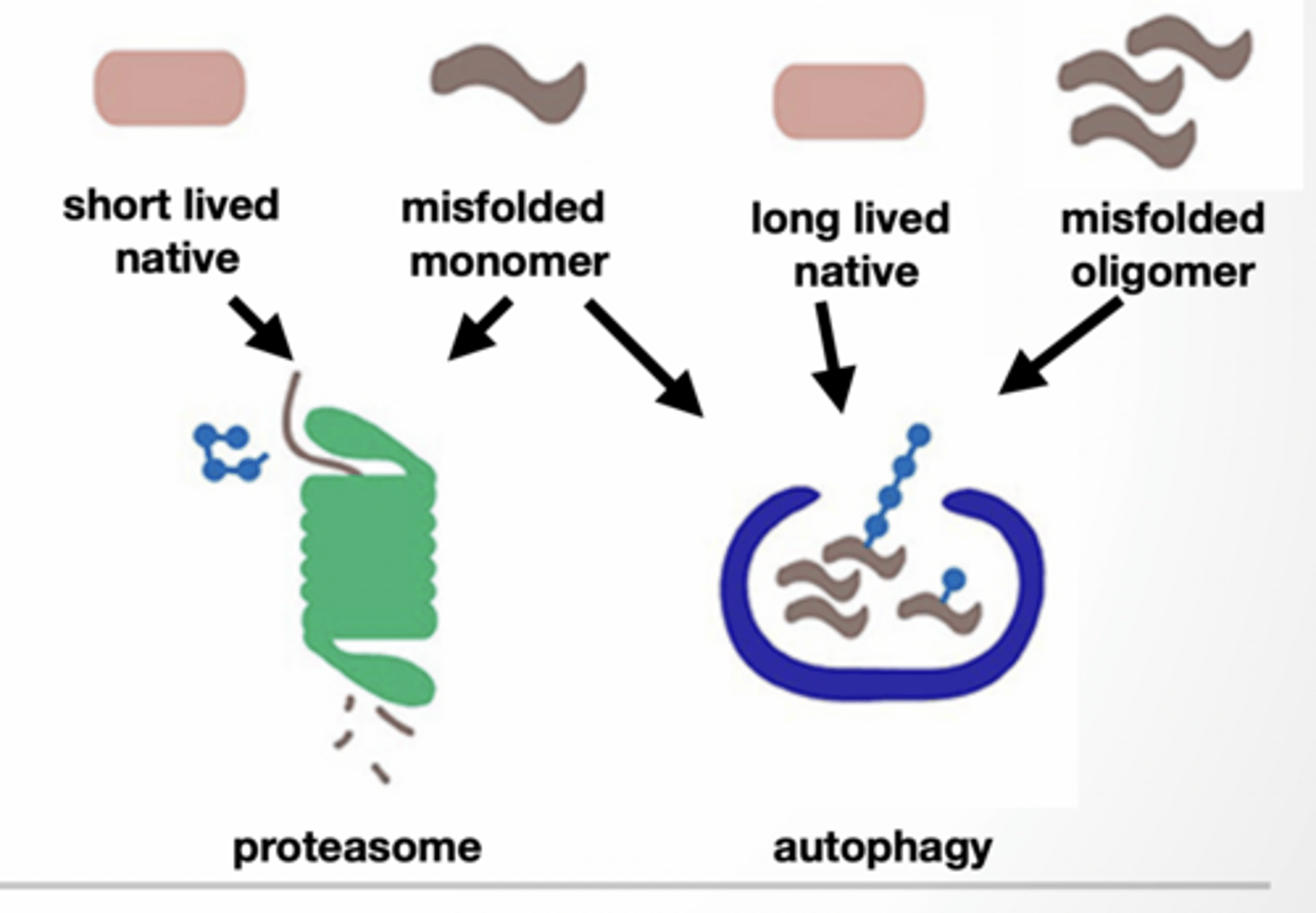
PROTEIN NEEDS TO BE REMOVED DUE TO DAMAGE OR REGULATION OF ACTIVITY
Proteasome - direct degradation
Short lived proteins misfolded monomers
Autophagy - delivery to lysosome Long lived proteins misfolded oligomers/monomers
Both are tagged with ubiquitin
Autophagy is for
nutrient and energy homeostasis
removal of damaged organelles
removal of aggregate proteins
removal of intracellular pathogens
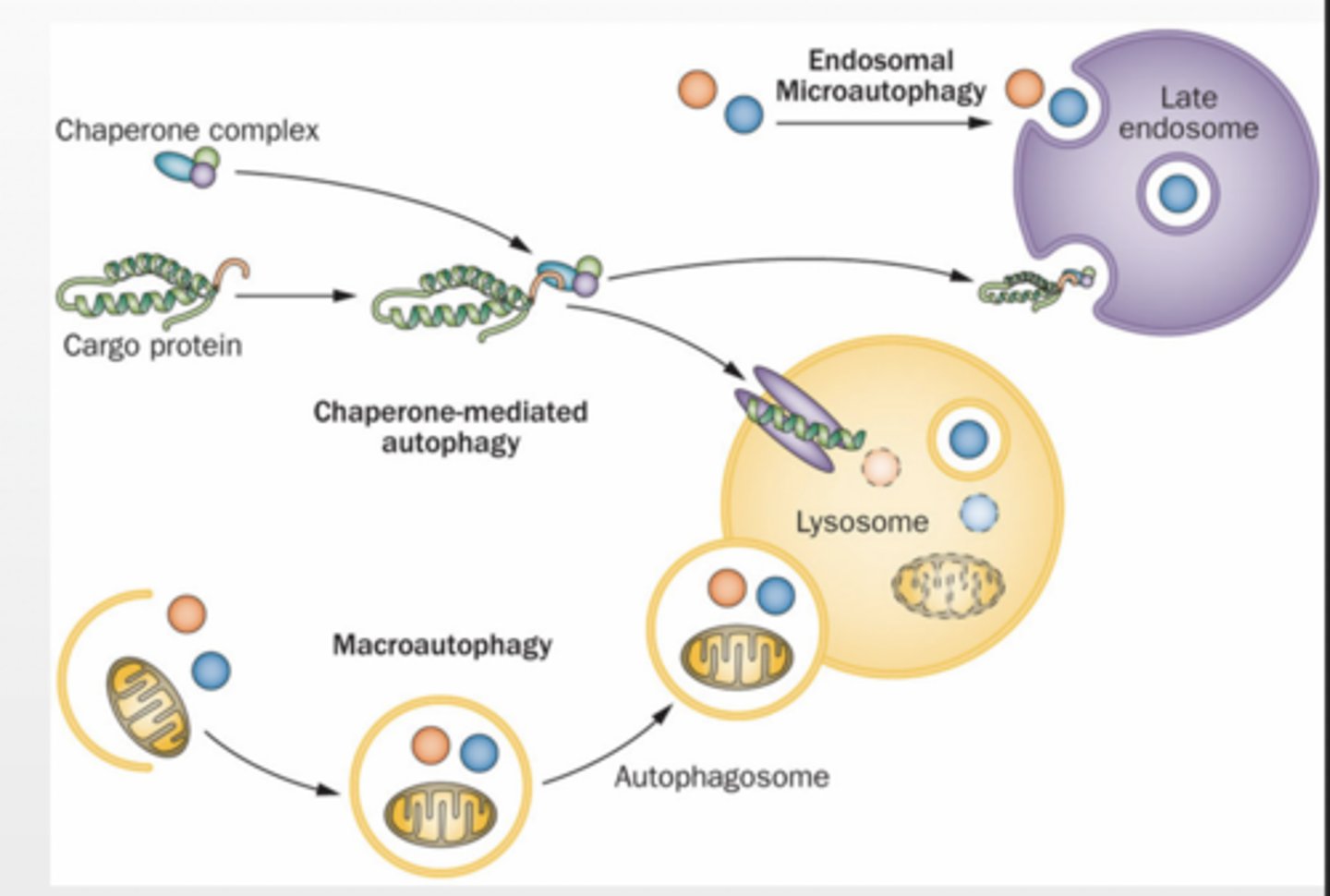
Macroautophagy
molecules are enveloped by a membrane to form autophagosome
membrane fuses with lysosome
Microautophagy small debris are sequestered into lysosome
Chaperone Mediated Autophagy (CMA) proteins are transported through a channel
3 types feet
feet
feet of process
feet
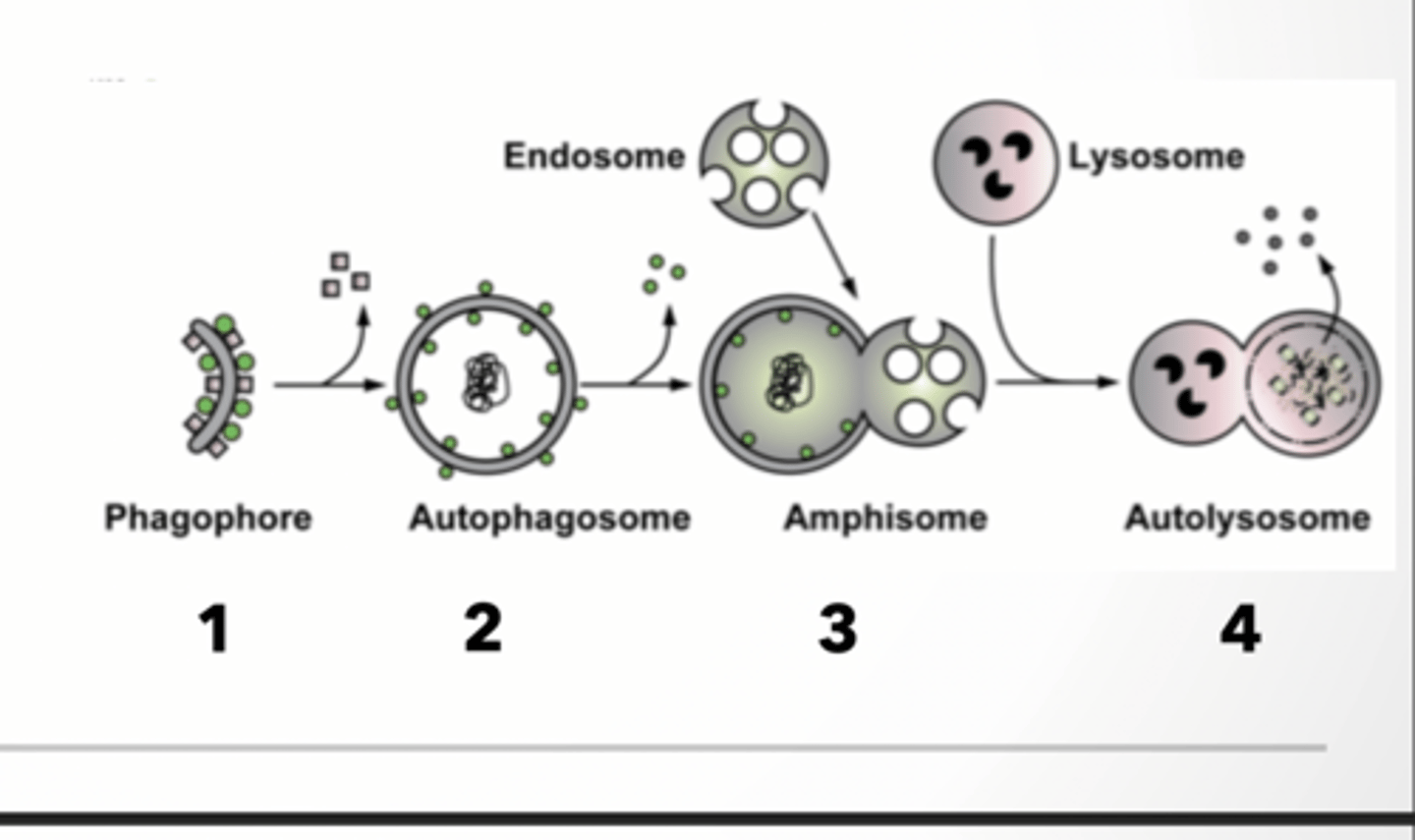
PROCESS OF AUTOPHAGY and Phagophore formation
1. Phagophore or isolation membrane forms from membrane 2. Cargo is brought to autophagosome
3. Autophagosome fuses with endosome (amphisome)
4. Amphisome fuses with lysosome (autolysosome)
PHAGOPHORE FORMATION
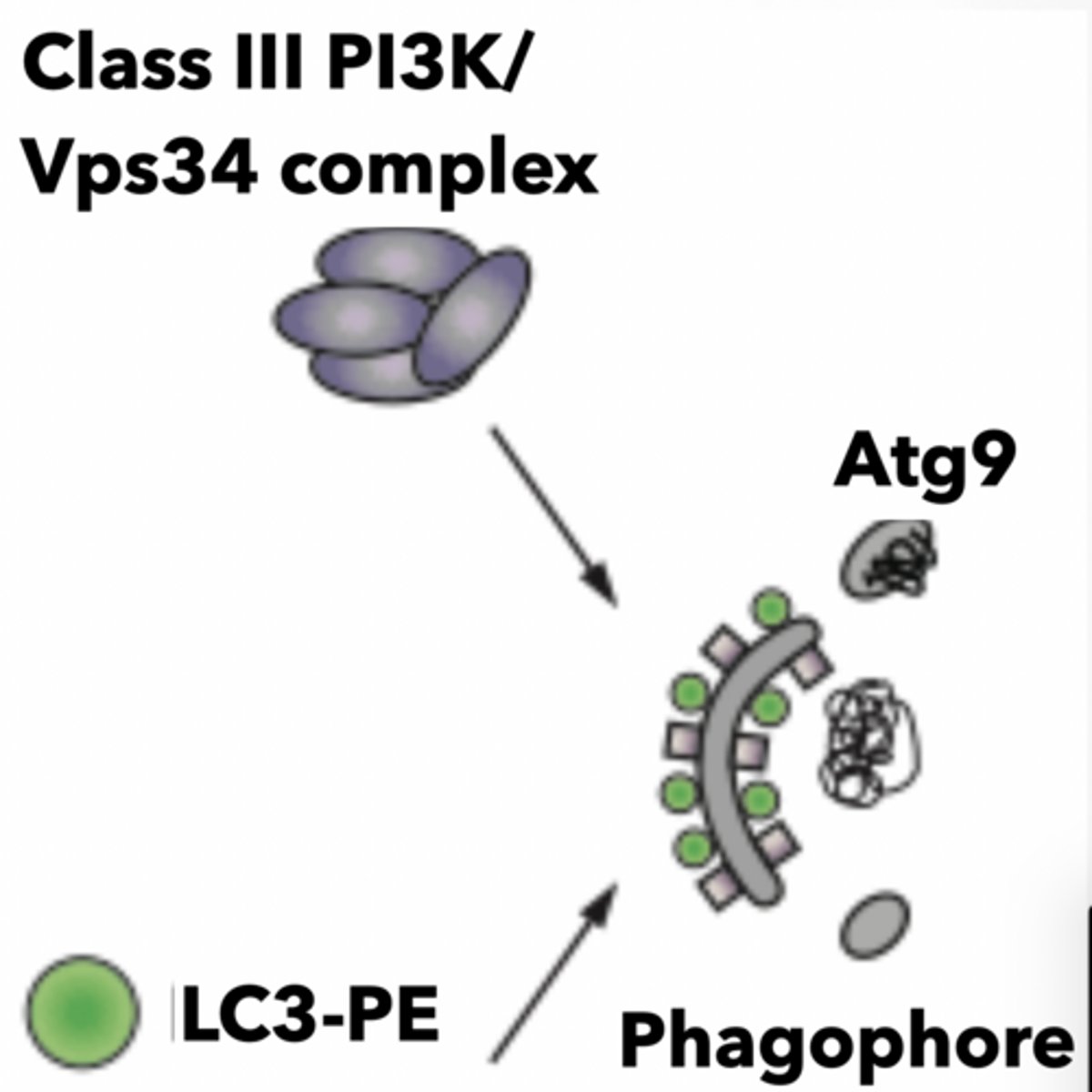
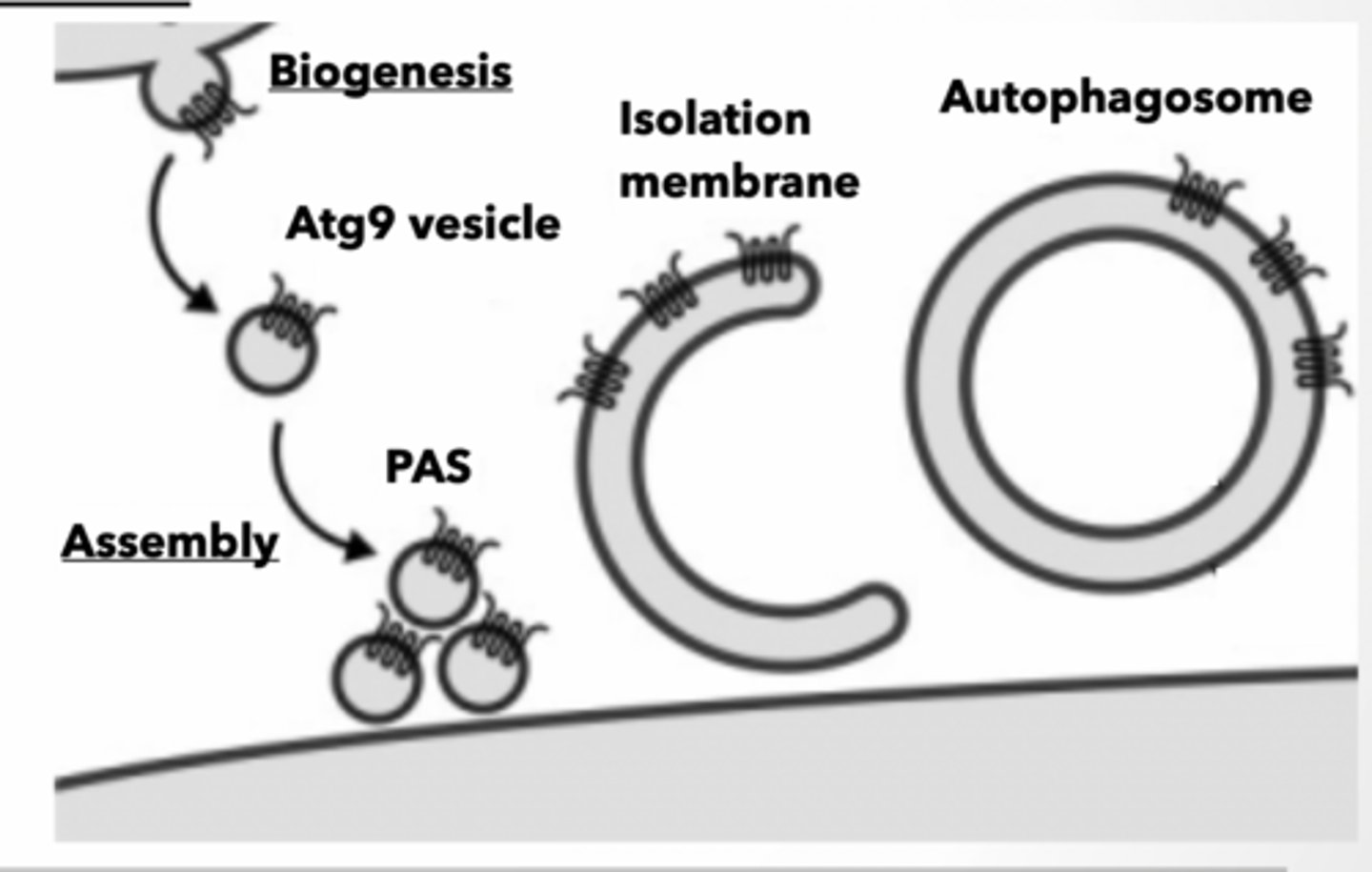
All autophagosomes start as an isolation membrane (IM) could come from Golgi, endosomes, ER, mitochondria associate membrane (MAM) interface
Complex mechanisms enable formation of autophagic vesicle
PI3P
VPS34 complex
ATG9 - membrane receptor
LC3 - autophagy receptor
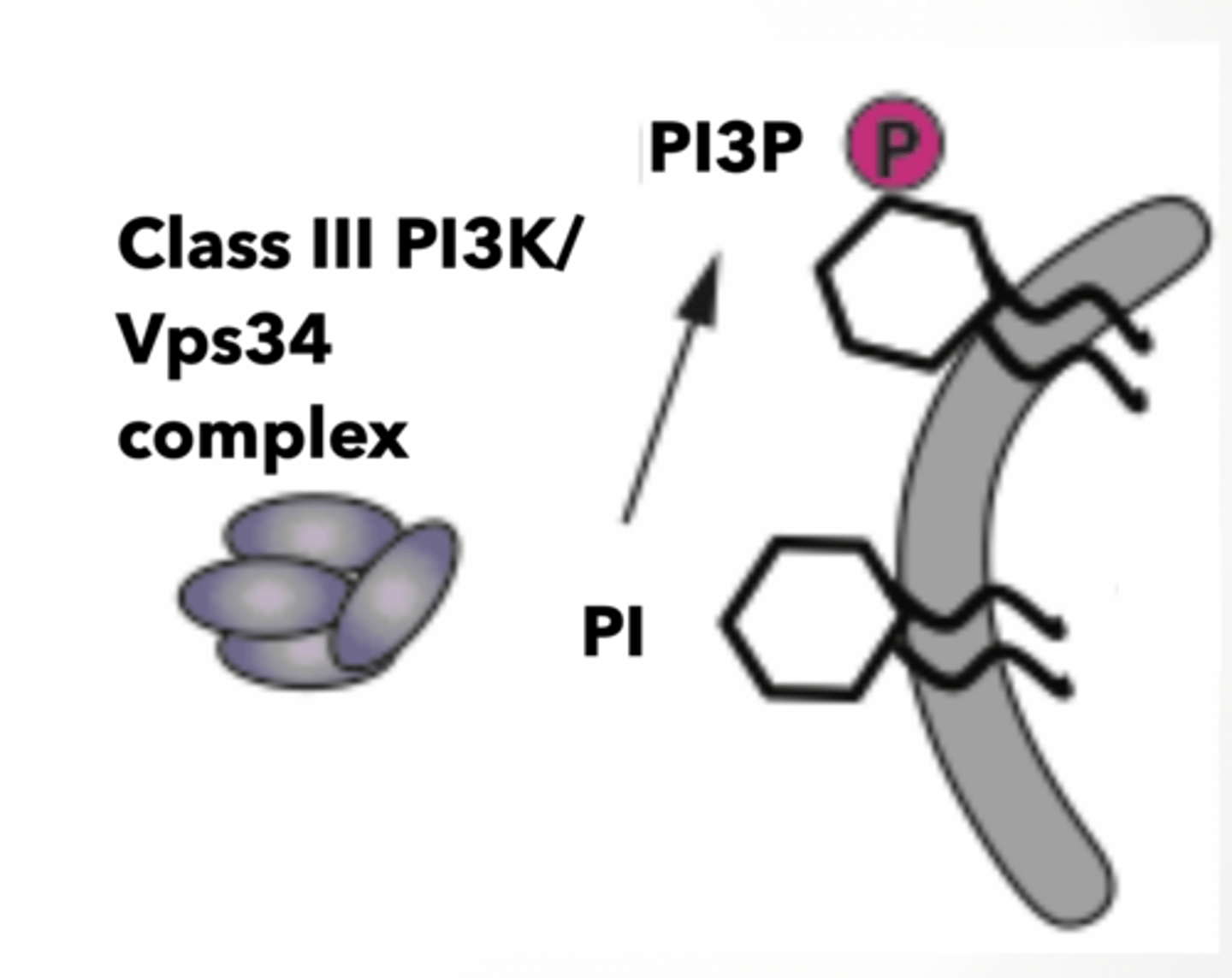
Vps34 is phosphotidylinositol kinase (PI3K)
each version of PIP has a different kinase
Turns PI to PI3P PI3P is sensed by other proteins
Feet of this bs
feet
Feet of Vps
feet
Autophagosome formation and LC3 - II
ATG9 containing vesicles bud from source membrane Vesicles assemble into preautophagosomal structure (PAS) PAS forms isolation membrane
ATG9 senses PI3P to bring new membrane to forming autophagosomes
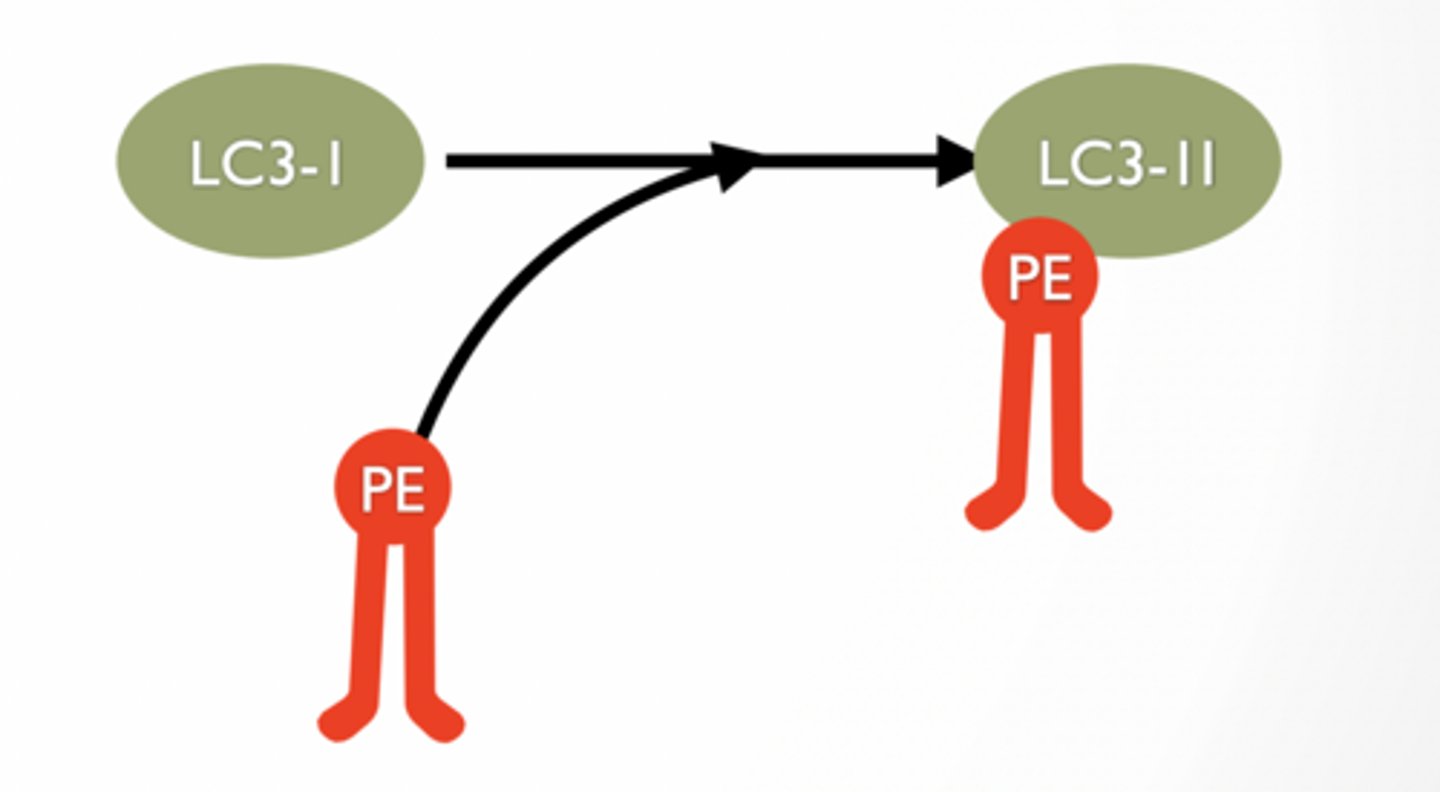
LC3-II IS A PHOSPHOLIPID TAG FOUND ONLY IN AUTOPHAGOSOMES
LC3-II is lipidated form of LC3-I
lipidation is when PE is added to LC3
LC3-II is a molecule used to “tag” PE
Found only in autophagosome
PE part of LC3-II can be inserted to membranes to allow ‘tag’ to be associated with membrane
Autophagy regulation
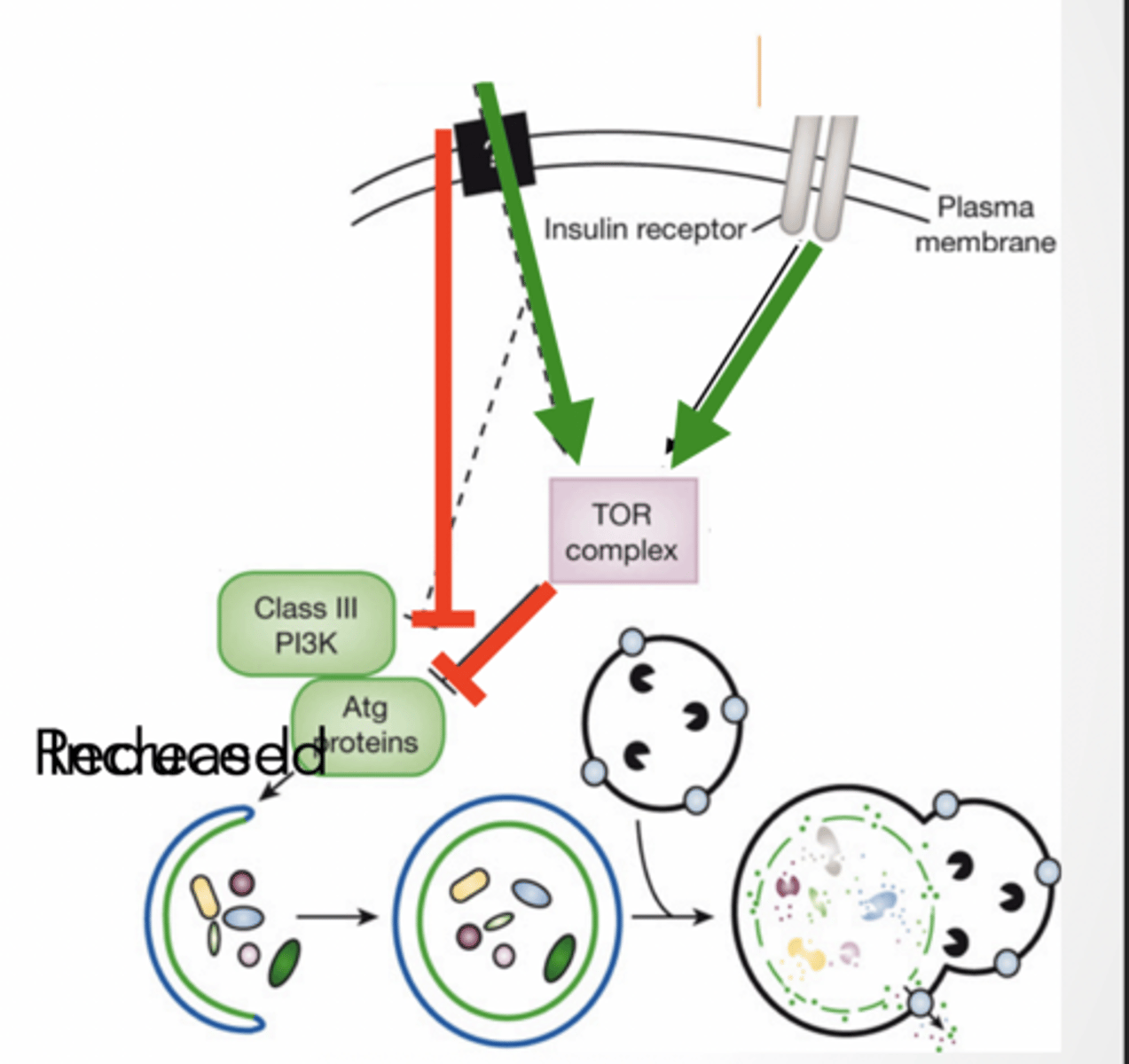
Starvation induced autophagy
TOR complex inhibits autophagy
anything that inhibits TOR will activate autophagy and vice versa
insulin and amino acids activate TOR Class III PI3K (kinase) activates autophagy
ATG proteins - autophagy related work with kinase to form protein complexes
Feet of LC3
feet
Feet of regulation
feet
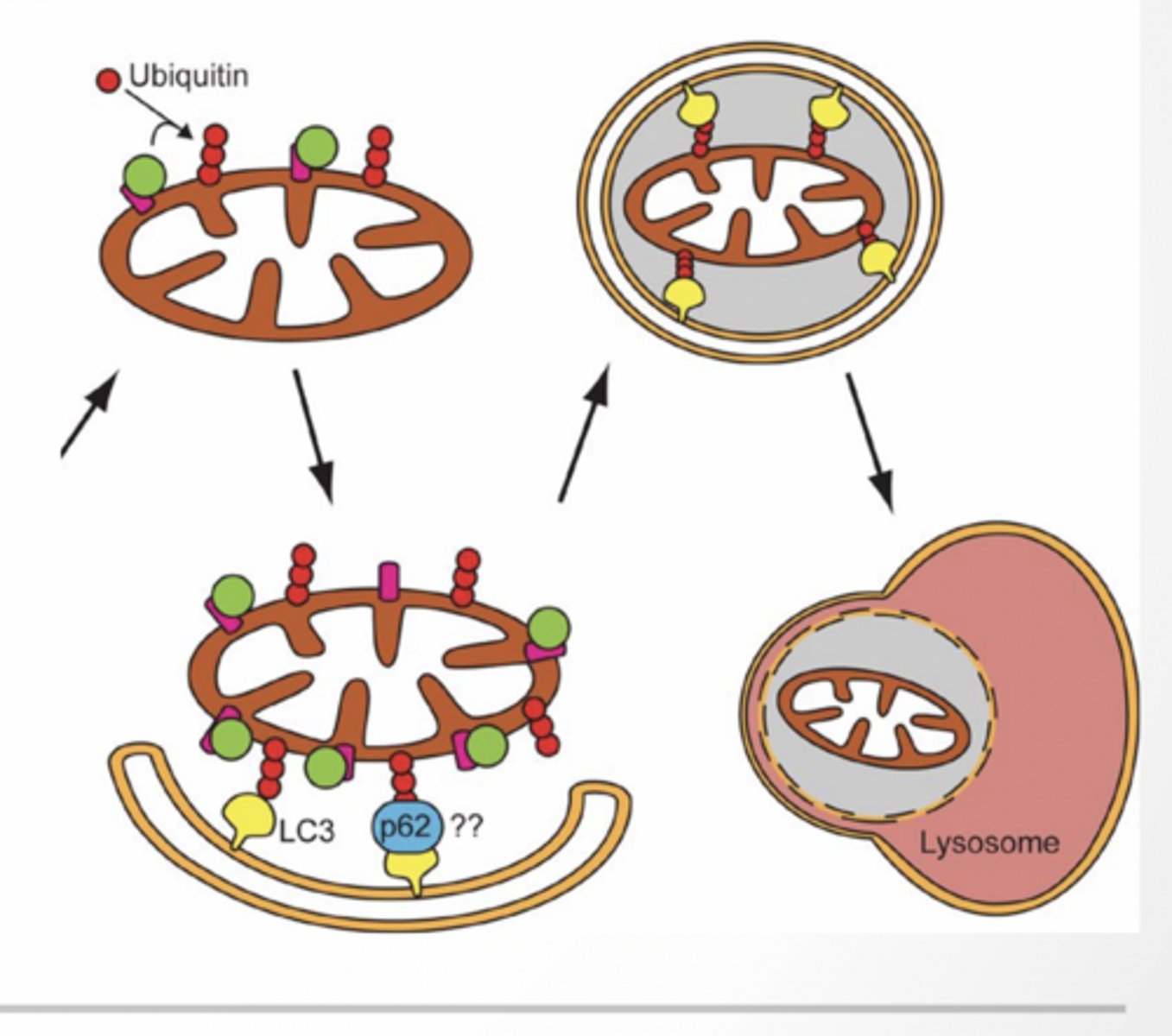
Mitophagy
CARGO IS LOADED INTO PHAGOPHORE AND BECOMES THE ISOLATION MEMBRANE
MITOPHAGY
Damaged mitochondria ubiquinated
Ubiquitin recognizes LC3II (potentially using an adaptor protein P63)
Mitochondria is loaded into the auotphagosome
Pros
Aging - removes damaged organelles
Neurodegeneration - prevents accumulation of aggregate proteins
Infection - this is one of the way that cells get rid of viruses and bacteria that get inside
Cancer - removes damaged organelles
cons
Neurodegeneration - accumulation of autophagic vesicles may be involved in AD
Infection - some bacteria have evolved to subvert the system for their lifestyle
Cancer - can help cancer cells resist cancer drugs and survive with low nutrients
Extracellular matrix
EXTRACELLULAR MATRIX
Supports and anchors cells
Cell communication
Composition:
Mix of proteins and polysaccharides
Secreted from cells via exocytosis
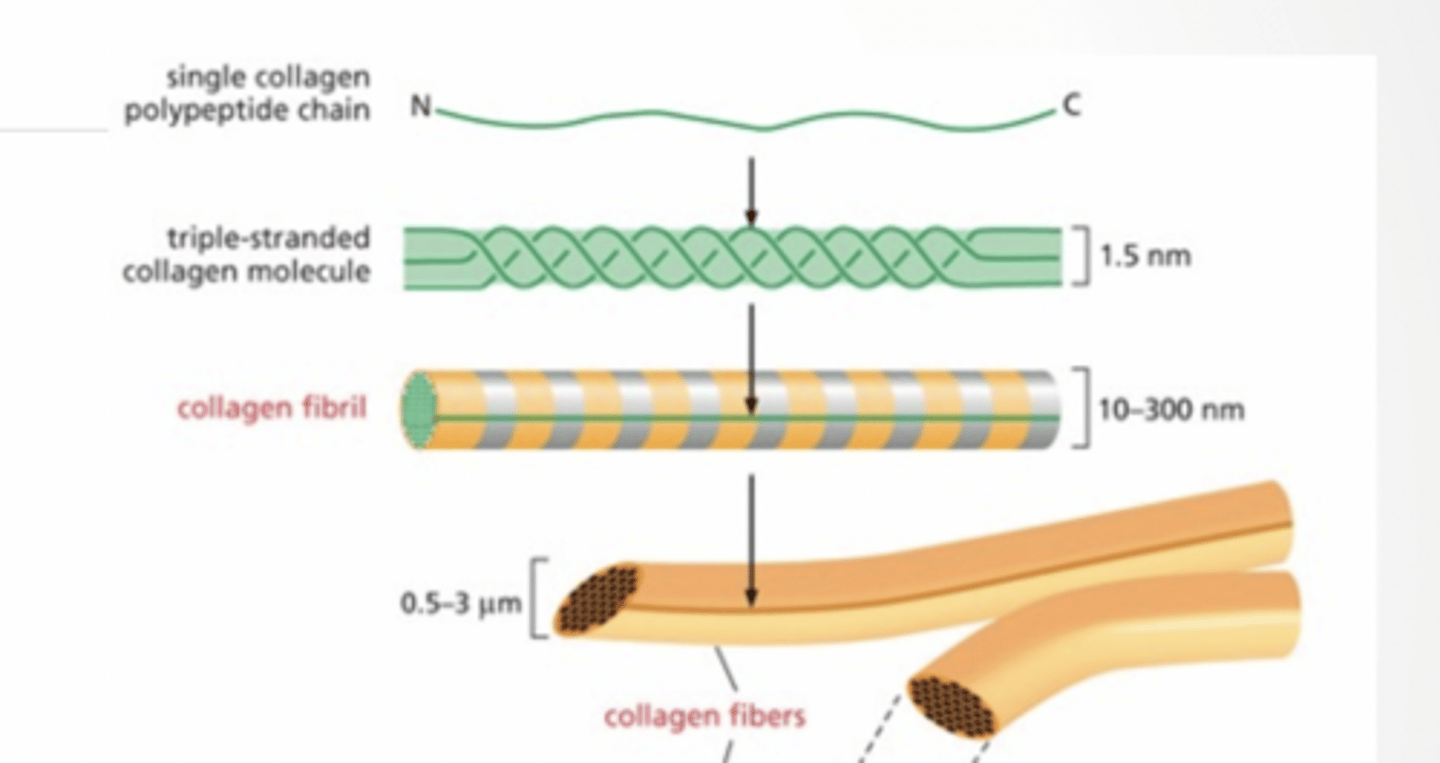
COLLAGEN
Single protein chain
Becomes triple strand
Strands assemble into fiber
Fibers assemble into bundles
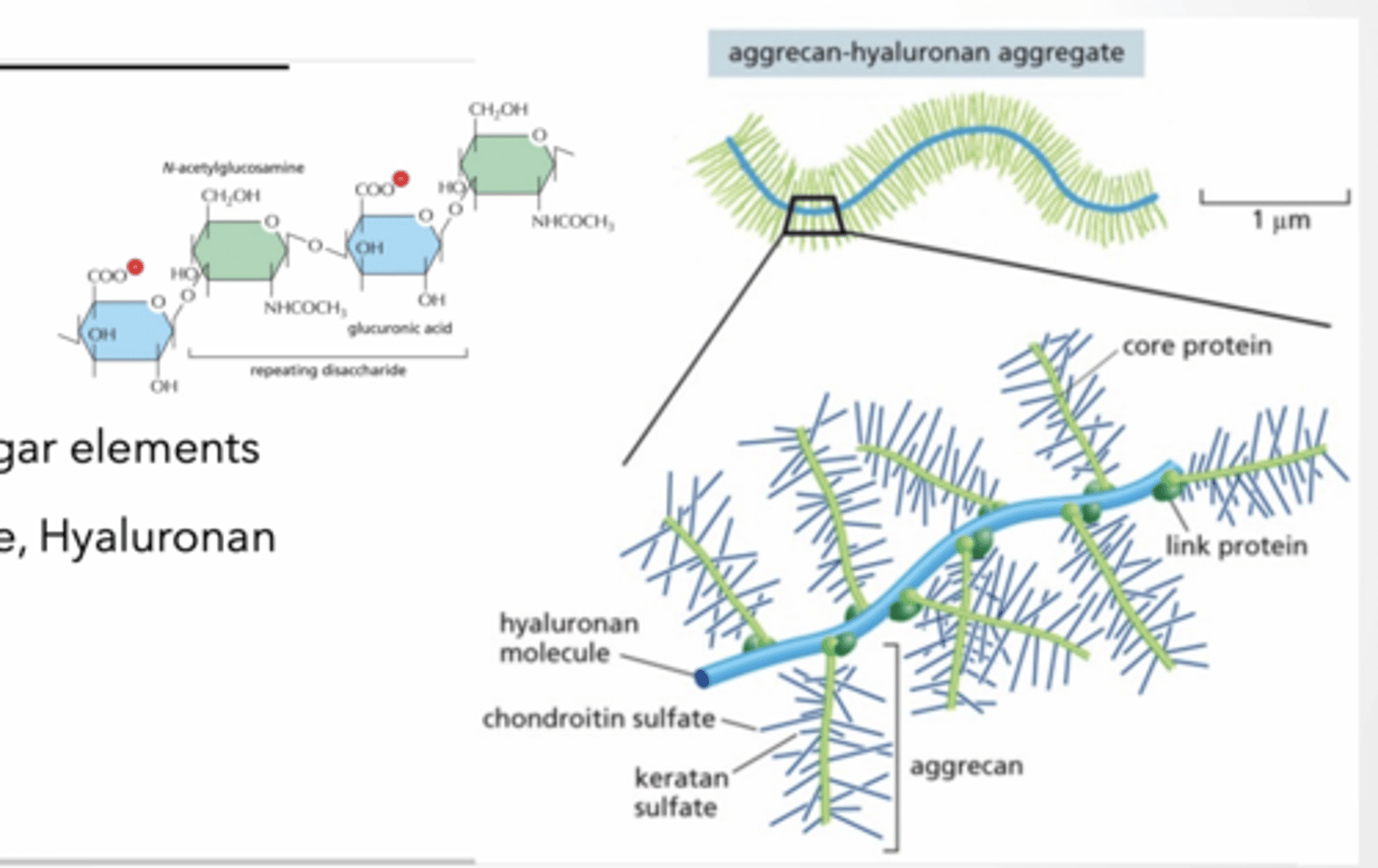
PROTEOGLYCANS
GAGs made of repeated sugar elements
Keratin, Chondrotin sulfate, Hyaluronan
Proteins link together GAGs
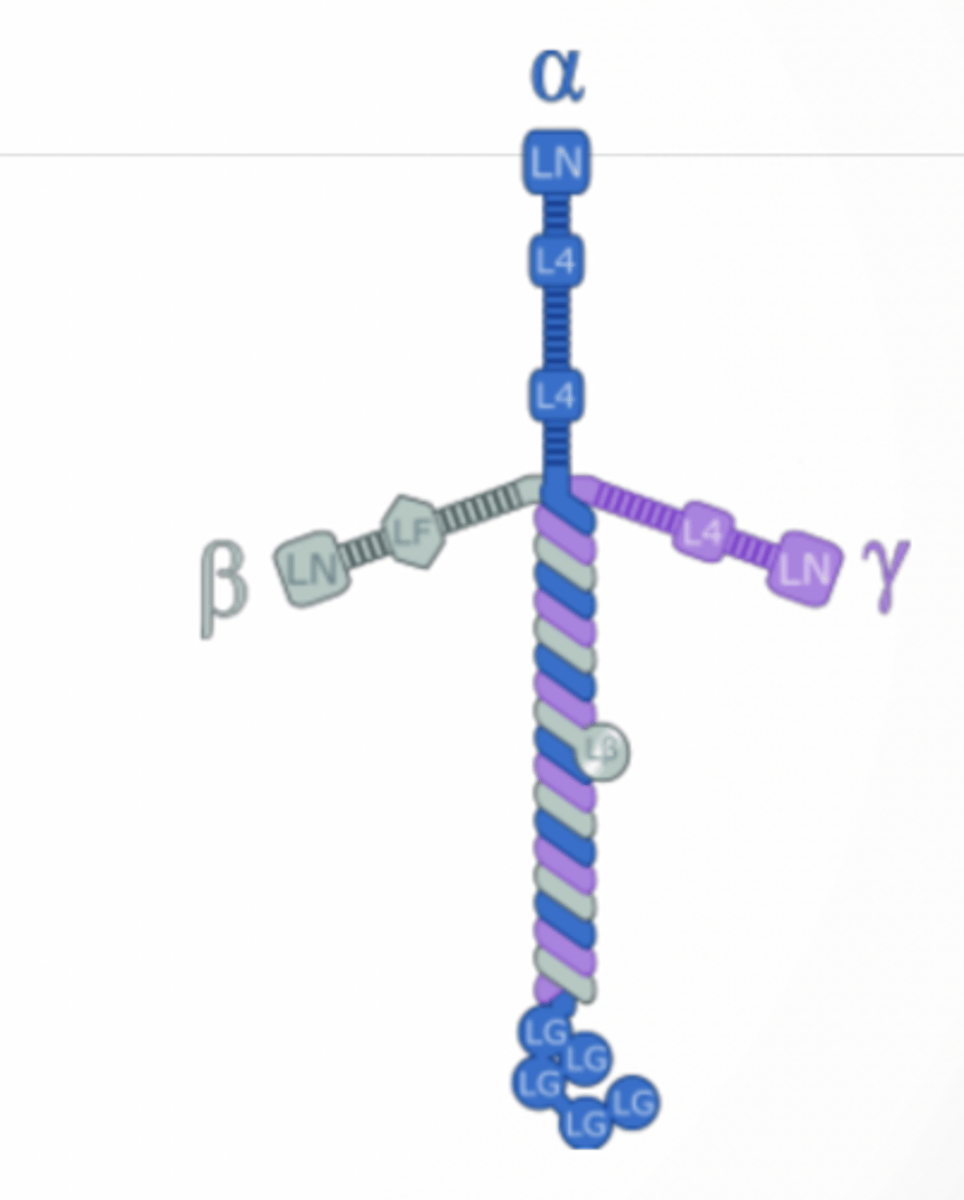
LAMININ
Fibrous proteins
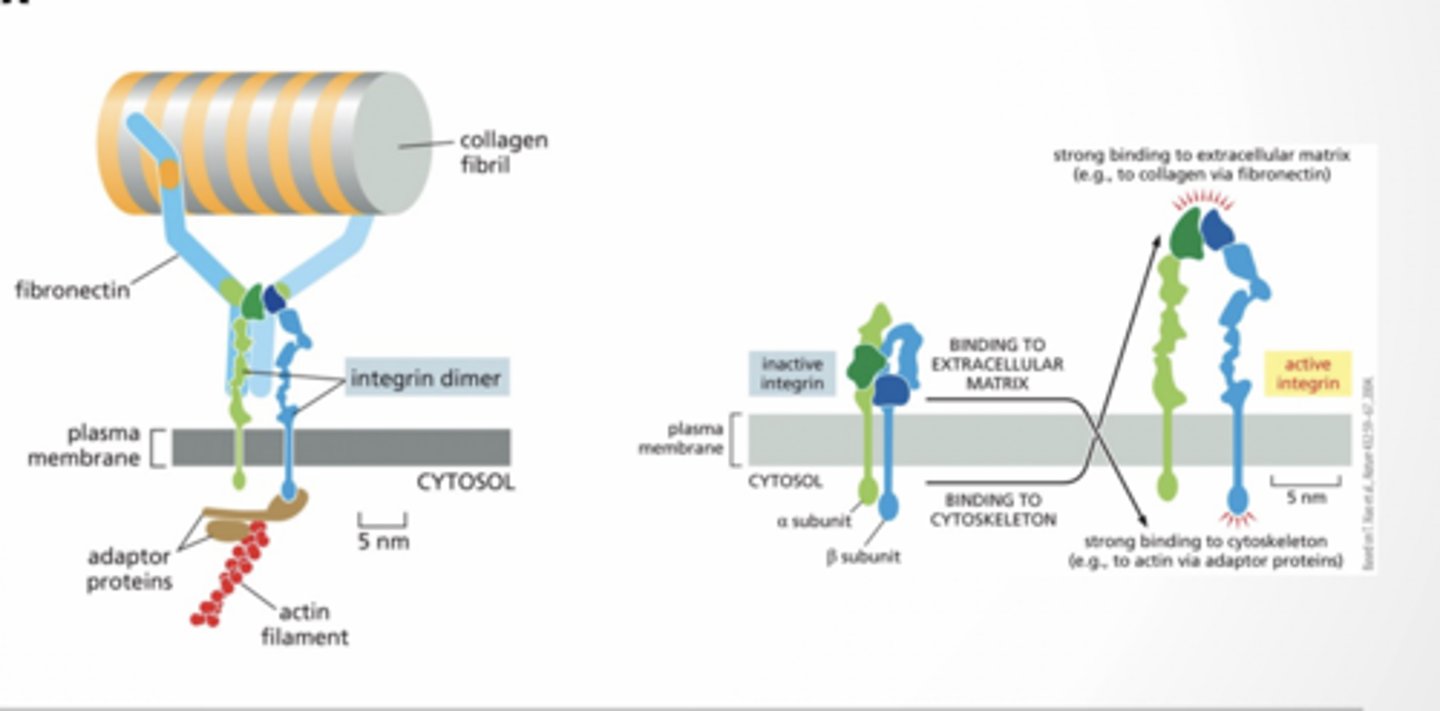
INTEGRINS
Used to connect cells to extracellular matrix
Proteoglycans feet
feet
Laminin feet
feet
Integrins feet
feet
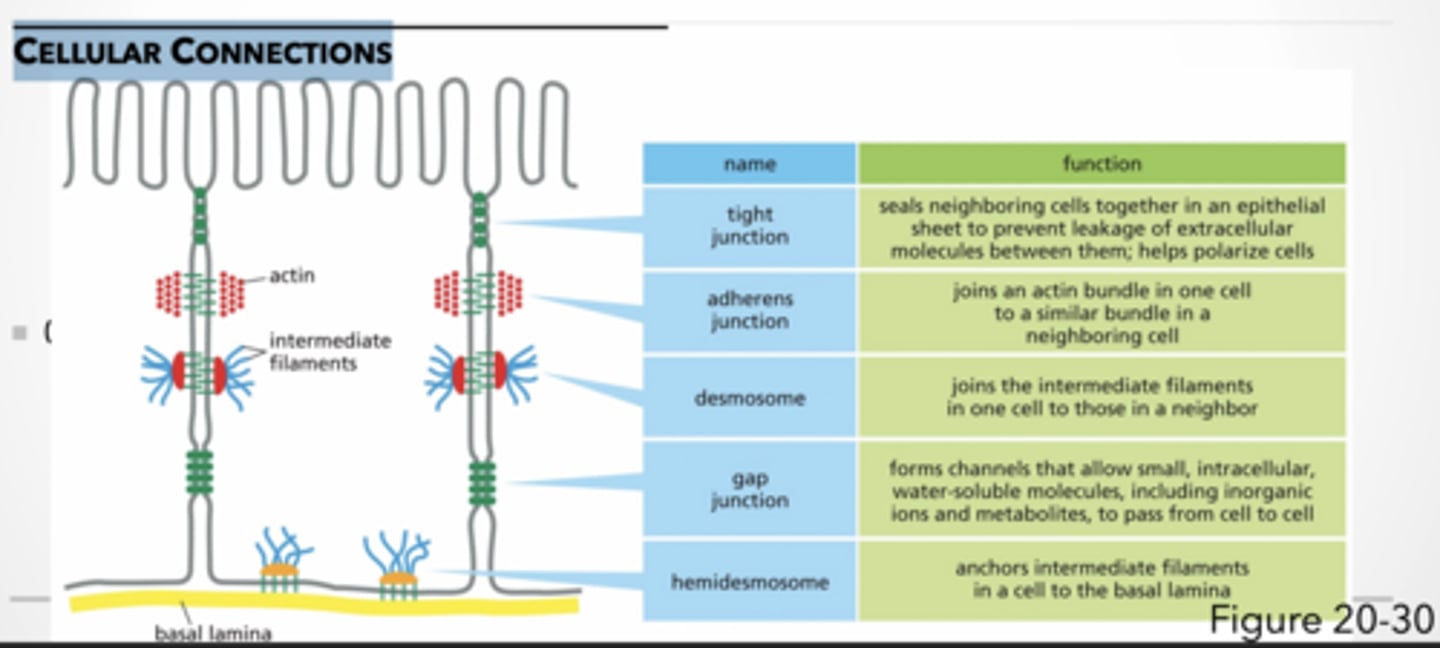
CELLULAR CONNECTIONS
CELLULAR CONNECTIONS
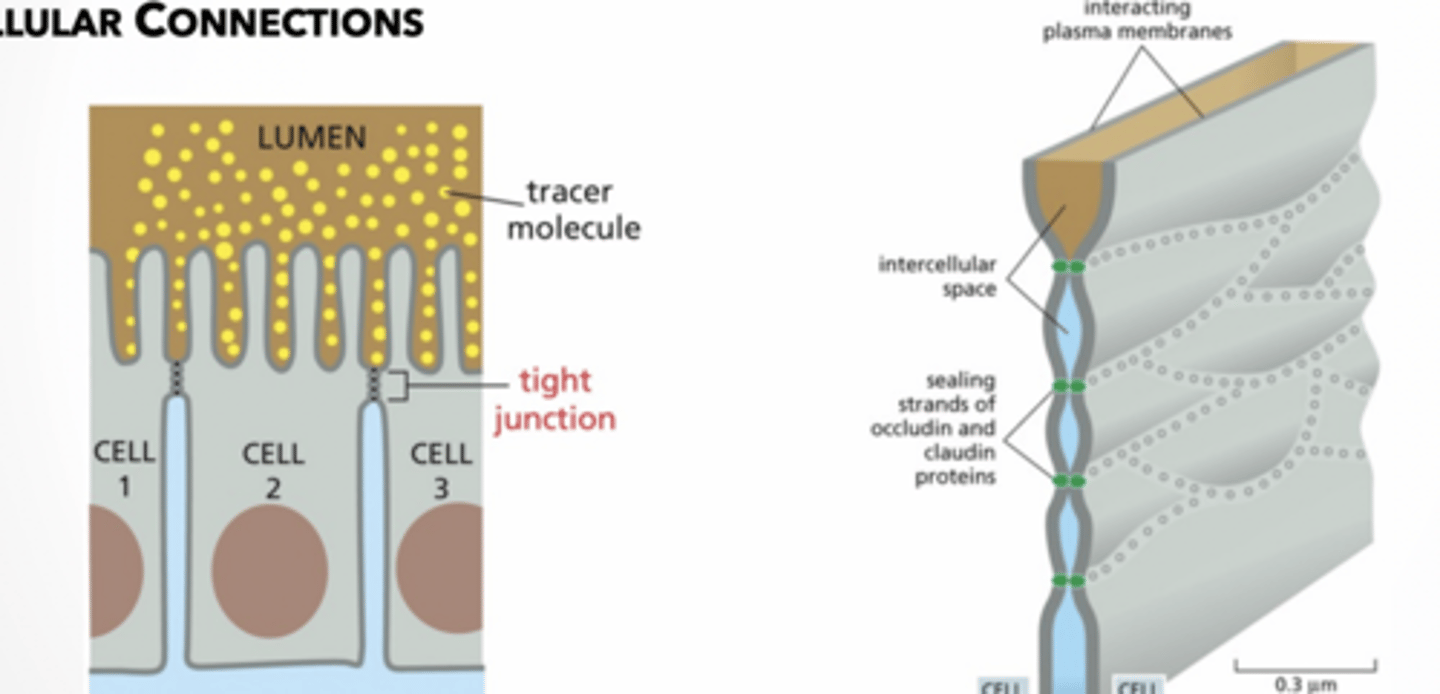
tight junction → seals neighboring cells together in an epithelial sheet to prevent leakage of extracellular molecules between them; helps polarize cells
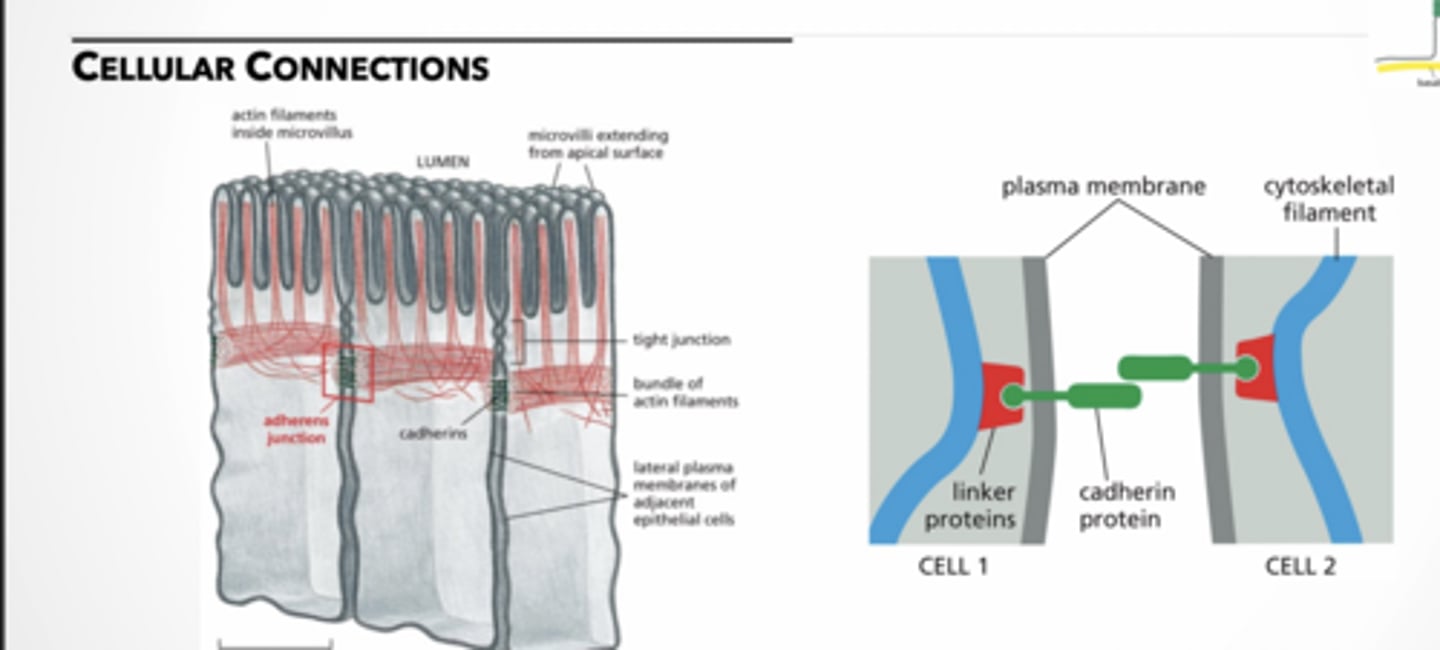
adherens junction → joins an actin bundle in one cell to a similar bundle in a neighboring cell
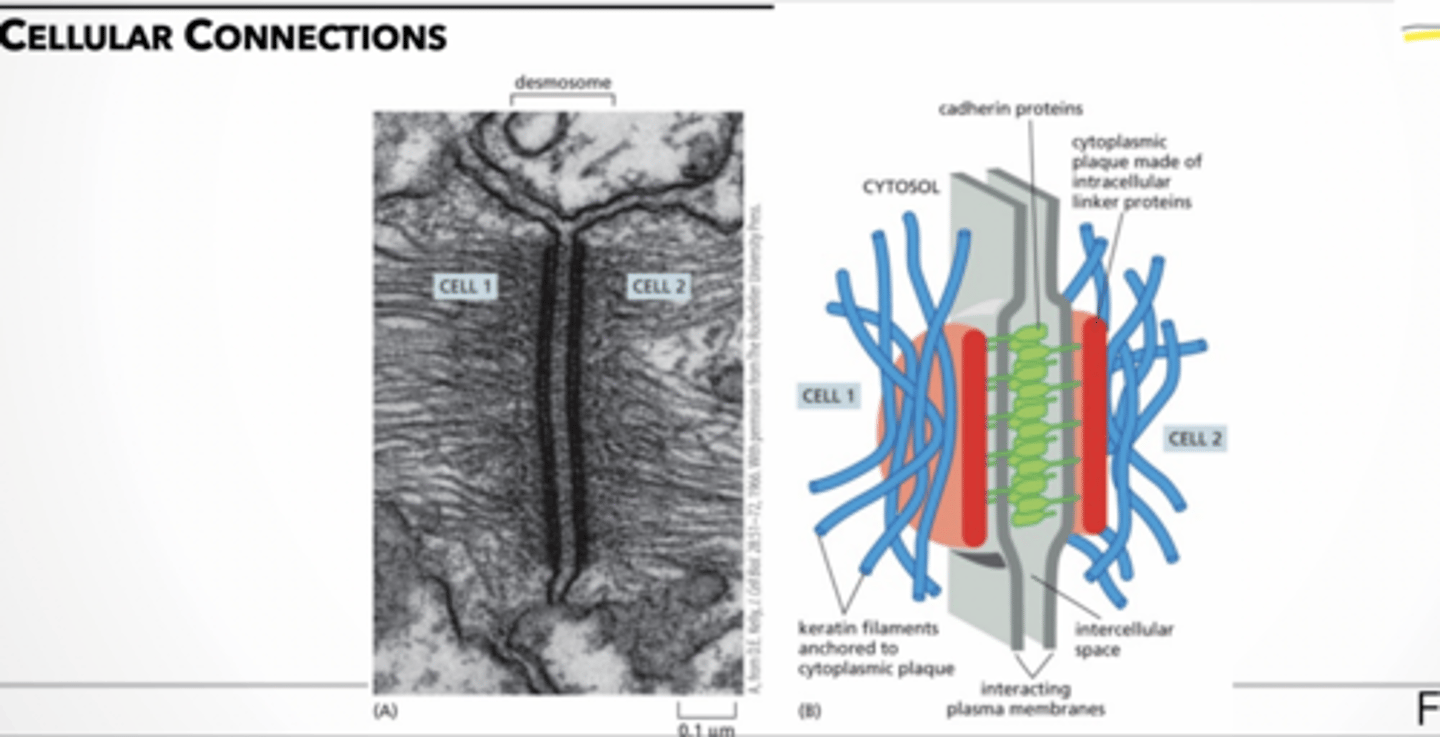
desmosome → joins the intermediate filaments in one cell to those in a neighbor
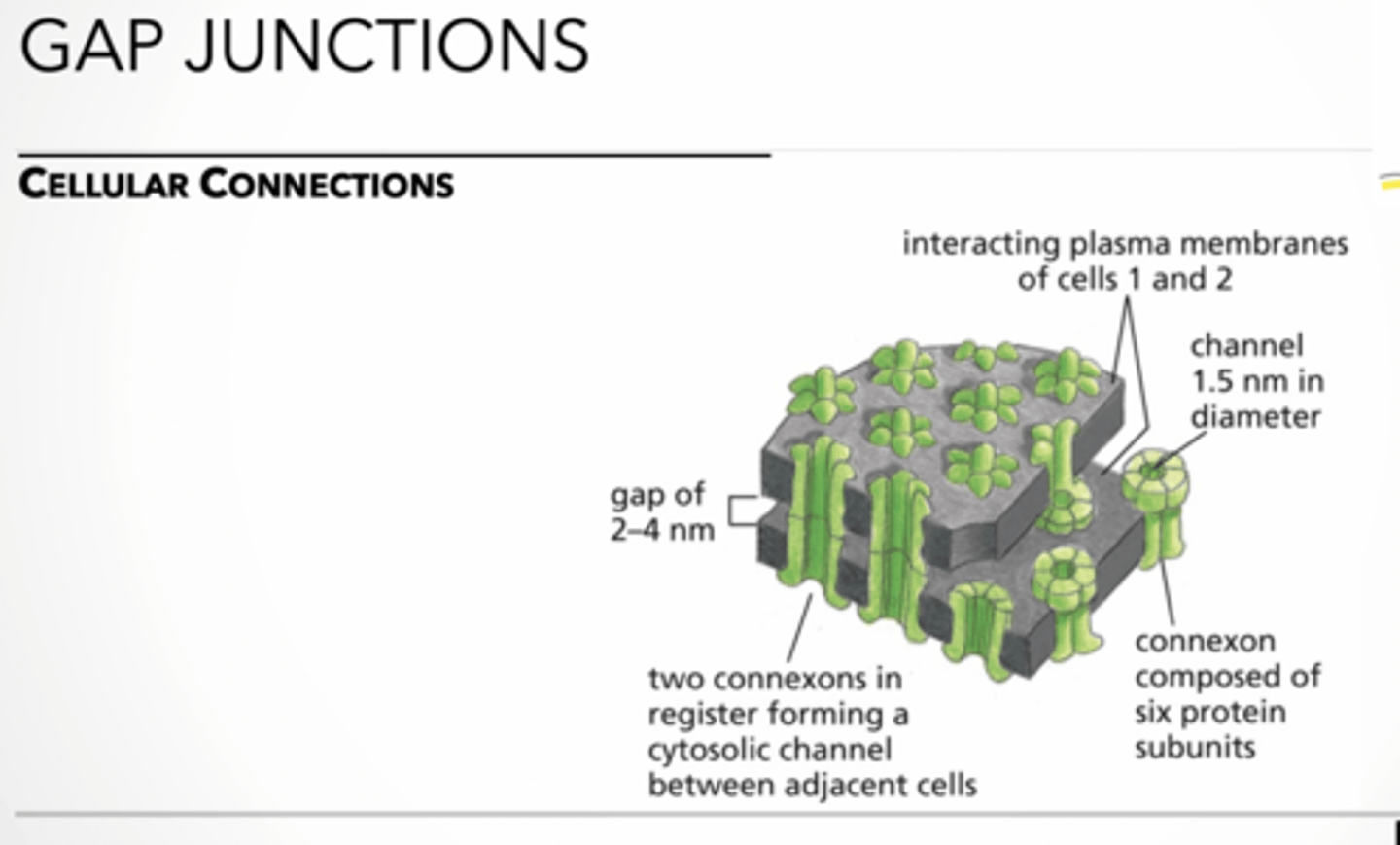
gap junction → forms channels that allow small, intracellular, water-soluble molecules, including inorganic ions and metabolites, to pass from cell to cell
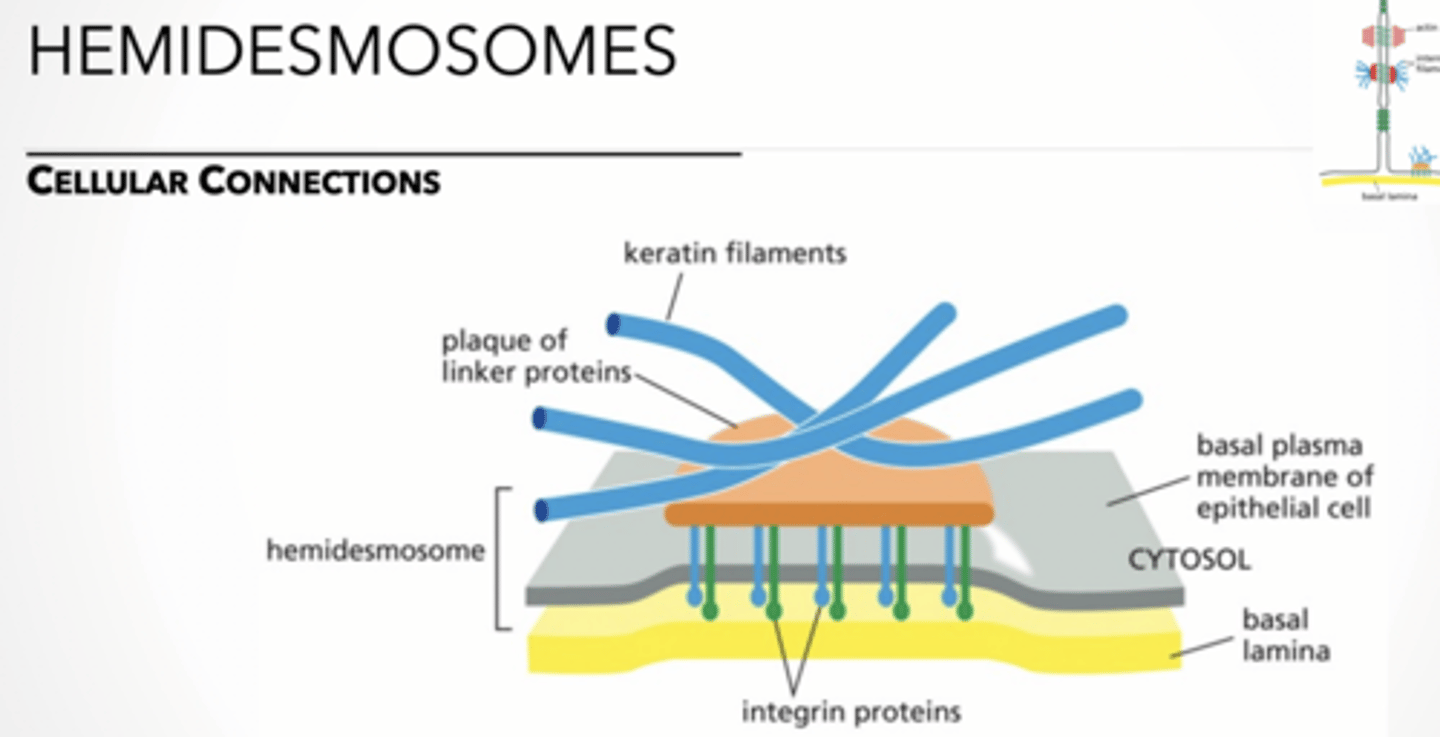
hemidesmosome → anchors intermediate filaments in a cell to the basal lamina
TJ feet
feet
adherin feet
feet
demosome feet.
feet
Gap junction feet
feet
Hemidemosome feet
feet
Tissues , Epithelial
TISSUES
TISSUE ARE MADE OF MULTIPLE TYPES OF CELLS
Organs = multiple tissue layers working together
Skin is NOT just one cell type
It has:
epithelium (top layer)
connective tissue (middle)
fat (bottom)
Epithelial types
columnar
cuboidal
stratified
squamous
basal lamina
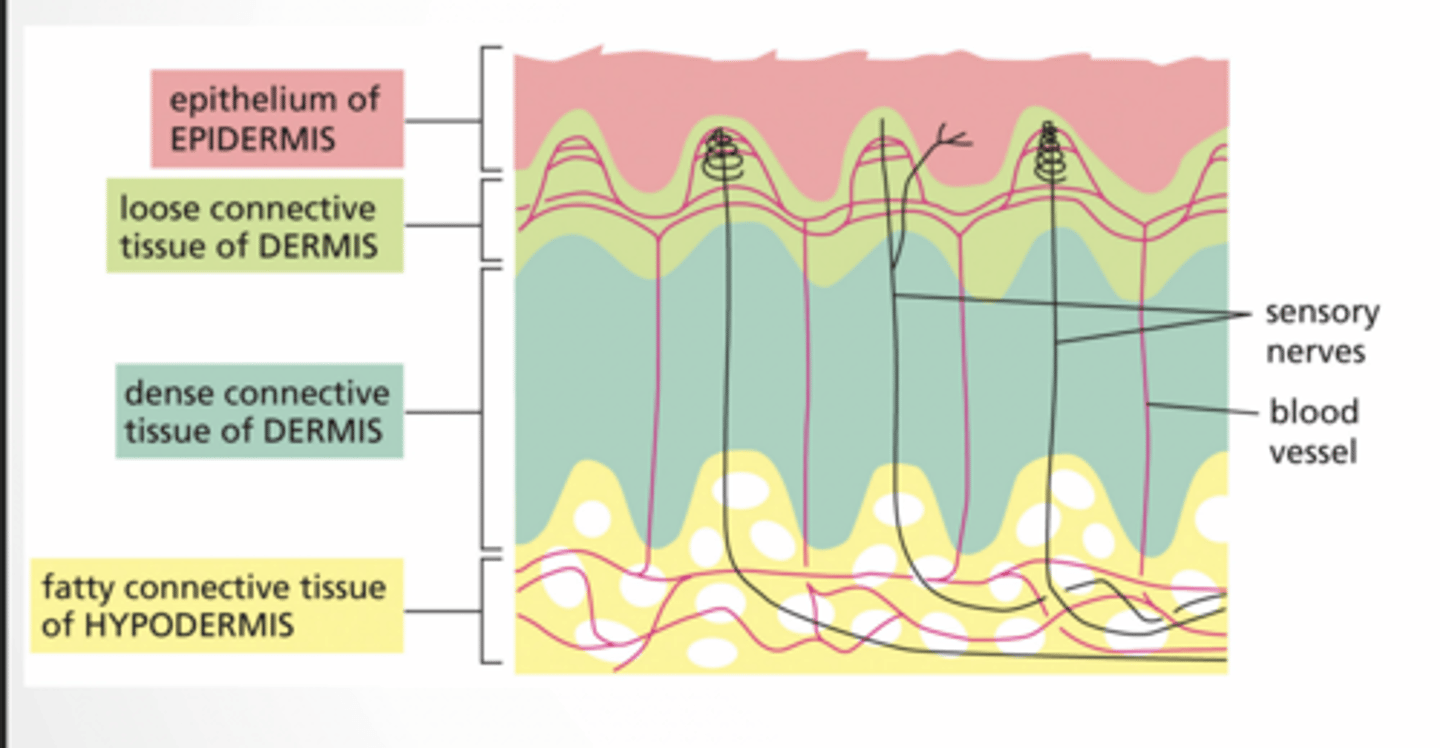
Epidermis
top = dead protective layer
keratinocytes → structure
melanocytes → pigment
Langerhans → immune
Loose connective tissue = spread out cells + immune cells
immune defense
flexible
dense connective tissue of dermis
Dense connective tissue = tight fibers
strong (tendons, dermis)
less space, fewer cells
QUORUM SENSING
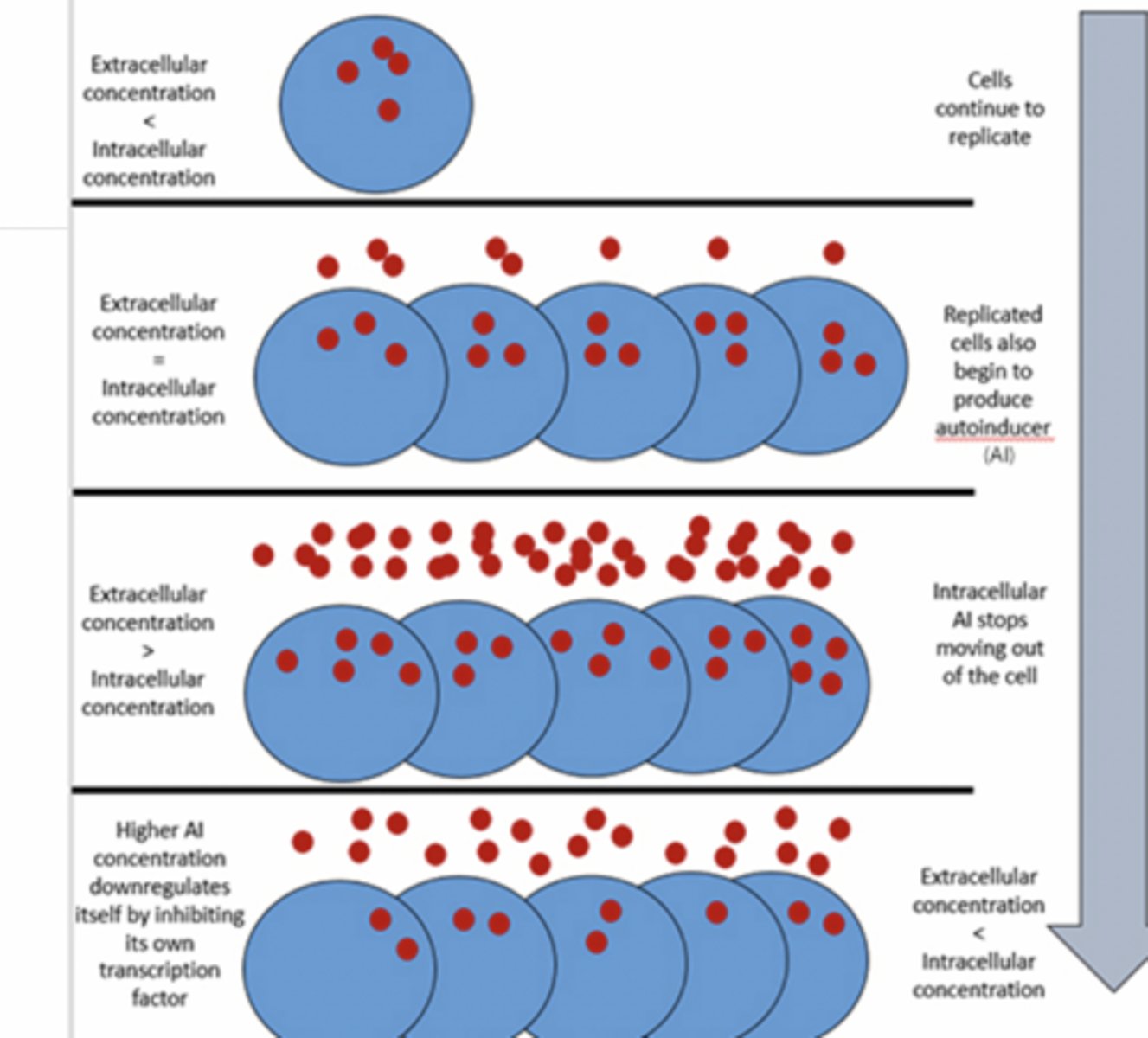
Allow bacteria to sense the density of their population
An auto inducer is produced by the bacteria constitutively
The bacteria have a receptor that binds with the auto inducer and starts a signaling cascade
Extracellular concentration < intracellular concentration
Cells continue to replicate
Replicated cells also begin to produce autoinducer (AI)
Intracellular AI stops moving out of the cell
Higher AI concentration downregulates itself by inhibiting its own transcription factor
WHAT ARE EXTRA CELLULAR VESICLES?
Lipid bi-layer vesicles critical for cellular communication
Released from lots of different kinds of cells - plants, nematodes, mammalian, microbes
Can not replicate
Carry proteins, nucleic acids, metabolites, organelles
Initially thought to be a way the cell got rid of trash

Regulation feet
feet
Extracellular vesicles
WHAT ARE EXTRA CELLULAR VESICLES?
Lipid bi-layer vesicles critical for cellular communication
Released from lots of different kinds of cells - plants, nematodes, mammalian, microbes
Can not replicate
Carry proteins, nucleic acids, metabolites, organelles
Initially thought to be a way the cell got rid of trash
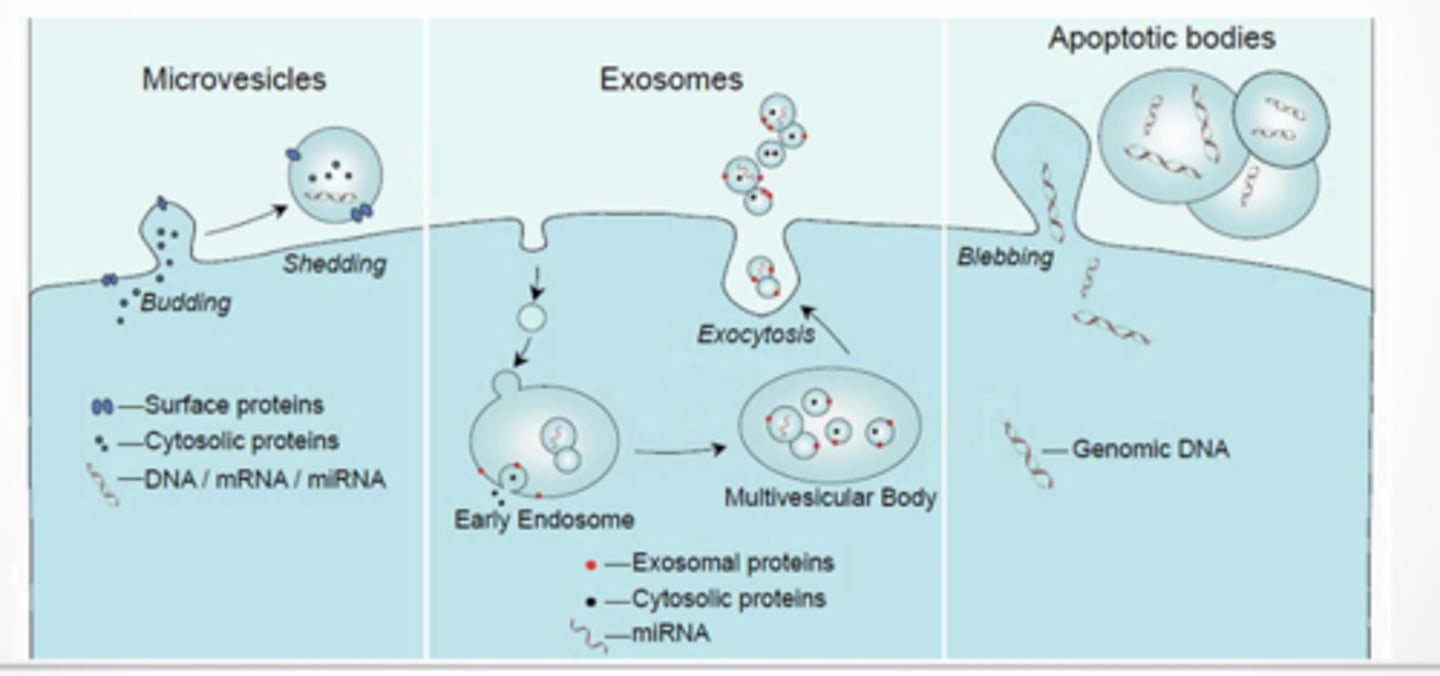
3 types
Microvesicles
Exosomes
Apoptotic bodies
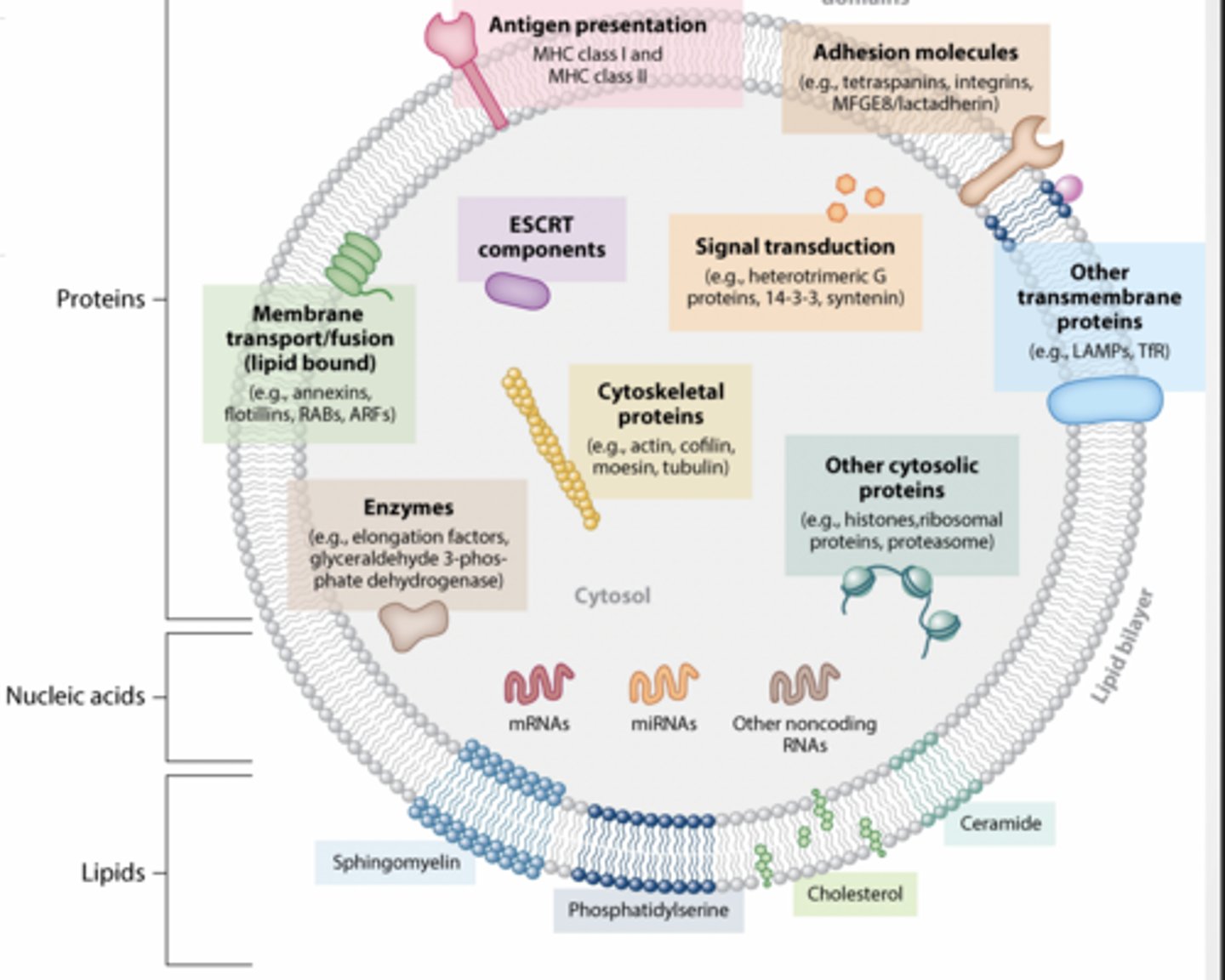
WHAT IS IN AN EV?
Proteins
Nucleic acids
Lipids
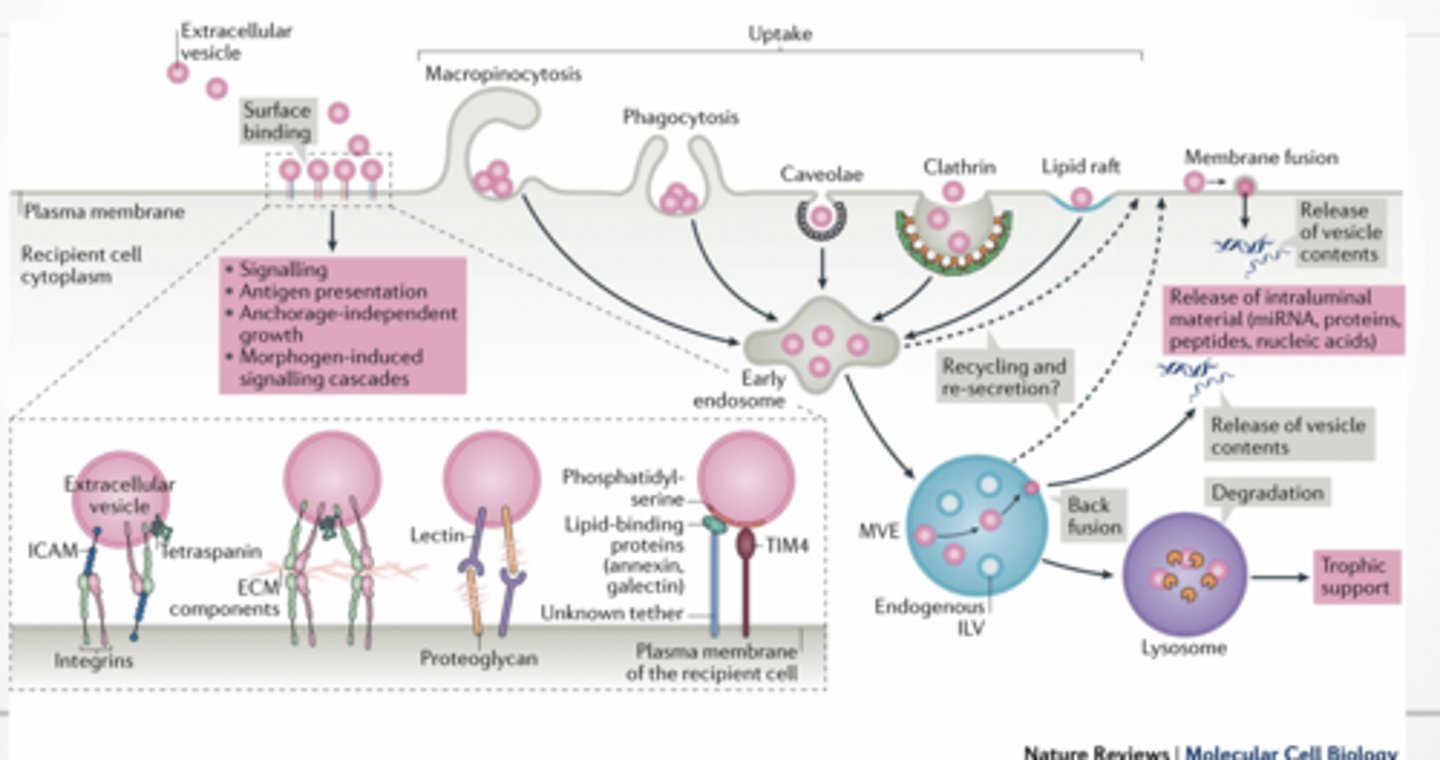
HOW ARE EVs RECEIVED BY OTHER CELLS
Phago/pinocytosis
membrane fusion
Scientists still don’t fully understand EVs
What is in feet
feet
How are they received
feet
Case study again intro
Jackie got Botox → develops drooping eyelid (ptosis)
Cause: Botox spread to the levator palpebrae superioris muscle
That muscle normally lifts the eyelid
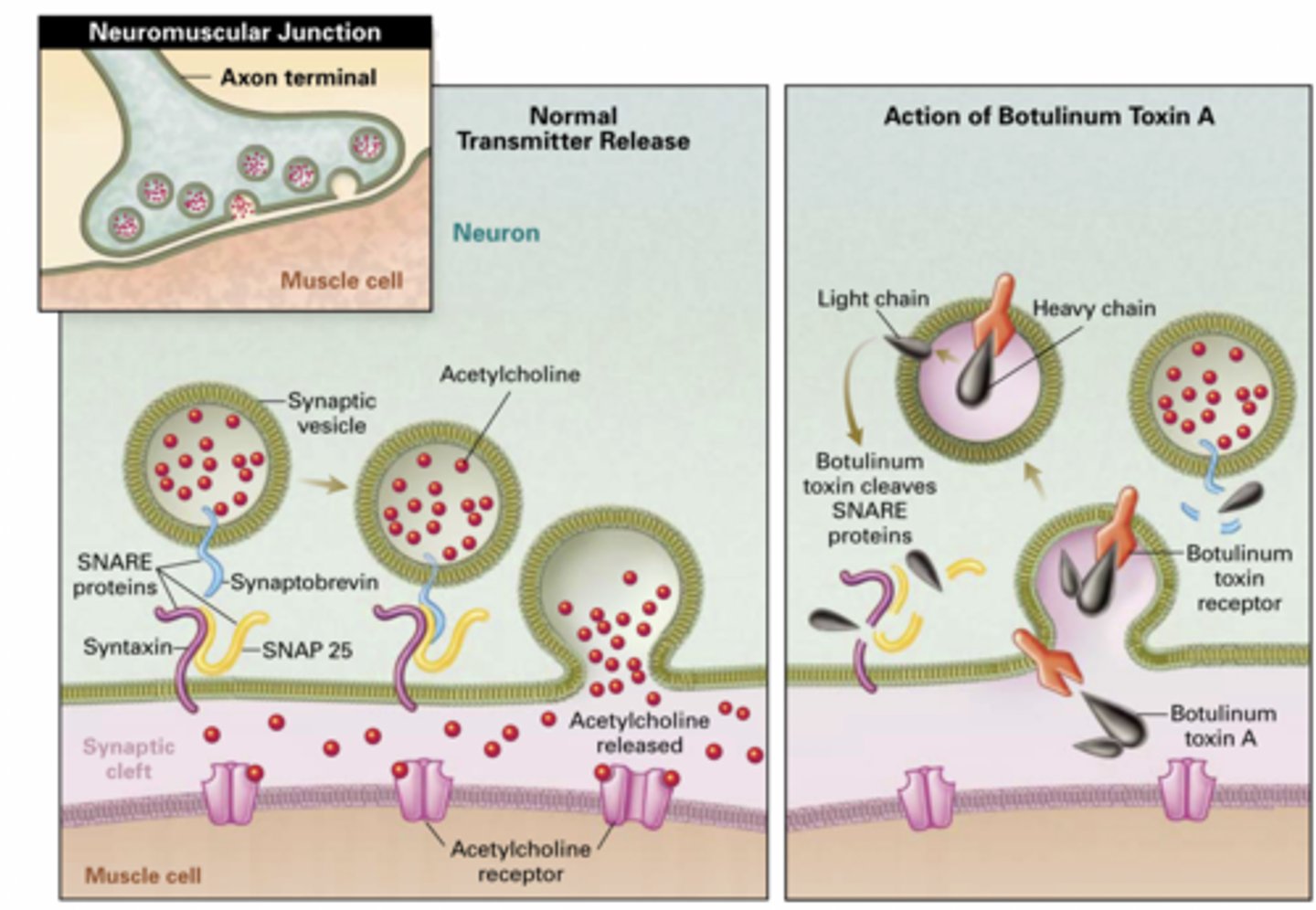
Botox = Botulinum neurotoxin (BoNT)
A neurotoxin that blocks neurotransmitter release by cleaving SNARE proteins.
It:
Enters neurons via receptor-mediated endocytosis (clathrin)
Gets into cytosol
Cleaves SNARE proteins
Blocks synaptic vesicle exocytosis
Prevents acetylcholine (ACh) release
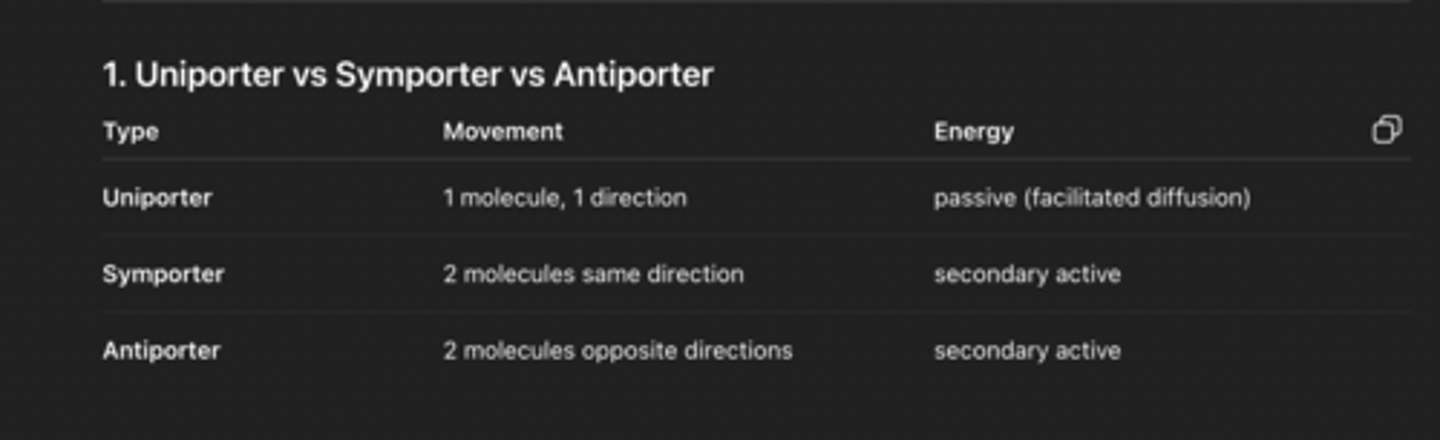
SNARE proteins
Proteins that allow vesicle fusion (synaptobrevin, syntaxin, SNAP-25)
No ACh → no muscle contraction → paralysis
More
Botox enters cells like LDL
Uses receptor-mediated endocytosis
Specifically: clathrin-dependent
Gets into cytosol
Targets SNARE proteins
Blocks exocytosis of ACh
Neuron cannot release acetylcholine-> Muscle does not contract = paralysis
Why are endocytosis and exocytosis active transport?
Because they require ATP (energy)
What is clathrin and its role?
Clathrin = protein that forms coated vesicles
Examples of proteins secreted by exocytosis
hormones (insulin)
neurotransmitters
enzymes
antibodies

Order
Correct order:
SNARE proteins interact
Vesicle fuses with plasma membrane
Acetylcholine is released from vesicle
Acetylcholine binds to the acetylcholine receptor
Receptor changes conformation
Part 2
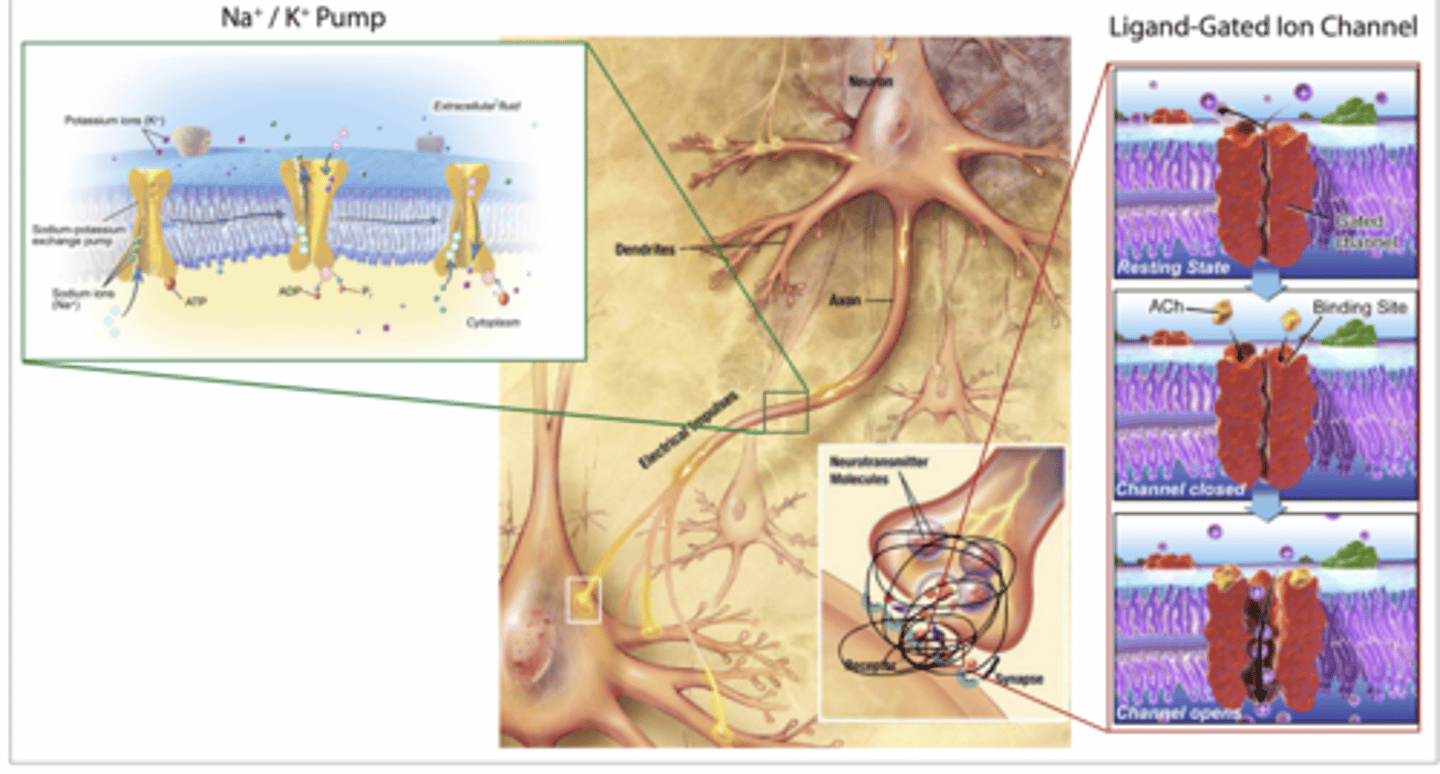
ACh binds to nicotinic acetylcholine receptors (nAChRs), which are ligand-gated ion channels, on skeletal muscle.
Binding of the ligand acetylcholine to the nAChRs induces these ion channels to open, allowing Na+ ions to rush into the cell
Channel opens → Na⁺ rushes INTO muscle cell
Membrane depolarizes → triggers contraction
This is facilitated diffusion, not active transport
it's a CHANNEL (not a transporter)
Ligand-gated ion channel
Allows Na⁺ diffusion
Muscarinic ACh receptor (GPCR pathway)
Steps:
ACh binds mAChR (GPCR)
Activates G protein (Gi type)
Effects:
↓ cAMP (inhibits adenylate cyclase)
Opens K⁺ channels
Cell becomes more negative (hyperpolarized)
Feet of this case shh
feet
Generally, after neurotransmitters are released in the neuromuscular junction, many of the neurotransmitters or some of their components are recycled by a reuptake mechanism inherent in the pre-synaptic neuron. Considering that the reuptake mechanism is often a sodium-dependent process that utilizes membrane potential, what type of membrane transport might you expect to be in play here?
secondary active transport (symporter)
Na⁺ moves down gradient
Neurotransmitter moves with it into neuron
LAST PART
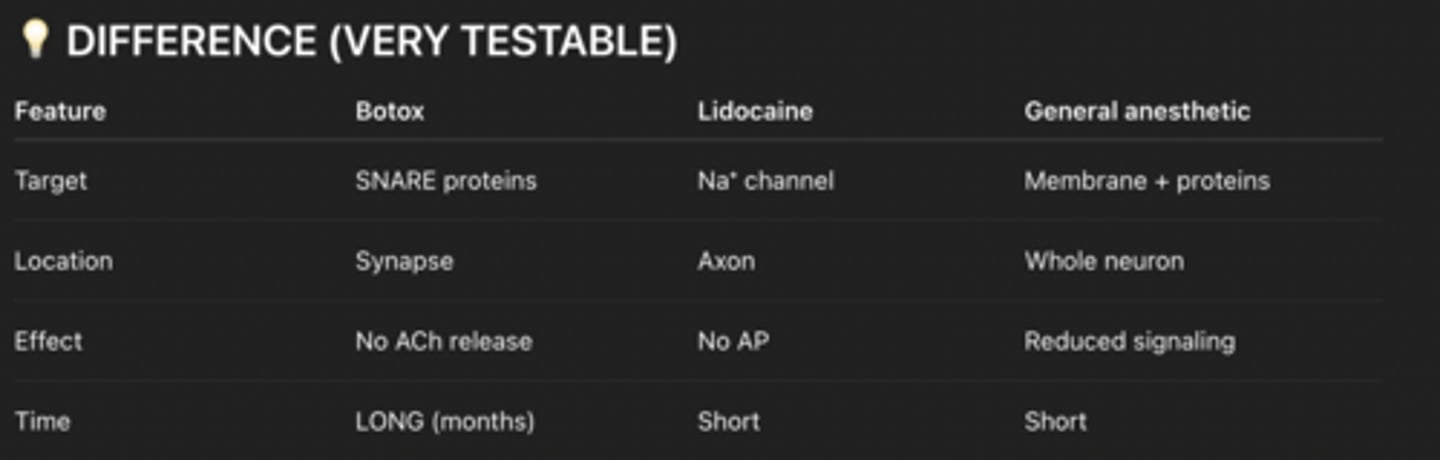
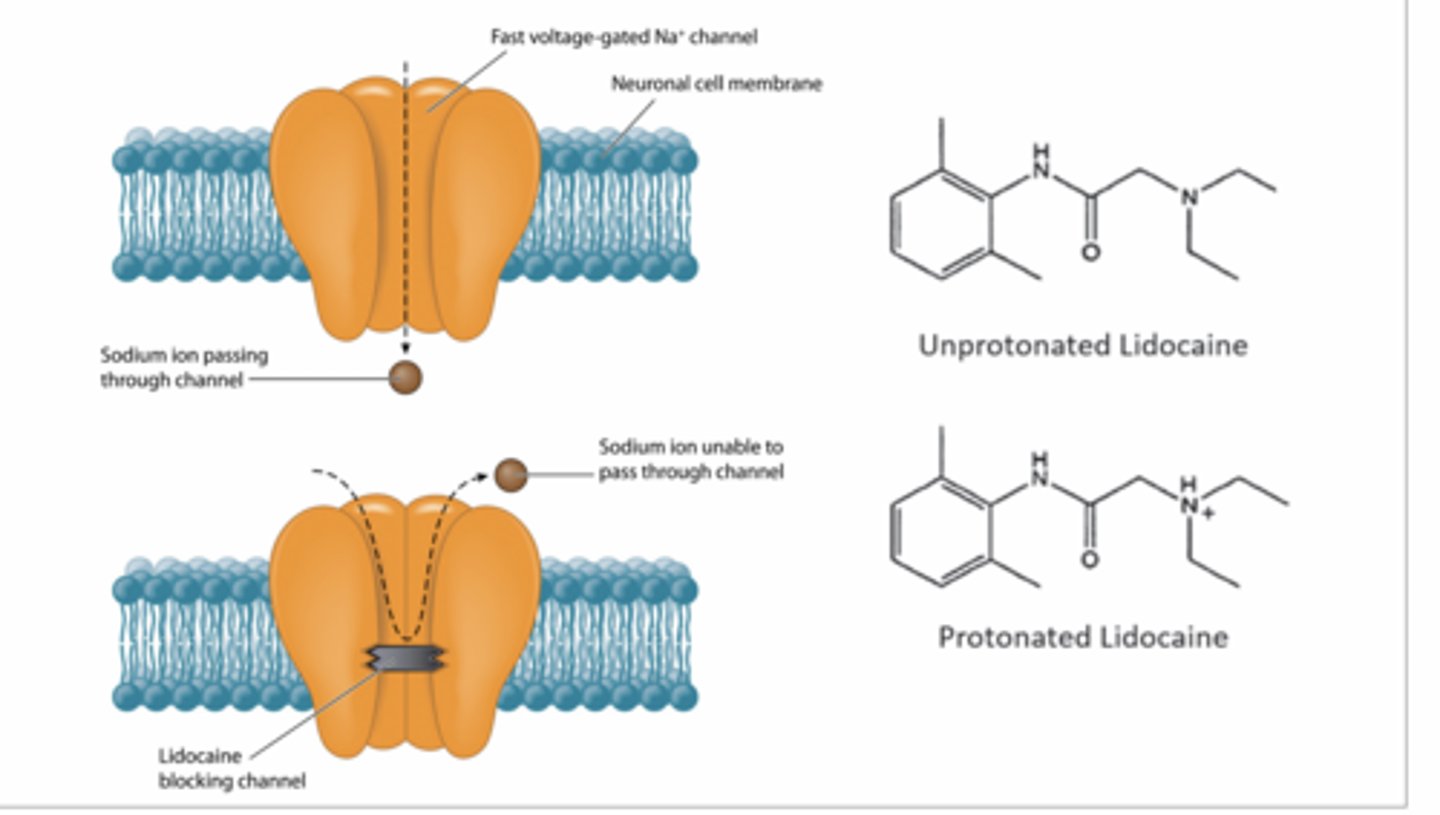
lidocaine,
Uncharged lidocaine diffuses into neuron
Becomes protonated (charged) inside
Binds inside Na⁺ channel
Physically blocks Na⁺ entry
Ach cant enter
No depolarization AND NO nerve signal
GENERAL ANESTHETICS
Don’t block one specific protein — they mess with the membrane itself
Mechanism:
Insert into lipid bilayer
Change:
membrane fluidity
membrane pressure
This alters ion channel shape → channels don’t open properly
Reduced neural activity (brain-wide)
WHY LOPIDINE HELPS
Botox paralyzes levator palpebrae (skeletal muscle)
Lopidine activates smooth muscle (tarsus superior)
Mechanism:
α-adrenergic agonist (GPCR)
Causes contraction → lifts eyelid slightly (~2 mm)
questions
Hydrophobic molecule in membrane — mimic anesthetic?
YES (likely)
Why:
General anesthetics work by inserting into membrane
Your molecule:
spans bilayer
disrupts lipid organization → could alter protein function
IV Botox (super important concept)
This would be VERY BAD
Effects:
Widespread paralysis
Respiratory failure (diaphragm)
No neuromuscular signaling
Why lidocaine stays inside cell
It’s about charge
Outside: uncharged → diffuses in
Inside: becomes protonated (charged)
Charged = can’t cross membrane
So it gets trapped inside neuron
α-latrotoxin (black widow toxin)
Observed:
First: muscle spasms
Then: paralysis
Opposite of Botox
Causes massive ACh release
Likely:
increases Ca²⁺ influx OR
forces vesicle fusion (SNARE activation)
Result:
Overstimulation → spasms
Neurotransmitter depletion → paralysis