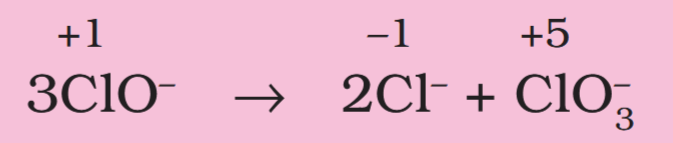Redox Reactions [ALMOST DONE]
1/114
Earn XP
Description and Tags
Suitable for IAT, NEST, NEET, JEE, etc. Revision of Redox Reactions from NCERT. Iodometry is remaining, and how to balance chemical equations I'm not gonna add for obvious reasons. Mostly everything else should be here. Question mode: Flashcards only. Answer mode: Answer with definition. Recommended: spaced repetition. Good luck with exams!
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
115 Terms
What is hypothetical charge?
hypothetical charge = (number of valence electrons in the isolated atom of that element) - (number of valence electrons after assigning all bond pairs to the more electronegative atom in a molecule)
What is the oxidation number of Cr in CrO5?
+6
What is the oxidation number of an atom in its elemental state?
0
What is the oxidation number of an alkali metal?
+1
What is the oxidation number of an alkali earth metal?
+2
What is the oxidation number of hydrogen when it combines with most elements?
+1
What is the oxidation number of hydrogen in metal hydrides?
−1
What is the oxidation number of oxygen in most compounds?
−2
What is the oxidation number of peroxide?
O2−2 oxidation number = −1
What is the oxidation number of superoxide?
O2− oxidation number = −21
What is the oxidation number of oxygen in OF2?
+2
What is the oxidation number of oxygen in O2F2?
+1
What is the maximum oxidation number of any element?
The maximum number of electrons it can lose i.e. the number of electrons in its valence shell
What is the minimum oxidation number of any element?
The maximum number of electrons it can gain to complete its octet (mostly 8 - valency)
Which two elements have the maximum oxidation state ever?
Osmium (Os+8) and Ruthenium (Ru+8)
What is the oxidation number of both A and B when there is a coordinate bond between them (A→B) and
electronegativity of B is greater than electronegativity of A?
A+2→B−2
What is the oxidation number of both A and B when there is a coordinate bond between them (A→B) and
electronegativity of A is greater than electronegativity of B?
A0→B0
What is the oxidation state of elements in an amalgum?
0
If an element is in its highest oxidation state, it will act as ________ agent.
(oxidising / reducing)
oxidising
If an element is in its lowest oxidation state, it will act as ________ agent.
(oxidising / reducing)
reducing
KMnO4 is a strong _______ agent.
(oxidising / reducing)
oxidising
K2Cr2O7 is a strong _______ agent.
(oxidising / reducing)
oxidising
HNO3 is a strong _______ agent.
(oxidising / reducing)
oxidising
metal hydrides are strong ________ agents.
(oxidising / reducing)
reducing
What is disproportionation?
the same element undergoes both oxidation and reduction.
What is comproportionation?
different atoms of the same element at different oxidation states react to produce that element at an intermediate oxidation state.
Zn(s)+CuSO4(aq)→ZnSO4(aq)+Cu(s)
This is _____________ reaction
(molecular / ionic / net ionic / half)
molecular
Zn(s)+CuSO4(aq)→ZnSO4(aq)+Cu(s)
Write the ionic reaction for this.
Zn(s)+Cu+2(aq)+SO4−2(aq)→Zn+2(aq)+SO4−2(aq)+Cu(s)
Zn(s)+CuSO4(aq)→ZnSO4(aq)+Cu(s)
Write the net ionic reaction for this.
Zn(s)+Cu+2(aq)→Zn+2(aq)+Cu(s)
Zn(s)+CuSO4(aq)→ZnSO4(aq)+Cu(s)
Write the half reactions for this redox reaction.
Zn(s)→Zn+2(aq)
Cu+2(aq)→Cu(s)
acidic strength of an oxo-acid is _________ proportional to the Oxidation number of the central atom in the acid.
(directly / inversely)
directly
Arrange HClO, HClO2, HClO3, HClO4 in decreasing order of acidic strength
HCl^{+7}O_4 > HCl^{+5}O_3>HCl^{+3}O_2>HCl^{+1}O
Which is more acidic, H2SO3 or H2SO4?
H2SO4
Which is more acidic, HNO2 or HNO3?
HNO3
Arrange in order of acidic strength: H3PO2, H3PO3, H3PO4
H_3P^{+1}O_2 > H_3P^{+3}O_3 > H_3P^{+5}O_4
(it is an exception)
What is the qualitative definition of equivalent mass E?
Equivalent mass of a substance is the amount of substance which directly or indirectly combines with 1 gram of H2 / 8 grams of O2 / 35.5 grams of Cl2
How do you calculate the equivalent mass of elements?
E=valencyatomic mass
How do you calculate the equivalent weight of ions?
E=∣charge on ion∣molar mass of ion
How do you calculate the equivalent weight of acids?
E=basicitymolar mass of acid
How do you calculate the equivalent mass of bases?
E=aciditymolar mass of base
How do you calculate the equivalent mass of an ionic compound?
E=magnitude of total charge on cation or anionmolar mass of the compound
How do you calculate the equivalent mass of an oxidising or reducing agent?
E=change in oxidation state per molecule or ionmolar mass of compound
How do you calculate the equivalent mass of a compound using its n-factor?
E=n-factor / valency factormolar mass
What is the n-factor of acids?
the basicity of the acid
What is the n-factor of bases?
the acidity of the base
What is the n-factor of salts in a non-redox reaction?
n-factor = charge on the cation that is replaced * number of cations replaced
What is the n-factor of salts in a redox reaction where only one element of the salt undergoes change in oxidation state?
= change in oxidation state of that element * number of atoms of element
What is the n-factor of salts in a redox reaction where two elements of the salt are either both oxidised or both reduced?
= sum of (change in oxidation state of element * number of atoms of element) of both elements
What is the n-factor of salts in a redox reaction where two elements of the salt undergo oxidation and reduction respectively?
n-factor = (change in oxidatoin state of element * number of atoms of element) for either element
What is the n-factor of hydrogen peroxide when it acts as an oxidising or reducing agent?
2
What is the n-factor of an element that undergoes disproportionation?
let n1 be the n-factor of the oxidation half-reaction
let n2 be the n-factor of the reduction half-reaction
n-factor of molecule / element = n1+n2n1n2
How do you calculate the number of equivalents of a molecule in a reaction, when you have the equivalent weight of molecule?
neq=E (equivalent mass)W (molecular mass)
How do you calculate the number of equivalents of a molecule in a reaction, when you have the n-factor of the molecule?
neq=number of moles∗n-factor
How do you calculate the number of equivalents of a molecule in a reaction, when you have the normality and volume of the molecule?
neq=NV
How do you calculate the number of equivalents of a molecule in a reaction, when you have the molarity, volume, and n-factor of the molecule?
neq=M∗V∗n-factor
2Na(s)+H2(g)→2NaH(s)
What is being reduced, what is being oxidised?
sodium is being oxidised, hydrogen is being reduced.
Half reactions that involve loss of electrons are called __________ reactions.
(oxidation / reduction)
oxidation
Half reactions that involve gain of electrons are called __________ reactions.
(oxidation / reduction)
reduction
Place a strip of metallic zinc in an aqueous solution of copper nitrate for about an hour. What do you observe?
The strip becomes coated with reddish metallic copper and the blue colour of the solution disappears.
What colour is ZnS?
white
Zn(s)+Cu2+(aq)→Zn2+(aq)+Cu(s)
The state of equilibrium for this reaction favours products or reactants?
products
Cu(s)+2Ag+(aq)→Cu2+(aq)+2Ag(s)
The state of equilibrium for this reaction favours products or reactants?
products
Co(s)+Ni2+(aq)→Co2+(aq)+Ni(s)
The state of equilibrium for this reaction favours products or reactants?
neither
Arrange these in the order of increasing electron releasing tendency:
Zn, Cu, Ag
Ag < Cu < Zn
What is the oxidation number of aluminium in most of its compounds?
+3
What is Stock Notation (when representing oxidation numbers)?
According to this, the oxidation number is expressed by putting a Roman numeral representing the oxidation number in parenthesis after the symbol of the metal in the molecular formula.
All decomposition reactions are redox reactions. True or false?
false. not all of them are redox reactions.
Which 3 alkaline earth metals are good reductants?
Ca, Sr, Ba
All alkali metals and some alkaline earth metals (Ca, Sr, and Ba) which are very good reductants, will displace hydrogen from _______ water.
(hot / cold)
All alkali metals and some alkaline earth metals (Ca, Sr, and Ba) which are very good reductants, will displace hydrogen from cold water.
Less active metals such as magnesium and iron react with _________ to produce dihydrogen gas.
(cold water / hot water / steam)
Less active metals such as magnesium and iron react with steam to produce dihydrogen gas.
Metals which are not good enough reductants to displace hydrogen from water, are capable of displacing hydrogen from acids. True or false?
True.
Under which condition can a metal displace a different metal in a metal displacement reaction?
The reducing (displacing) metal is a better reducing agent than the one that is being reduced (displaced).

Na is a very good reducing agent. what is being reduced?
Hydrogen.
Which three metals are used to prepare hydrogen gas in a laboratory? What sort of reaction is it?
The metals used are Zn, Mg, and Fe.
The reaction is a nonmetal displacement reaction.
How can the reactivity of metals be measured?
The reactivity of metals is reflected in the rate of hydrogen gas evolution.
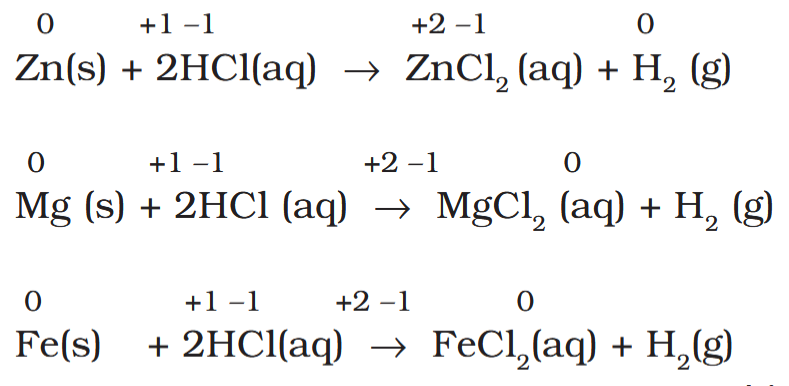
These reactions are used in the laboratory to produce hydrogen gas. Which is the slowest reaction, and which is the fastest reaction?
Third is slowest.
Second is fastest.
What are two examples stated in NCERT of metals that do not react with hydrochloric acid?
silver and gold.
Fluorine is so reactive that it can replace chloride, bromide and iodide ions in solution.
True or false?
True
Fluorine is so reactive that it attacks water and displaces the oxygen of water.
True or false?
True
What is the reaction between fluorine gas and water?

Why is it that the displacement reactions of chlorine, bromine and iodine using fluorine are not generally carried out in aqueous solution?
Fluorine is so reactive that it attacks water and displaces the oxygen of water, so it doesn’t displace halogens.
Chlorine can displace bromide and iodide ions in an aqueous solution.
True or false?
true
Fluorine can displace chloride, bromide and iodide ions in an aqueous solution.
True or false?
False, fluorine displaces the oxygen in water instead
Bromine can displace iodide ion in aqueous solution.
True or false?
True
What is the ‘Layer test’?
Reactions (7.41) and (7.42) form the basis of identifying Br– and I– in the laboratory through the test popularly known as ‘Layer Test’.
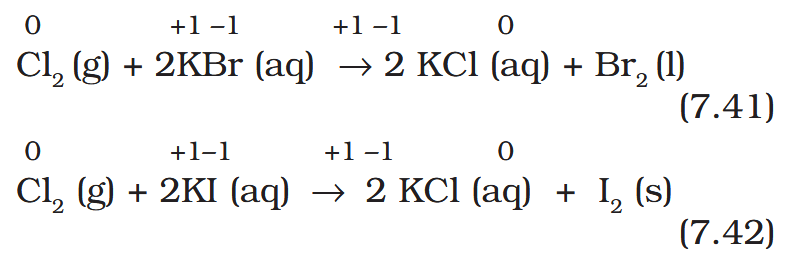
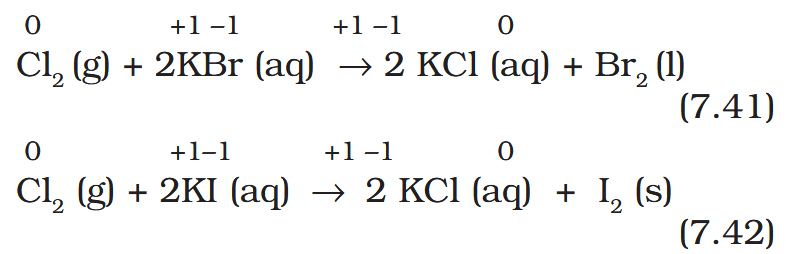
How can the formation of Br2 and I2 be identified? What is the solvent used?
As Br2 and I2 are coloured and dissolve in CCl4, can easily be identified from the colour of the solution.
What is the way to convert F- ions to F2 by chemical means?
There is now way, it can only be done through electrolytic means.
There are chemical means to oxidise Cl–, Br– and I– into Cl2, Br2 and I2, but not fluorine. Why is that?
fluorine is the strongest oxidising agent so nothing can oxidise it chemically
What is the condition for a substance to be capable of undergoing disproportionation?
It should have at least three possible oxidation states.
Which sort of redox reaction is the decomposition of H2O2?
disproportionation
what is the oxidation state of the oxygen atom in H2O2?
−1
Phosphorous, sulphur and chlorine undergo disproportionation in the ________ medium.
(acidic / alkaline)
Phosphorous, sulphur and chlorine undergo disproportionation in the alkaline medium.
Depict the disproportionation of phosphorus in alkaline medium in equation form.
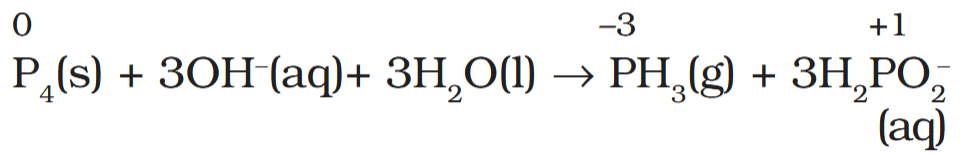
Depict the disproportionation of sulphur in alkaline medium in equation form.
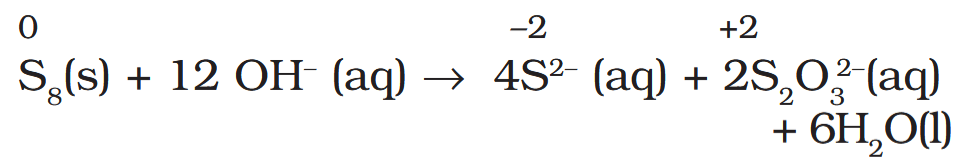
Depict the disproportionation of chlorine in alkaline medium in equation form.
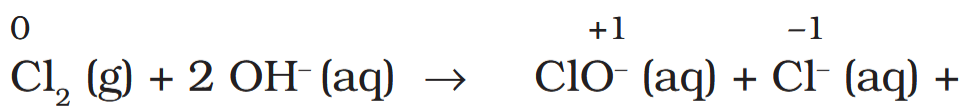
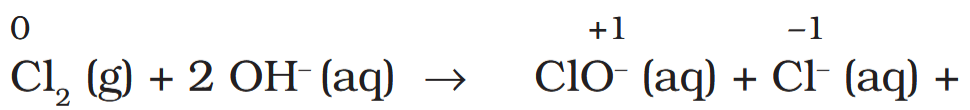
How does the product act as a bleaching agent?
The hypochlorite ion (ClO–) formed in the reaction oxidises the colour-bearing stains of the substances to colourless compounds.
Depict the reaction of fluorine and alkali in equation form.
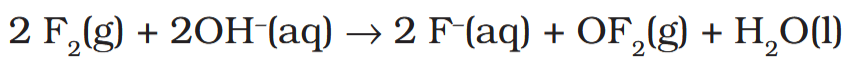
Among halogens, which element does not undergo disproportionation?
fluorine.
Which of the following species, do not show disproportionation reaction?
ClO–, ClO2–, ClO3– and ClO4–
ClO4–
Chlorine is in its highest oxidation state (+7) and it cannot exhibit an oxidation state higher than that
Depict the disproportionation reaction of ClO– in equation format