CHD pt. 4
1/59
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
60 Terms
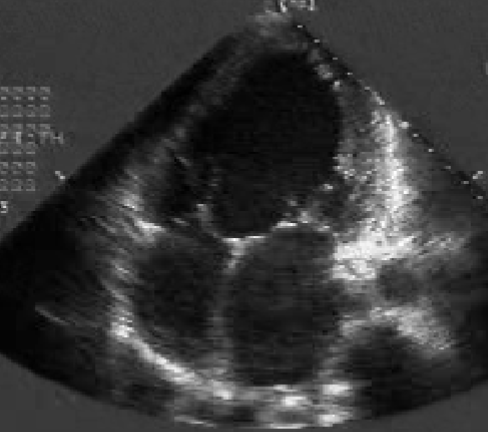
“Crab Claw” view
Showcases normal SVC presentation
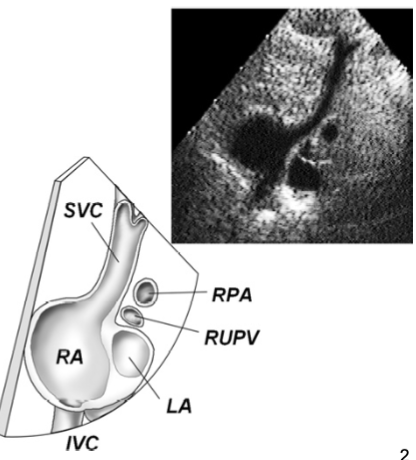
Persistent Left Superior Vena Cava
most common congenital anomaly that affects the systemic veins
The LSVC drains into the Coronary Sinus for blood to return to the RA
No tx is necessary
No complications
Where should contrast be injected in order to diagnose a persistent lt SVC?
Left arm
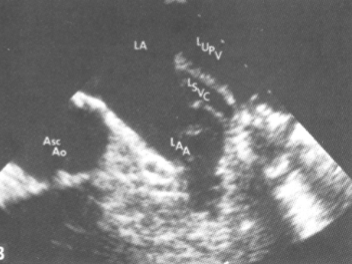
Persistent Left SVC:
dilated Coronary Sinus
May follow vessel
May mimic:
Pericardial effusion
Anomalous pulmonary venous return
Descending aorta
Cor Triatriatum
fibromuscular membranous partition
LA divided into 2 parts
Called “Sinister” or “Sinistrum”
Pulm venous return must pass through a false “chamber” / compartment posterior to the LA
membrane may have fenestrations
Diagnose obstruction of flow in the LV
mimics MS
RA division
rare
thought to be remnant of fetal circulation
called “Dextrum” or “Dexter”
Cor Triatriatum Sinister
associated w/:
ASD
PHTN
complications:
CHF
d/t increased pressure in LA and PA w/ MS
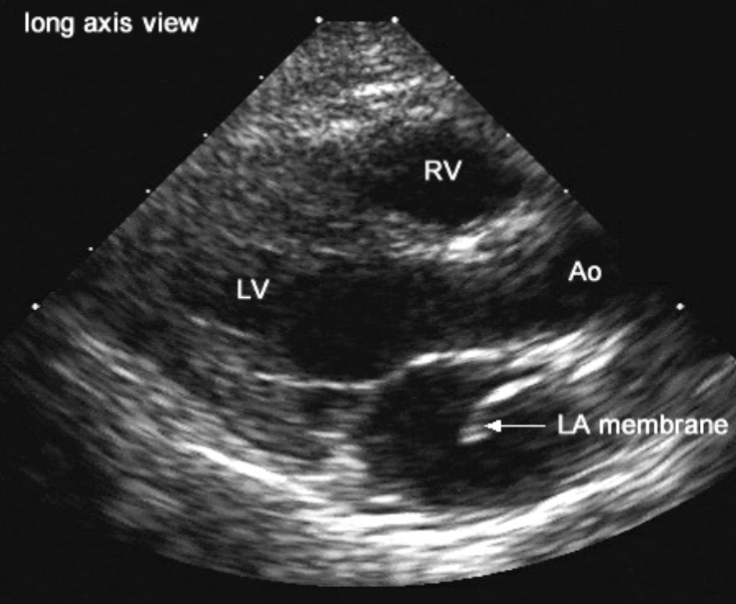
Cor Triatriatum Sinister 2D findings:
best views:
PLAX / A4C
see membrane within LA cavity
Kawasaki’s DIsease
acute febrile vasculitis
thought to be an autoimmune disorder
1800 cases/yr
50% of pt’s are <2 y/o
affects ~6 months - 5 yrs
only 20% have CA involvement
Kawasaki’s Syndrome findings:
Aneurysmal Coronary Arteries
may be lined w/ thrombus
>3mm <5 y/o
>4mm >5 y/o
may affect myocardial perfusion
causes WMA’s
MR + AI
Kawasaki’s Disease symptoms:
FUO lasting > 5 days
unresponsive to antibiotics or acetaminophen
swollen cervical lymph nodes
body rash
erythema of hands and feet
skin peeling occurs within 2-4 weeks
swollen, cracked, reddened lips
strawberry tongue**
Kawasaki’s Disease clinical signs:
elevated C-reactive protein, alpha-1 antitrypsin protein, and WBC
proteinuria
Conotruncal Defects
failure of conotruncal septation may result in following CHD’s:
TOGV
ToF
Pulmonary Atresia w/ VSD
DORV w/ VSD
truncus arteriosus
More common in infants born to DM mothers
Cyanotic Heart Disease
cyanosis may be present in many CHD’s
blue discoloration of mucus membranes, skin, lips, nailbeds, etc
cyanosis that is cardiac in origin will:
increase w/ crying
not be affected by O2 administration
these lesions allow systemic venous blood to mix w/ the systemic arterial circulation
Congenital defects that most commonly cause cyanosis:
TOGV
ToF
Pulmonary Atresia
Tricuspid Atresia
TAPVR
Truncus Arteriosus
Single Ventricle / Atrium
Hypoplastic Lt Heart
DORV
Ebstein’s Anomaly
d/t R → L ASD
What is one method to treat cyanotic heart disease?
give Prostaglandin to improve systemic O2 saturation
dilates the:
PDA
maintains a shunting source
Pulm arterioles
systemic vessels
Transposition of Great Vessels (TGV)(TGA)(TOGV)
Dx when the Ao is anterior to the Pulm A
PSAX
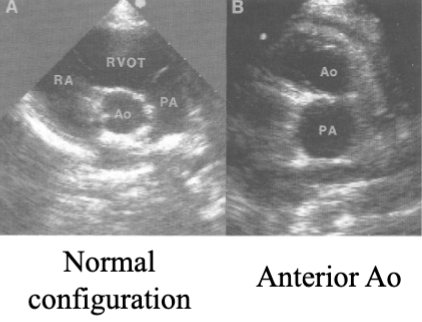
2 types of TOGV:
D-Transposition
D = dextro
latin for “right”
L-Transposition
L = levo
latin for “left”
D-Transposition of the Great Vessels (DTGV)
there is one wrong connection / mismatch
correct atrium is connected to correct ventricle
the wrong great vessel is attached to each ventricle
aka complete transposition
unsaturated bl→RA→RV→ Ao
saturated bl→LA→LV→PA
associated w/ cyanosis
must have large ASD, VSD, or PDA to survive
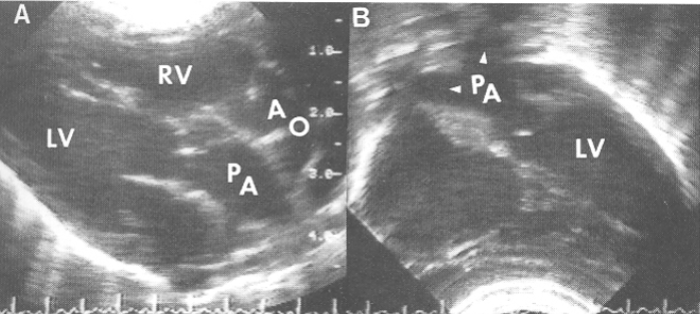
What type of open defect is DTGV most associated with?
membranous VSD
DTOGV surgical tx:
atrial switch
mustard or senning repair
re-direct atrial blood to exit correct great vessel
jatene repair
switches the great vessels
Helpful hints to remember D-transpos:
D = death
must have surgery to switch/redirect to survive
L-Transposition of the Great Vessels
2 wrong connections
atria are connected to the wrong ventricles
ventricles are connected to the wrong great vessels
overall connection is “correct”
“Congenitally Corrected”
“Double Discordance”
unsaturated bl→RA→LV→PA
saturated bl→LA→RV→Ao
LTOGV findings:
Lt A-V valve is the morphologic TV
Ebsteinization often present
not associated w/ cyanosis
less common than D-transposition
LTGV is associated w/:
Ao Coarctation
EKG abnormalities
conduction system in abnormal location
Tetralogy of Fallot (TOF)
Combination of 4 congenital lesions:
membranous VSD
anterior + rt displacement
overriding Ao (over the IVS)
may be infundibular / subinfundibular stenosis
usually d/t posterior malalignment of IVS from overriding Ao
PV or PA may be atretic
RVOTO / PS
RVH
d/t obstruction
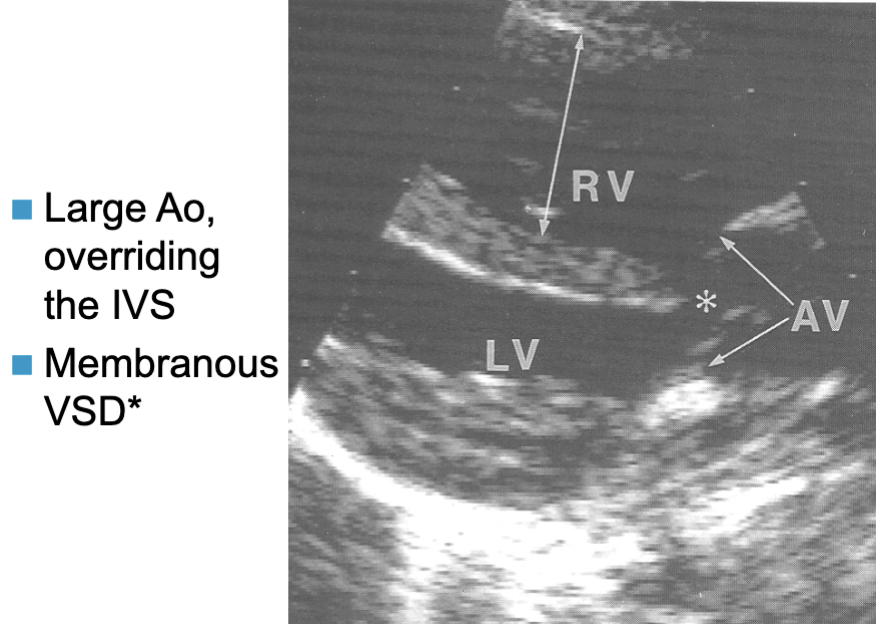
Tetralogy of Fallot key findings:
membranous VSD
overriding Ao (over the IVS)
RVOTO / PS
RVH
TOF blood flow:
since there is RVOTO + large membranous VSD, flow from either ventricle may exit via the Ao
this results in:
cyanosis
R → L shunt
also depends on the amount of resistance met in each great vessel
TOF 2D findings:
PLAX
overriding Ao
VSD
RVH
PSAX
VSD (mid)
PS/RVOTO (base)
TOF Dp findings:
of a subvalvular obstruction
dynamic obstruction → peaks in late systole
dagger shaped spectral trace
PW to localize region of stenosis
CW for max pressure gradient across the stenosis
TOF chest x-ray findings:
“boot shaped” heart
rt sided Ao arch
What is TOF associated with?
CA anomalies**
LAD arising from the RCA and crossing over the RVOT
single CA where a major branch crosses the RVOT
digit clubbing
cyanotic nail beds
Pulmonary Atresia
atresia of the PV / PA
no pulmonary blood flow exists
except through:
PDA
systemic to pulm A collaterals
“bronchiolar collaterals”
arise from the Desc Ao
poor prognosis
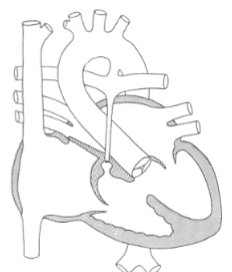
What is Pulmonary Atresia associated with?
Tetralogy of Fallot
Turner’s Syndrome
Noonan’s syndrome
What are the 2 types of Pulmonary Atresia?
VSD
intact IVS
VSD Pulm Atresia
similar to severe TOF
all ventricular blood leaves via the Ao
causes cyanosis
pulm flow is provided through PDA
Intact IVS (IIVS) Pulm Atresia
associated w/:
TV atresia
creates rt → lt ASD
RV hypoplasia
RV Hypoplasia
RV underdevelopment
due to compromised RV flow in fetal life
caused by:
TV atresia / stenosis
PV/PA atresia w/ IIVS
results in a small noncompliant RV that contracts minimally
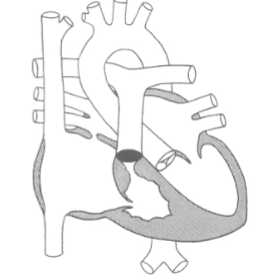
The 2 types of Anomalous Pulmonary Venous Return include:
Partial (PAPVR)
Total (TAPVR)
Partial Anomalous Pulm Venous Return (PAPVR)
some Pulm V’s drain into venous structures and return blood to the RA
frequency of occurence in order:
RPV → SVC
RVP → RA
RPV →. IVC
LPV → Coronary Sinus
associated w/ Sinus Venosus ASD (15%)
Total Anomalous Pulm Venous Return (TAPVR)
all 4 Pulm V’s drain into the RA or to one of the systemic veins
venous connection to RA
venous connection “above heart”
venous connection “below diaphragm”
critical need for a large ASD
so blood can reach the left heart
TAPVR venous connection “above heart” connects to:
SVC
azygous V
Lt innom V
Coronary Sinus
TAPVR venous connection “below diaphragm” connects to:
Portal V
Ductus venosus (PV to IVC to bypass liver)
Persistent Trucus Arteriosus
one large great vessel
carries both RVOT and LVOT
serves as both Ao and MPA
Rt + Lt PA’s usually arise from truncus
has a single common S-L Valve
abn w/ >3 cusps
usually incompetent
loud diastolic murmur
associated w/ VSD
both ventricles eject bl to truncus via large VSD
Depending on PA’s size, PTA will demonstrate:
diminished pulm blood flow
causes cyanosis
increased pum bl flow
causes CHF
Truncus Arteriosus anatomy:
1 - PA arises from truncus + shares common valve annulus
2 + 3 - PA branches arise separately
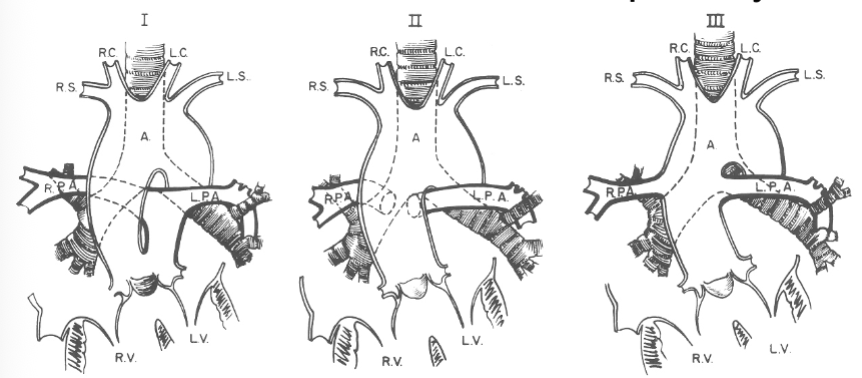
Truncus Arteriosus 2D findings:
PLAX
large overriding truncal root
VSD
PSAX
# of leaflets present
absence of MPA is its usual location
examine truncal root for PA origin
Truncus Arteriosus surgical tx:
Rastelli Procesure
dacron conduit w/ a prosthetic valve b/w the RA and PA
Single Ventricle aka:
double inlet LV
common ventricle
univenticular heart
Univentriular heart
both A-V valves connect to a single ventricular chamber which then directs blood to both great vessels
volume of blood flow to each GV is determined by the reisstane in each arterial circuit
2D findings of a single ventricle:
absence of IVS
best view → A4C
Double Outlet RV (DORV)
both great vessels arise from the morphologic RV
determine great vessel relation to each other
normal
PA ant + left of the Ao
side-by-side
in save TRV plane
dextromalposition
Ao ant + to the rt
levomalposition
Ao ant + to the lt
What is DORV always associated w/?
VSD
sole outlet for the LV
usually large
most often membranous
DORV is often associated w/:
ASD
PDA
MV abnormalities
LVOTO
pulm stenosis and/or RVOTO (50%)
causes diminished pulm blood flow
causes cyanosis
those w/o RVOTO will have increased pulm blood flow
causes CHF
What is the most severe form of LVOTO?
Hypoplastic Left Heart Syndrome
Hypoplastic Left Heart Syndrome
small underdeveloped LV w/ poor contractility
may be caused by:
Ao atresia
Ao hypoplasia
severe AS
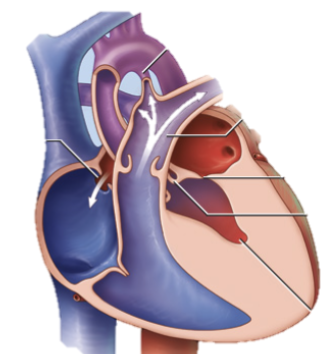
Hypoplastic Left Heart Syndrome includes:
MV + AoV atresia
endocardial thickening
small LA
What does HLHS have to be associated with?
PDA
pt given prostaglandins to maintain patency
blood shunts from MPA directly to desc Ao to maintain systemic flow
rt → lt shunt
will observe retrograde flow in asc ao
must feed CA’s
Hypoplastic Left Heart Syndrome is also associated w/:
RVE
small Ao root (usually <5mm)
dilated PA
HLHS prognosis:
poor → d/t organ hypoperfusion
HLHS Tx : Norwood Procedure
concerts RV into main pumping chamber
Blalock-Taussig shunt
shunt inserted btwn reconstructed Ao and MPA
supplies blood t lungs
Glenn operation
SVC anastomosed w/ MPA (bi-directional Glenn)
BT shunt removal
initial separatio of pulm _ systemic circulations
Fontan shunts
IVS anatomoses w/ MPA
Pulm A recives all systemic venous return
all systemic blood flow originates from RV