Orgo II Exams 2-3 Reagents
1/115
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
116 Terms
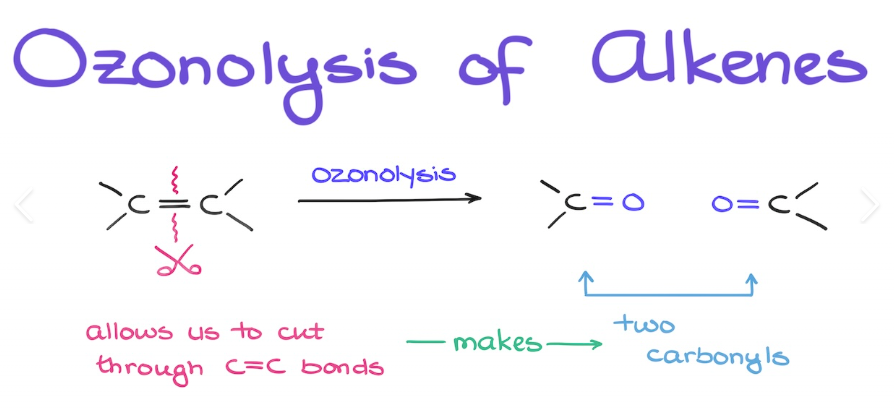
What Synthesis of alkene uses these conditions? 1) O3, -78* C 2) H2O, Zn
Ozonolysis of Alkenes
What are the reagents/results of Ozololysis of alkenes?
1) O3, -78* C 2) H2O, Zn; The double bond of the alkene breaks, leaving each carbon in the double bond being a carbonyl (C=O)
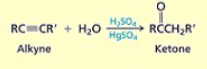
What synthesis of alkynes uses these conditions? H2O; H2SO4+HgSO4
Hydration of Alkynes
What are the conditions and results of hydration of alkynes?
H2O; H2SO4+HgSO4
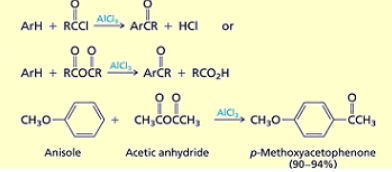
What synthesis of arenes uses these conditions? RCOCl, AlCl3
Friedal-crafts acylation of arenes
What are the conditions and results of friedal-crafts acylation of arenes?
RCOCl, AlCl3
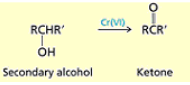
What happens when you combine 2nd degree alcohols with PCC, CH2Cl2 or Na2Cr2O7, H2O, H2SO4
2nd degree alcohol → a ketone.
What happens when you combine 1st degree alcohols with PCC, CH2Cl2?
You get aldehydes!
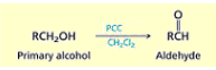
What reagents are used and what is the effect of reduction of aldehydes and ketones to hydrocarbons?
Wolff-Kishner reduction: H2NNH2, KOH, heat
Clemmensen reduction: Zn(Hg), HCl
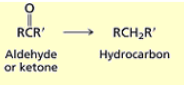
What happens when you add Zn(Hg) and HCl to an aldehyde or ketone?
Clemmensen reduction
What happens when you add H2NNH2, KOH, and heat to an aldehyde or ketone?
Wolff-Kishner reduction
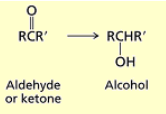
What happens when you add NaBH4, CH3OH or 1) LiAlH4 2) H3O+ to an aldehyde?
Aldehydes are reduced to primary alcohols
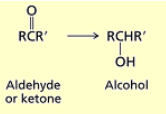
What happens when you add NaBH4, CH3OH or 1) LiAlH4 2) H3O+ to an ketone?
Ketones are reduced to secondary alcohols
What happens if you add 1) RMgBr 2) H3O+ or 1) RLi 2) H3O+ to formaldehyde?
Formaldehyde is converted to primary alcohols.
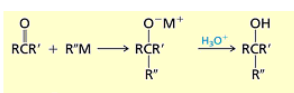
What happens if you add 1) RMgBr 2) H3O+ or 1) RLi 2) H3O+ to aldehydes?
Aldehydes are converted to secondary alcohols.
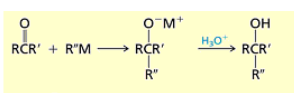
What happens if you add 1) RMgBr 2) H3O+ or 1) RLi 2) H3O+ to ketones?
Ketones are converted to tertiary alcohols
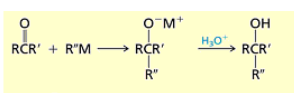
What is the result of adding 1) NaCN 2) HCl to an aldehyde or ketone?
What happens when you add ROH, H + (alcohol and acid catalyst) to an aldehyde?
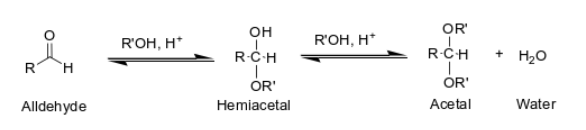
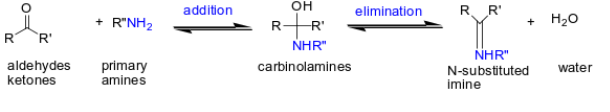
What happens when you add RNH2, H + to an aldehyde or ketone?
imine formation
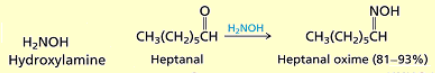
What happens when you add hydroxylamine (H2N-OH) to an aldehyde or ketone?
Oximes are formed.
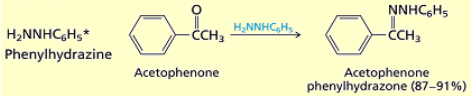
What happens when you add hydrazines (R-NH-NH2) to an aldehyde or ketone?
Hydrazones are formed.
What happens when you add R2NH, H + (Secondary amines (R2NH)) and aldehydes or ketones?
carbinolamines are formed
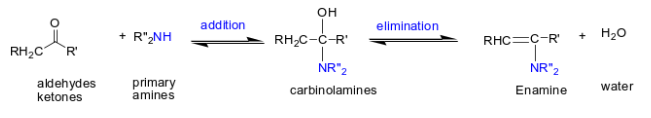
What happens when you add Ph3P(+) (a phosphorus -ylide) and (-)CR2 to aldehydes or ketones?
Alkene is formed.
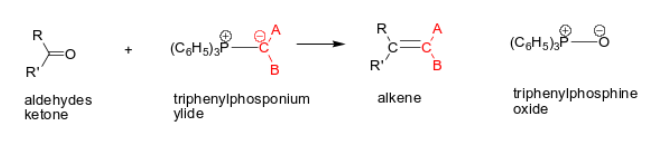

What happens when you add this molecule (C6H5CO2OH) to ketones?
Baeyer-Villiger oxidation: Oxygen insertion occurs between the carbonyl carbon and the larger of the two groups attached to it.
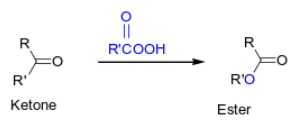
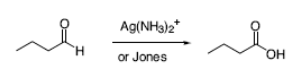
How do you oxidize an aldehyde to a carboxylic acid?
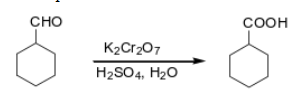
What are the grignard reagents?
Grignard reagents are organomagnesium compounds with the general formula RMgX, where R is an organic group and X is a halogen.
What happens when you add grignard reagents and carbon dioxide together?
you get carboxylic acids
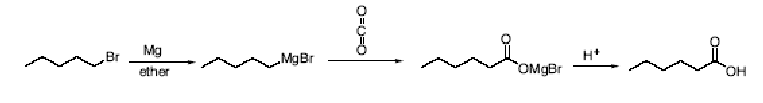
What happens when you oxidize alkylbenzenes?
A primary or secondary alkyl side chain on an aromatic ring is
converted to a carboxyl group by reaction with a strong oxidizing agent such as
potassium permanganate or chromic acid.
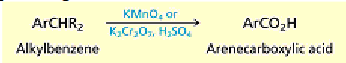
How do you oxidize alcohols into carboxylic acids?
Potassium permanganate and chromic acid convert alcohols to carboxylic acids by way of the corresponding aldehyde.
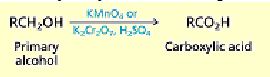
How do you convert aldehydes to carboxylic acids?
Aldehydes are particularly sensitive to oxidation and are converted to
carboxylic acids by a number of oxidizing agents, including potassium permanganate,
chromic acid and Tollens reagent.

What is the product of a reaction of an aldehyde and this reagent arrow?
Carboxylic Acids
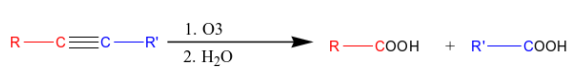
What happens when you add 1) O3 and 2) H2O to an internal alkyne?
Carboxylic acid is formed.
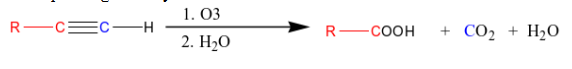
What happens when you add 1) O3 and 2) H2O to an terminal (or marginal) alkyne?
you yield water and carbon dioxide.
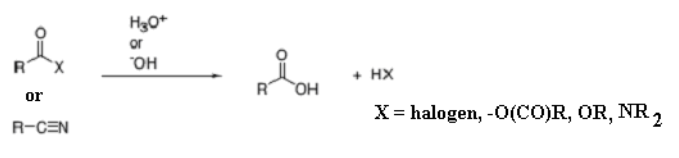
What happens when you use this arrow (water in the presence of an acid or base) on any carboxylic acid derivatives?
Hydrolysis. You get carboxylic acids.
What happens when you mix SOCL2 with carboxylic acids?
Acyl Chlorides
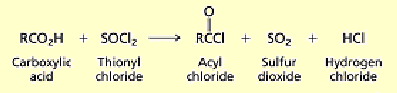
What happens when you add 1) LiAlH4, diethyl ether 2) H2O to a carboxylic acid?
Lithium aluminium hydride reduction, reduced to primary alcohol
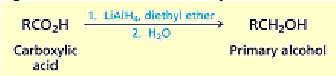
What happens when you add an acid catalyst (H+) to carboxylic acids?
Esterification: The reaction is an equilibrium process but can be driven to favor the ester by removing
the water that is formed.
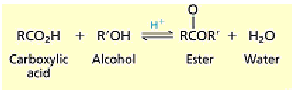
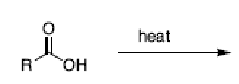
NR. Carboxylic acids are usually very stable
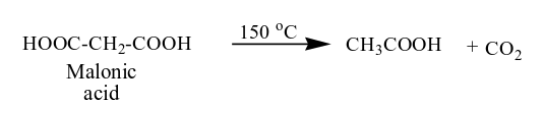
What happens in the reaction between malonic acid and heat?
one carboxylic acid replaces with a H and you yield CO2.
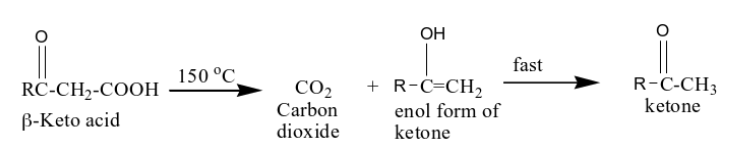
What happens when you react a beta-keto acid with heat?
you yield a ketone
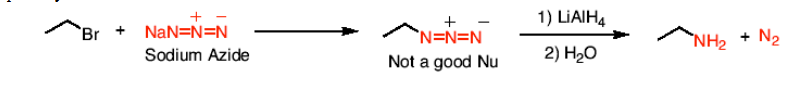
How do you synthesize primary amines using nucleophilic substitution?
with sodium azide and using 1) LiAlH4 2)H2O to reduce to a primary amine.
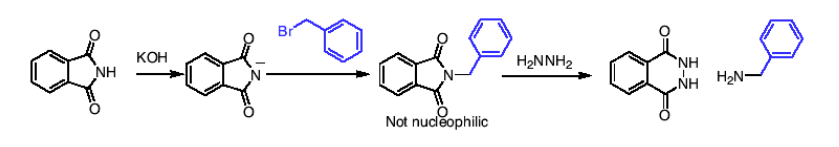
How do you synthesize primary amines using Gabriel synthesis?
1) Phthalimide deprotonated (using KOH) and nucleophile attacks the (-) on the N and attaches.
THEN
2) H2HHH2 to release the final amine
What is the general way to synthesize amines?
You need 1) A nitrogen nucleophile that can only be alkylated once, or alternatively a higher oxidation state of nitrogen can be reduced to a primary amine.
What happens in the reaction of toluene (benzene with one CH3 group), and HNO3, H2SO4?
You nitrate the ring at the para or ortho position.
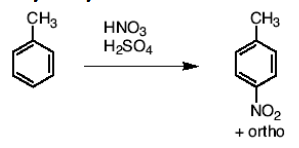
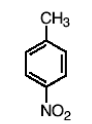
What happens in a reaction to an aromatic nitro group and 1) Fe, HCl (or Sn and HCl) and 2)H2O?
How do you react a nitrile to form a primary amine?
Reduce using 1) LiAl4 and 2) H2O
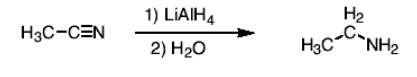
How do you react a ketone or aldehyde to form a primary amine?
NH3 and a reducing agent (NaBH3CN)
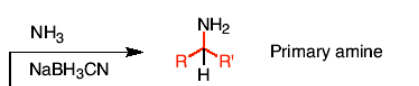
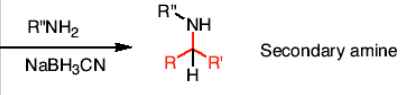
How do you react a ketone or aldehyde to form a secondary amine?
R’’NH2 and a reducing agent (NaBH3CN)
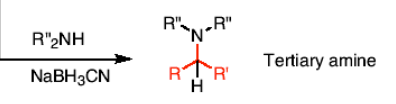
How do you react a ketone or aldehyde to form a tertiary amine?
R’’2NH and a reducing agent (NaBH3CN)
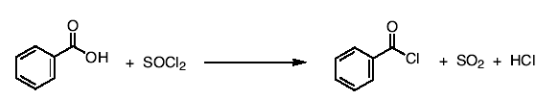
How do you synthesize acid chlorides from carboxylic acids?
you add SOCl2
What do acid chlorides react with?
All nucleophiles, they are the most reactive of carboxylic acid derivatives.
How do you synthesize anhydrides from acid chlorides?
Add a carboxylic acid and a base, the base deprotonates the OH and then that new (-) attacks the Cl as the nucleophile
How do you get a carboxylic acid from an acid chloride?
You add H2O (hydrolysis) and yield carboxylic acid and HCl
How do you replace the Cl in an acid chloride with a nucleophile?
with a nucleophile + H and a base to deprotonate that
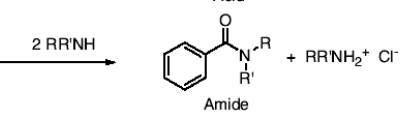
How do you get an amide from an acid chloride?
you add 2RR’NH
How do you synthesize anhydrides from carboxylic acids? Two ways, cyclic and non-cyclic
1) You add an acyl chloride and pyridine (a 6-member ring with N in it and a base) to neutralize the HCl that is a product in the reaction. (-OH deprotonates and Cl attacks, leaving just an O connecting the two. This can be used for R and R’ being either the same or different.
or
2) Cyclic anhydrides in which the ring is five- or six-membered are sometimes prepared by
heating the corresponding dicarboxylic acids in an inert solvent
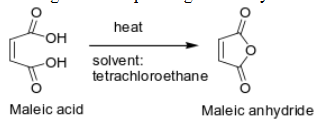
What happens in a reaction between an alcohol and an anhydride?
One side is a carboxylic acid and one side is a ester (alcohol deprotonates, H attacks center O and forms carboxylic acid, then the O- from the alcohol attaches where the center O was)
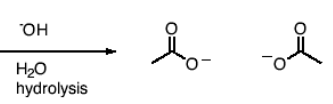
What happens in a reaction between an anhydride and H2O?
Breaks into carboxylic acids at center O. (one side takes O from anhydride and proton from H2O and the other side takes the -OH from H2O).
What happens in a reaction between an anhydride, a base (OH-) and H2O?
Hydrolysis. Anhydride breaks at center O, and leaves O- on each carbon attached to the center O.
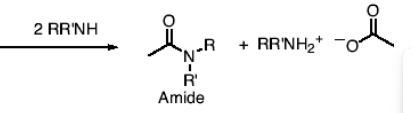
What happens in a reaction between an anhydride and 2RR’NH?
You form an amide and
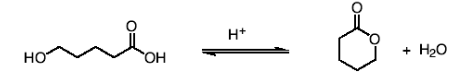
How do you form lactones and what reactions do they go through?
The mechanism for formation is identical to the esterification mechanism. Lactones undergo all the
same reactions as esters.
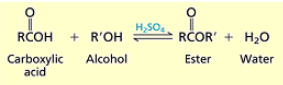
How do you synthesize esters from carboxylic acids?
Carboxylic acid + alcohol + H2SO4. (R group on alcohol replaces H on carboxylic acid) you yield water
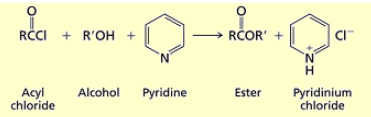
How do you synthesize esters from acyl chlorides?
you add an alcohol and pyridine (base to deprotonate the alcohol) R’O on alcohol replaces Cl.
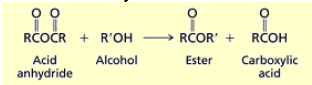
How do you synthesize esters from carboxylic acid anhydrides?
You add an alcohol and an acid or base to catalyze. split at O, attach R’O from alcohol. there
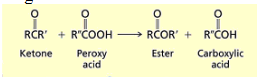
What happens when you add a peroxy acid to a ketone?
You yield an ester and a carboxylic acid. O inserts between the carbonyl and the largest R group.
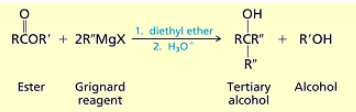
What happens when you add an ester, two equivalents of grignard reagent, 1. diethyl ether, and 2. H2O
You get tertiary alcohols.
first one R’’ from grignard attacks carbon and
then the OR group leaves
then another grignard attacks
then the O protonates and you get tertiary alcohol
What happens in a reaction between an ester and LiAlH4?
reduction, LiAlH4 cleaves and gives you two alcohols. RO leaves and is protonated, then carbonyl group becomes -OH
What happens when you treat an ester with an aqueous acid (eg. H2O and H3O+)
you get an alcohol and a carboxylic acid. -OR is replaced with -OH and -OR is protonated to form an alcohol.
TO MAKE THIS IRREVERSABLE, ADD NaOH first.
What happens to esters under basic conditions?
Saponification. you get glycerol and a fatty acid salt. In an example using NaOH as the base plus H2O, the OR of the ester leaves and is protonated. -ONa replaces it on the center carbon. (more intermediates than that but that is the effect on the molecules).
REMEMBER: The last step, when the H on the carboxylic acid intermediate leaves and protonates the alcohol is IRREVERSABLE. this is important because you can do this first (using NaOH) in hydrolysis of esters using aqueous acid to make it not possible to revert back to an ester.
What happens when you react an ester with ammonia (NH3)?
you yield an amide and an alcohol. OR group leaves and is protonated, and NH2 adds to carbon.
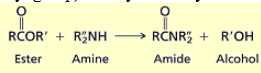
What happens when you react an ester with primary or secondary amines?
you yield amides and alcohols. OR group leaves and is protonated with H from amine and replaced with NRR from amine.
What reactions do thioesters go through?
The same reactions as esters, but faster because the S stabilizes the carbonyl less than the O from an ester.
How do you react amides to form carboxylic acids?
They are hard to hydrolyze, so we use a strong acid or base + heat.
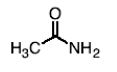
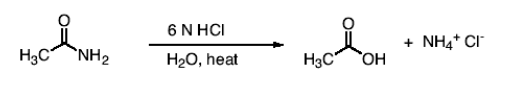
What happens in the reaction between an amide and 6N HCl, H2O, and heat? What does 6N mean?
You form a carboxylic acid (NH2 leaves and -OH comes in) and NH4+ Cl- as product.
6N basically means it’s a strong acid, which you need to hydrolyze amides. N means Normality or a measure of the number of H+ ions the solution can provide.
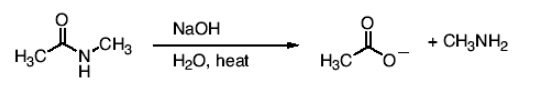
What happens in the reaction between an amide and NaOH, H2O, and heat?
You get unprotonated carboxylic acid (conj. base), and R-NH(-) leaves and is protonated immediately to form R-NH2
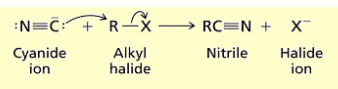
How do you synthesize nitriles from alkyl halides?
Add cyanide. you yield halide anion and nitrile.
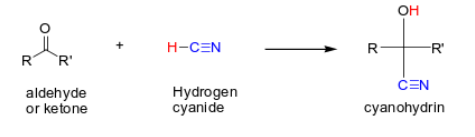
How do you replace the carbonyl with a nitrile and -OH group in aldehydes and ketones?
You add HCN. HCN dissociates and CN(-) attacks carbonyl, then H+ protonates it to an -OH. you form cyanohydrin. (C=O → C-CN and C-OH)
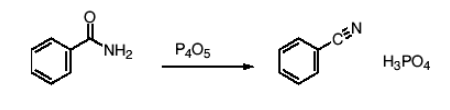
How do you synthesize nitriles from amides?
Dehydration. You replace -CONRR and replace with -C≡N. You can form both aryl and alkyl nitriles by adding a dehydrating agent. (P4O5, SOCl2, POCl3, PCl5 are the most common).
What happens to a nitrile in basic or acidic conditions? (plus, H2O and heat)
hydrolysis
in acidic: you yield carboxylic acid and NH4+
in basic: you yield carboxylic acid conj. base and NH3
What happens to a nitrile when you add a grignard reagent (R’MgX), 1) Diethyl ether, 2) H3O+ and heat?
IN GENERAL: R-C≡N group is replaced with R-CO-R’
R from Grignard attacks center carbon and one of the bonds from C≡N breaks and N protonates.
then H3O+ and heat gives a ketone. (-NH is replaced with -OH)
In a tautomerization reaction of aldehydes and ketones, which product is usually preferred?
They are in equilibrium, but generally the keto form is preferred because C=O is stronger than C=C.
What happens in a reaction between an aldehyde and itself under basic conditions?
You get an aldol (a molecule with an alcohOL and an ALDehyde)
OH⁻ → remove α-H → enolate → attack → protonate
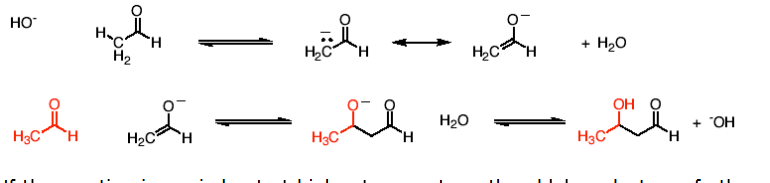
What happens in a reaction where an aldol (still in presence of base) is heated?
aldol condensation: Aldol + heat → lose H₂O → form C=C between α and β carbons
What happens in the aldol reaction of ketones?
Ketones also have acidic α-hydrogens and electrophilic carbonyl carbons, so they can undergo aldol reactions as well. In the case of ketones, however the equilibrium favors the ketones over the aldol product.
What are the conditions for the reaction between two different aldehydes giving primarily one product instead of 4? (Practical mixed/crossed aldol reactions).
A crossed aldol reaction can give primarily one product if:
• One reactant does not have any α-hydrogens.
• One of the reactants is more reactive toward nucleophilic addition than the
other
If the conditions are correct, what happens in a reaction between two different aldehydes?
Crossed aldol: one forms enolate → attacks the other → C–C bond → β-hydroxy aldehyde
How do you form beta-ketoesters using two esters?
Claisen condensation: 2 esters + matching base → β-ketoester (then H₃O⁺) Strong base makes enolate → enolate attacks aldehyde → alcohol forms → heat eliminates water → α,β-unsaturated carbonyl
What happens in a mechanism with a diester, 1) NaOMe and 2) H3O+?
Dieckmann reaction (intramolecular Claisen): forms a ring β-ketoester
What happens in a mixed Claisen condensation with two different esters (NaOEt, then H₃O⁺)?
β-ketoester forms (if only one ester has α-H)
Mechanism (what to draw):
Base (NaOEt) removes α-H → enolate forms
Enolate attacks carbonyl of second ester → C–C bond forms
Tetrahedral intermediate collapses → –OR leaves
Enolate forms again (drives reaction)
H₃O⁺ protonates → β-ketoester
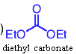
What happens when a ketone reacts with diethyl carbonate (NaOEt, then H₃O⁺ )?
β-ketoester forms
What happens when a ketone reacts with LDA, then an alkyl halide (R–X)?
Alkyl group adds at α-carbon (C–C bond forms)
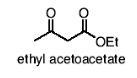
What happens in the acetoacetic ester synthesis? WHY IS THIS USEFUL
-COOEt is replaced by the R from alkyl halide.
This method is very useful for preparing methyl ketones. (if you need any R group off of a ketone)
One hydrogen in between the carbonyls is removed, then the carbon bonds to the alkyl group, forms a new C-C bond, and -COOEt leaves.

What happens in the malonic ester synthesis? (NaOEt, R–X, then NaOH, H₃O⁺, heat)
Substituted acetic acid forms (–CH₂COOH with R added)
What happens when an aldehyde or ketone reacts with X₂ under acidic conditions (H₃O⁺)?
α-halogenation (adds ONE halogen at α-carbon)
What happens when an aldehyde or ketone reacts with X₂ under basic conditions (OH⁻)?
α-halogenation (adds MULTIPLE halogens at α-carbon)
What happens when a carboxylic acid reacts with Br₂ (or Cl₂) and P or PBr₃ (or PCl₃)?
α-halogenated carboxylic acid forms (Br/Cl at α-carbon) COOH + X₂ + P → add X at α
What happens when a strong nucleophile (e.g., RLi, LiAlH₄) reacts with an α,β-unsaturated carbonyl?
1,2-addition → alcohol forms (adds to carbonyl carbon)
What happens when a weak nucleophile (e.g., CN⁻, amine, enolate) reacts with an α,β-unsaturated carbonyl?
1,4-addition → adds to β-carbon → carbonyl remains
What happens in a Michael addition?
Enolate adds to α,β-unsaturated carbonyl → 1,5-dicarbonyl
Mechanism (draw this)
Base makes enolate
Enolate does 1,4-addition (β attack)
Protonate → product (Two carbonyls separated by 3 carbons)