CH. 15 | Microbial Mechanisms of Pathogenicity
1/15
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
16 Terms
15 - 1 Identify the principal portals of entry
Pathogens enter the body through 3 primary avenues:
Mucous membranes
Skin
Parenteral route
Key Concept: Entering the body does not always guarantee disease. Many pathogens have a preferred portal of entry that is required for them to cause disease
Mucous Membranes
Most common pathway for bacteria and viruses
Respiratory Tract: Easiest and most frequent entry point — Microbes are inhaled via moisture drops or dust
COVID-19, Influenza, Tuberculosis, Measles
Digestive Canal: Entry via contaminated food, water, or fingers — Most microbes are killed by hydrochloric acid, but those that survive can CAUSE disease
Typhoid fever, cholera, Hepatitis A, giardiasis
Genital System: Sexually contracted — Pathogens may cross unbroken membranes or enter through small abrasions
HIV, Chlamydia, Syphilis, Gonorrhea
Conjunctiva: Membrane lining the eyelids and eyeballs
Pink eye
Skin
The largest organ in the body, and a primary defense
Unbroken Skin: Mostly impenetrable (impossible to pass through) by microbes
Openings: Pathogens can enter through hair follicles or sweat ducts
Direct Infection: Some fungi grow on skin keratin, and certain larvae (hookworm) can bore through intact skin
Parenteral Route
Occurs when pathogens are deposited directly into tissues beneath the skin or membranes because the barriers have been injured
Causes: Punctures, injections, bites, cuts, wounds, surgery, or skin splitting due to swelling or drying
Examples: HIV, Hepatitis viruses, Tetanus
15 - 2 Define ID50 and LD50
ID50 (Infectious Dose)
The number of microbes required to cause an active infection in 50% of a sample population
It measures the virulence (degree of pathogenicity) of a microbe
Lower ID50 = Higher Efficiency
High ID50 = Low Efficiency
LD50 (Lethal Dose)
The amount of a toxin or substance required to cause death in 50% of a sample population
It measures the potency of a toxin
Lower LD50 = More deadly
High LD50 = Less deadly
15 - 3 Using examples, explain how microbes adhere to host cells
To understand how microbes cause disease
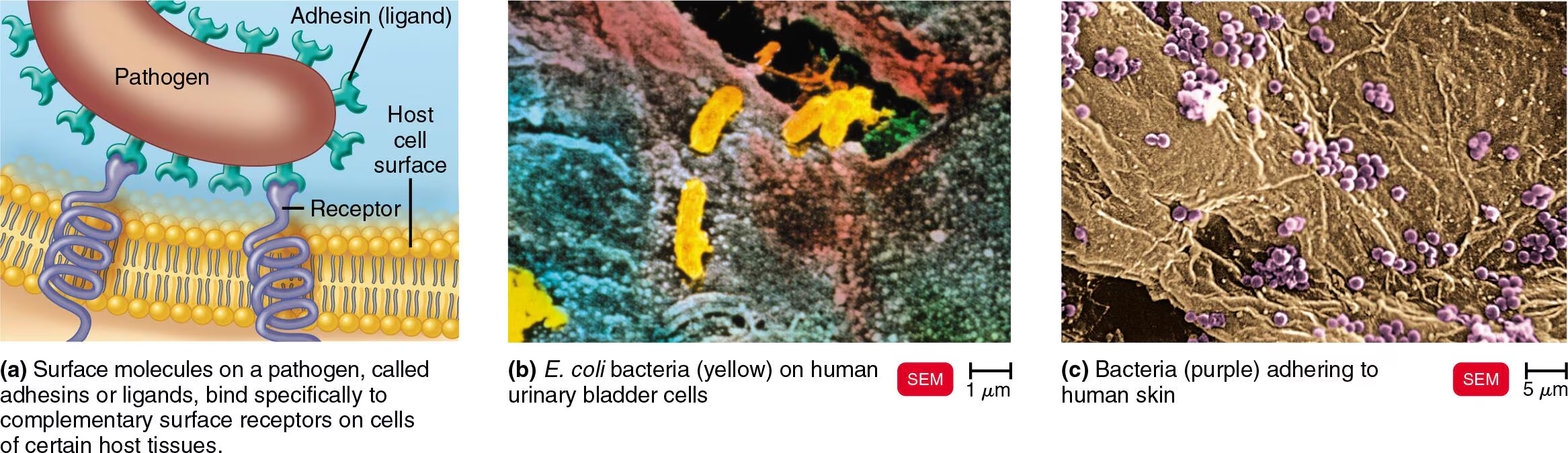
Adherence (Adhesion)
Microorganisms attach to surfaces, such as host tissues, using specialized molecules called adhesins
Attachment phase and is a necessary step for most pathogens to become harmful
Lock (body cells) & Key (pathogen) system
Mechanism: Adhesins & Receptors
Microbes physically bind to nearby cells using specific molecules
Adhesins (Ligands): These are surface molecules on the pathogen
Usually made of glycoproteins or lipoproteins
Receptors: These are complementary surface molecules on the host cell
Typically sugars, like mannose
Examples of Adherence in Action
Dental Plaque (Streptococcus mutans)
This bacterium uses an enzyme to turn glucose into a sticky substance called dextran
This forms a “sticky net” — glycocalyx, which allows the bacteria to cling to your teeth
Respiratory Infections (Influenza & COVID-19)
Influenza: Uses a spike protein called HA to bind to sialic acid on your lung cells
COVID-19: Uses its famous S (spike) proteins to attach to a specific receptor called ACE2 on human cells
Overall, this is an important concept because if we can figure out how to alter or block either the adhesin or the receptor, then we can prevent the infection from ever starting
15 - 4 Explain how capsules and cell wall components contribute to pathogenicity
To understand how microbes defend themselves
Capsules
A capsule is a sticky layer of glycocalyx material that surrounds the bacterial cell wall
It works by increasing virulence by impairing phagocytosis. Normally, your immune cells (phagocytes) wrap around a bacterium to destroy it. However, the chemical nature of the capsule makes the bacterium “slippery”, thus preventing the immune cell from sticking to it
However, to work around this. If your body produces antibodies against that specific capsule, the immune system can get ahold of the bacterium and destroy it
Examples: S. pneumoniae: Strains with capsules cause pneumonia; Strains without them are harmless because your body eats them immediately through phagocytosis
Cell Wall Components
Certain chemicals in the cell wall help bacteria stick to you and resist being digested
M Protein: Found on the surface and fimbriae of Streptococcus pyogenes
Function: It is heat-resistant and acid-resistant. It helps the bacteria attach to your cells and helps them resist phagocytosis by white blood cells
Opa Protein: An outer membrane protein used by N. gonorrhoeae
Function: It works with fimbriae to attach the bacteria firmly to host cells. Once attached, the host cell actually pulls the bacteria inside
Mycolic Acid: A waxy lipid found in the cell wall of Mycobacterium species, etc
Function: Even if a white blood cell manages to swallow the bacteria, the waxy mycolic acid prevents the bacteria from being digested
The bacteria can actually multiply inside the immune cell
15 - 5 Compare the effects of coagulases, kinases, hyaluronidase, and collagenase
To understand how bacteria use enzymes to spread or hide
Coagulases
An enzyme that turns fibrinogen (a blood protein) into fibrin (clotting threads)
They create a blood clot around the bacteria, and from here the clot acts like a fortress that protects the bacteria from phagocytosis and isolates them from other immune defenses
Produced by some species of Staphylococcus
Kinases
An enzyme that break down fibrin and digest clots
They dissolve the clots the body creates to “wall off” an infection, and the benefit of this is that it allows the bacteria to spread through the body instead of staying trapped in one spot
Fibrinolysis (Streptokinase)
Hyaluronidase
An enzyme that digests hyaluronic acid, a sugar that holds connective tissue cells together
They cause tissue blackening and allow microbes to spread from the initial site of infection to other places
Produced by Streptococci, etc
Collagenase
An enzyme that breaks down collagen, the main protein in the connective tissue of muscles and organs
They destroy the structural framework of tissues, facilitating the spread of gas gangrene
Produced by Clostridium species
15 - 6 Define and give an example of antigenic variation
Antigenic Variation
The process by which some pathogens alter their surface proteins (antigens) so that the body’s antibodies can no longer recognize or bind to them
Examples
Neisseria gonorrhoeae: This bacterium has several different copies of the Opa-encoding gene. It can express different antigens over time, making it very difficult for the immune system to clear the infection
Influenzavirus (The Flu): This virus constantly undergoes antigenic changes, which is why you need a flu shot every year, because the antibodies from last year wouldn’t be able to recognize this year’s version
15 - 7 Describe how bacteria use the host cell’s cytoskeleton to enter the cell
To understand how bacteria enter a cell
Mechanism (Hijacking Actin)
While some bacteria enter through simple attachment, others take a more active approach by manipulating the host’s internal “scaffolding.”
Invasins: These are surface proteins produced by certain bacteria (like Salmonella and E. coli) upon contact with the host cell’s plasma membrane that can rearrange nearby actin filaments of the cytoskeleton, allowing the bacteria to be engulfed by the host cell
Entry via “Membrane Ruffling”
When Salmonella makes contact, its invasins cause the host cell’s plasma membrane to look like the splash of a liquid hitting a solid surface
The “ruffle” is this disruption of the cytoskeleton that creates “ruffles” in the membrane
Macropinocytosis: The bacterium sinks into these ruffles and is engulfed by the host cell. This is essentially the cell “drinking” the bacterium by mistake
15 - 9 Describe the function of siderophores
Bacteria are living organisms that need nutrients to grow, and for most pathogenic bacteria, iron is the most critical resource.
Siderophores
Bacterial iron-binding proteins
How it works
They work by taking iron away from the host’s iron-transport proteins by binding the iron even more tightly
Once the siderophore has formed into an iron-siderophore complex, it binds to specific siderophore receptors on the surface of the bacterium
Then, the bacterium pulls the whole complex inside, releasing the iron into its own cytoplasm to power its growth and reproduction
Alternative Methods of Stealing Iron
Direct Binding: Some pathogens have receptors that bind directly to our iron-transport proteins or hemoglobin, taking the whole unit into the bacterial cell
Cell Murder (Toxins): When iron levels are low, some bacteria release toxins that kill host cells, thereby making their iron more accessible and available for the bacteria
15 - 10 Provide an example of direct damage, and compare this to toxic production
Direct Damage
This occurs when a pathogen physically destroys the host cell it is currently inhabiting or attached to
Mechanism: As pathogens multiply inside a host cell, the cell becomes crowded with metabolic waste and new microbes. Eventually the pressure causes the cell to rupture (lyse)
Once the cell bursts, the newly formed pathogens are released and can immediately infect neighboring cells
Examples include viruses, as well as intracellular bacteria and protozoa
Toxin Production
Toxins are poisonous substances produced by microorganisms that contribute significantly to their pathogenicity
15 - 11 Contrast the nature and effects of exotoxins and endotoxins
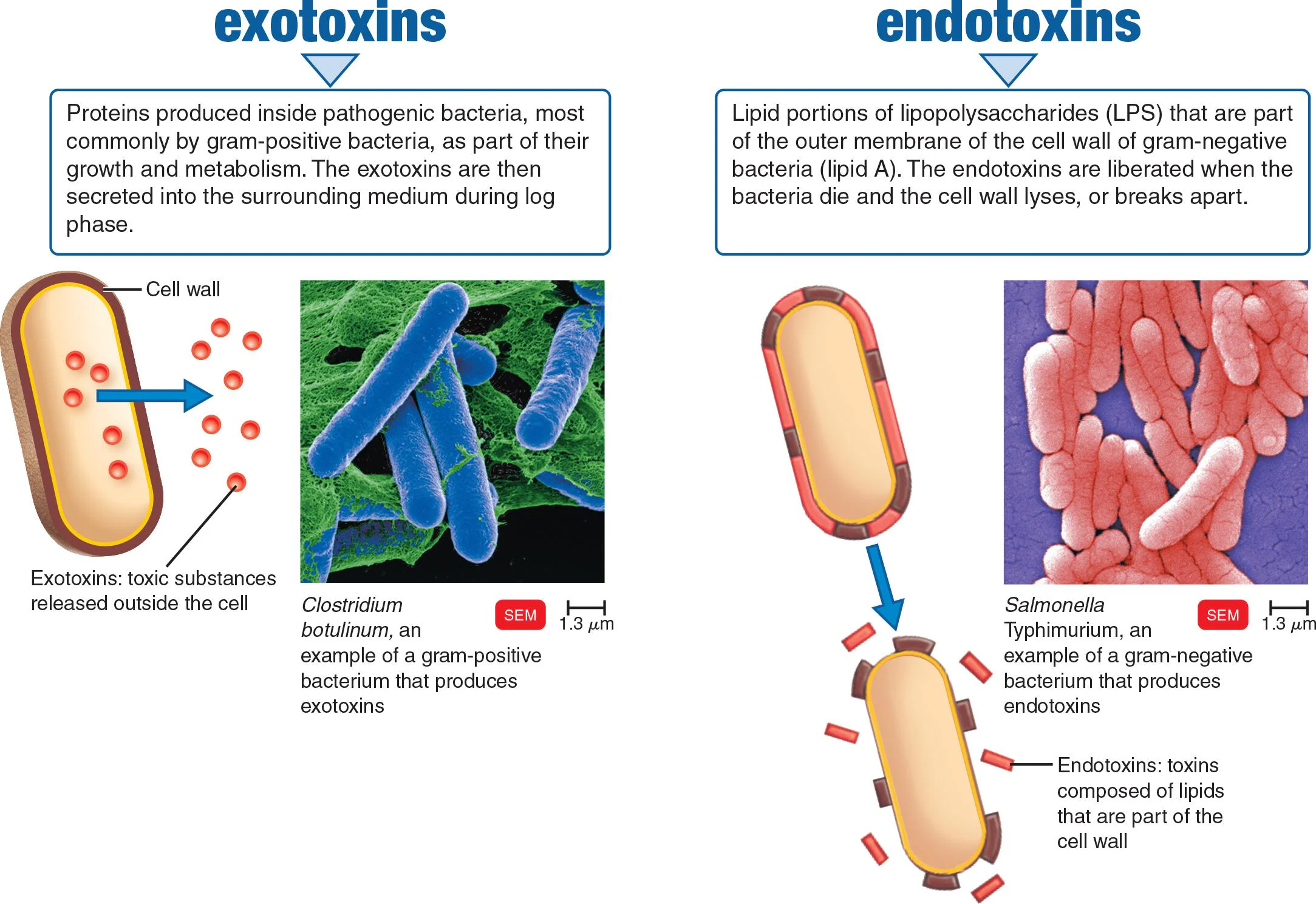
Exotoxins
A protein toxin released from living, mostly gram-positive bacterial cells
Highly specific — targets particular cells or metabolic functions
Extremely potent even in small amounts
Soluble in body fluids, easily spread through blood
The body produces antitoxins against them
Can be inactivated into toxoids for use as vaccines
Endotoxins
Part of the outer portion of the cell wall (lipid A) of most gram-negative bacteria: released on destruction of the cell
Lipid portions of lipopolysaccharides (LPS) — part of the cell wall
Produced only by gram-negative bacteria
Released when the bacteria die and the cell wall lyses
Less specific in their effects
Produce the same signs and symptoms, regardless of the species of the microorganism
15 - 12 Outline the mechanisms of action of A-B toxins, membrane-disrupting toxins, superantigens, and genotoxins
A-B Toxins
Bacterial exotoxins consisting of two polypeptides
Two components: A (active/enzyme) and B (binding)
B component binds to the host cell receptor
Toxin enters via receptor-mediated endocytosis
A and B are separated inside the cell
A component disrupts cell function, often by inhibiting protein synthesis
B component is recycled back to the cell membrane
Membrane-Disrupting Toxins
Cause lysis of host cells by disrupting plasma membranes, either by:
Forming protein channels in the membrane
Disrupting the phospholipid portion
Key Types:
Leukocidins — Kill pathogenic WBCs (neutrophils and macrophages) by forming protein channels; produced mainly by staphylococci and streptococci
Hemolysins — Destroy red blood cells via protein channels; streptococcal hemolysins are called streptolysins
Superantigens
An antigen that activates many different T cells, thereby eliciting a large immune response
Binds to proteins on macrophages, nonspecifically stimulating T cell proliferation
T cells release massive amounts of cytokines
Excessive cytokines cause fever, nausea, vomiting, diarrhea, shock, and even death
Genotoxins
A-B toxins that target DNA or RNA
Cause mutations, disrupt cell division, and may lead to cancer
Produced by gram-negative bacteria; Salmonella, and some E. coli
15 - 13 Identify the importance of the LAL assay
Limulus Amebocyte Lysate (LAL) Assay
A test to detect the presence of bacterial endotoxins
Detects endotoxins in drugs, medical devices, and body fluids (even minute amounts)
Critical because sterilized materials can still contain endotoxins when when no live bacteria are present
15 - 14 Using examples, describe the roles of plasmids and lysogeny in pathogenicity
Plasmids and Pathogenicity
Plasmids are small, circular DNA molecules that replicate independently from the main chromosome
Two Key Groups:
R (resistance) factors — carry antibiotic resistance genes
Virulence factors — carry genes that determine a microbe's pathogenicity
Examples of virulence factors encoded by plasmids:
Tetanus neurotoxin
Heat-labile enterotoxin (E. coli)
Staphylococcal enterotoxin D
Adhesins and coagulase (S. aureus)
Fimbria specific to enteropathogenic E. coli
Lysogeny and Pathogenicity
Bacteriophage — A virus that specifically infects and destroys bacteria
Prophage — A bacteriophage (virus) genome that has inserted itself into a host bacterium's DNA
Bacteriophages can incorporate their DNA into the bacterial chromosome, becoming a prophage — this state is called lysogeny
A change in microbial characteristics due to a prophage is called lysogenic conversion
Lysogenic cells are medically important because some bacterial pathogenesis is caused by the prophages they carry
Examples of toxins/virulence factors encoded by phage genes:
Diphtheria toxin
Botulinum neurotoxin
Erythrogenic toxins
Shiga toxin in E. coli O157
Cholera toxin — lysogenic phages in V. cholerae can even transmit the toxin gene to nonpathogenic strains, increasing the number of pathogenic bacteria
15 - 15 List 11 cytopathic effects of viral infections
Cytopathic Effects (CPE)
A visible effect on a host cell, caused by a virus, that may result in host cell damage or death
Cell junction disruption — junctions between cells are broken down (e.g., SARS-CoV-2 and Alphainfluenzavirus disrupt pulmonary alveoli junctions; cilia also stop moving)
Cytokine storm — excessive cytokine production increases inflammation, damaging tissues and organs, sometimes fatally (e.g., SARS-CoV-2, Alphainfluenzavirus)
Macromolecular synthesis stops — some viruses irreversibly halt mitosis within the host cell (e.g., Simplexvirus)
Lysosome release — host cell lysosomes release their enzymes, destroying intracellular contents and causing cell death
Inclusion bodies — viral nucleic acids or proteins accumulate in the cytoplasm or nucleus (e.g., Negri bodies in rabies); useful diagnostically
Syncytium formation — adjacent infected cells fuse into a large multinucleate giant cell (e.g., measles, mumps, common cold)
Altered cell function with no visible changes — host cell function is changed without visible morphological effects (e.g., measles virus reduces IL-12 production via CD46 receptor)
Antigenic changes on cell surface — viral proteins alter the cell surface, triggering host antibody response that kills the infected cell even if the virus is noncytocidal
Chromosomal changes — chromosomal breakage occurs; oncogenes may be activated or contributed by the virus, potentially leading to cancer
Cell transformation — cancer-causing viruses transform host cells into abnormally shaped cells that lose contact inhibition, leading to unregulated cell growth
Interferon production — infected cells produce alpha and beta interferons (coded by host DNA) which protect neighboring cells by inhibiting viral protein synthesis and triggering apoptosis of infected cells; however, most viruses can partially evade interferons by blocking their synthesis
15 - 16 Discuss the causes of symptoms in fungal, protozoan, helminthic, and algal diseases
Fungi
Generally lack well-defined virulence factors; toxic effects are indirect (fungus already growing in/on host)
Chronic infections (e.g., athlete's foot) can provoke allergic responses
Disease caused by toxin production
Can be caused by capsules, toxins, and allergic responses
Protozoa
Symptoms triggered by the presence of protozoa and their waste products
Some protozoa change their surface antigens while growing in a host, thus avoiding destruction by the host’s antibodies
Mechanisms vary by organism:
Plasmodium — invades and reproduces within host cells, causing rupture (malaria)
Toxoplasma — enters macrophages via phagocytosis, prevents acidification/digestion, survives and grows inside
Giardia — attaches via a sucking disc, digests host cells and tissue fluids
Some evade the immune system through antigenic variation:
Trypanosoma continuously switches surface antigens, staying ahead of antibody responses; can produce up to 1,000 different antigens, allowing infection to last decades
Helminths
Symptoms caused by the physical presence of the parasite
Use host tissues for growth or form large parasitic masses, causing cellular damage
Example: Wuchereria bancrofti blocks lymphatic circulation → accumulation of lymph plasma → grotesque swelling of limbs (lymphatic filariasis)
Metabolic waste products of the parasites also contribute to disease symptoms
Algae
Disease caused by neurotoxin production during harmful algal blooms that can cause paralysis when ingested by humans
15 - 17 Differentiate portal of entry and portal of exit
Portal of Entry
Routes through which microbes enter the body
Microbes tend to use a preferred route of entry specific to the pathogen
Portals of Exit
Routes through which microbes leave the body
Found in secretions, excretions, discharges, or shed tissue
The respiratory tract & digestive canal are the most common
Skin or wound infections