Microbio Exam 3 comprehensive study guide
1/84
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
85 Terms
Disinfectants
Destroys harmful microorganisms (99.9% of bacteria and viruses)
antiseptics
destroys harmful microorganisms from living tissue (mouth wash)
Sterilants
removes and destroys ALL microbial life through sterilization
Biocides
(germicide) are treatments to KILL microbes but not necessarily endospores.
Biostatics
inhibiting microbial and fungal growth
Bio. Safety level 1:
microbes that don’t cause many diseases, low risk microbes
Bio safety Level 2:
microbes are where they generally belong and have varying severity in diseases.
Bio safety level 3:
Microbes are indigenous or exotic and could potentially be lethal, spread through respiratory system
Bio. safety level 4:
Very hard to kill, lethal high-risk microbes
require treatment or vaccines
Dry heat
Kills by oxidation
Flaming is very common in sterilizing loops
incineration used to deal with lots of garbage
hot air steer is usually electrical reaches high temps
Boiling (moist heat)
denatures enzymes and proteins
easiest at home or industry method
Pasteurization
Heat treatment that kills harmful pathogens and spoilage microorganisms in food
enhances food safety and extends shelf life
Autoclave
Applies steam under pressure
combo of heat and pressure
Kills ALL organisms and endospores
Hospitals use strips to know if somethings sterile
Application of filtration
Passage of liquids and air through a filter
Used for heat sensitive materials
HEPA air filters can remove endospores, microbes, and viruses as air flows through
Membrane filters deal with larger units and use a vacuum to draw out the solution
Smaller membrane filters use syringes
What conditions affect the activity of a antimicrobial agent
Population size; larger vs. smaller populations
Composition of population; microorganisms and sensitivity to antimicrobial agents
Concentration or intensity of an antimicrobial agent; higher concentrations kill faster
Duration of exposure; longer exposure = more organisms killed
Temperature; higher temp usually increases killing
Local environment; many factors impact effectiveness depending on the thing (pH, organic matter conc.)
Surfactant
Surface active agents; group of chemical Compounds that lower the surface tension of water.
Major ingredients in soaps and detergents
Goal of antimicrobial drugs
synthetic substances made to interfere with microbial growth
Chemotherapy
the use of chemicals to treat disease
antibiotics
Substance made by a microbe that inhibits another microbe in small amounts
Selective toxicity
selectively finding and destroying pathogens without hurting the host
Spectrum of activity of antimicrobial drugs
Narrow spectrum of microbial activity: drugs affect a narrow range of microbial types
Broad spectrum antibiotic: affects a broad range of graph positive or gram-negative bacteria
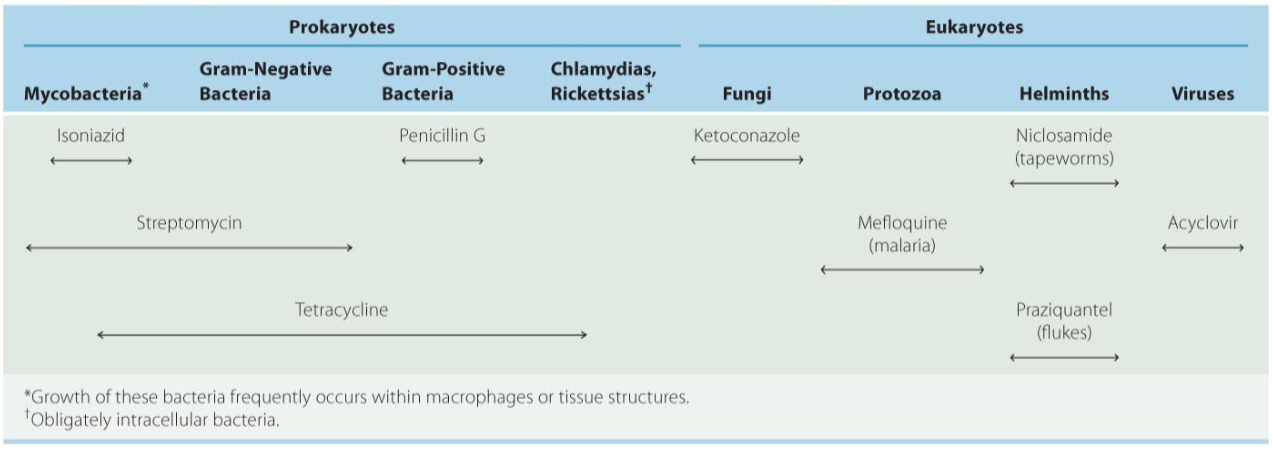
Compare regular toxicity to selective toxicity
Toxicity: side effects caused by drug
Selective toxicity: selectively kills or inhibits growth of microbial targets while causing minimal or no harm to the host
What is the significance of dosage?
amount of medication given during a certain time interval
ensures that optimum therapeutic drug levels are reached at the site of the infection without causing significant side effects
Whats the significance of the route of drug administration
Orally; pros are that patients can more conveniently take these drugs at home. Cons are that some drugs aren’t easily bodied by GI tract into bloodstream
Parenteral route (injection); pro is short time it takes to reach high concentrations in plasma. Is typically performed in health care settings.
What are factors and variables that can influence the side effects of a drug
Half life of drug: rate at which 50% of a drug is eliminated from plasma
Toxicity: side effects caused by drug
Selective toxicity: selectively kills or inhibits growth of microbial targets while causing minimal or no harm to the host
Bacteriostatic vs bactericidal activities
Bactericidal kills directly while bacteriostatic just inhibits growth
Synergism (+): 2 antibacterial drugs may be administered together to improve efficacy
Antagonism (-): can occur between 2 antimicrobials or between antimicrobials and non-antimicrobial used to treat other conditions. Interactions may cause loss of drug activity, decreased therapeutic levels due to increased metabolism and elimination, or increased potential for toxicity
How do drugs inhibit cell wall biosynthesis
cell wall made of peptidoglycan and proteins. Forming a beta-lactam ring stops peptidoglycan from being made
Natural penicillin; susceptible to penicillinases
Semisynthetic penicillin; contain chemically added side chains that make them resistant to penicillinases
How do drugs inhibit protein synthesis
targets different steps in bacterial translation.
Chloramphenicol: binds to 50S subunit of 70S ribosome to stop peptide bond formation. Can suppress bone marrow and affect blood cell formation
Tetracyclines: interfere with tRNA attachment to ribosomes
How do drugs inhibit membrane function
polypeptide antibiotics change membrane permeability. The membrane targeting mechanism isn’t a selective toxicity
How do drugs inhibit nucleic acid synthesis
Interferes with DNA replication and transcription. Stops DNA replication by inhibiting DNA polymerase and DNA helicase, and blocks transcription by inhibiting RNA polymerase
Not selective toxic for DNA, but is selectively toxic for RNA
How do drugs inhibit metabolic pathways
Antimetabolites compete with normal substrates for an enzyme.
Sulfanilamide competes with para-aminobenzoic acid (PABA), stopping the synthesis of folic acid
Explain drug resistance
Antimicrobial resistance happens when microbes develop the ability to defeat drugs designed to kill them
How do microbes use an efflux pump in drug resistance
transports antibiotics or other poisonous substances out of bacteria
uses transport proteins
builds resistance to multiple drugs at once
How do microbes use blocked penetration in drug resistance
just doesn’t let anything in
How do microbes use inactivation of enzymes in drug resistance
stops needed metabolic processes, which causes structural breakdown and death
How do microbes use target modification in drug resistance
changes the composition of structures like enzymes or ribosomes to stop antibiotics from binding
spontaneous mutations can change the targets of antibiotics and build drug resistance
Describe tests used to determine microbe susceptibility to an antibacterial drug.
Give drug in appropriate concentrations to destroy susceptible microbes
Give two or more drugs at same time
Use drugs only when necessary
Possible future solutions:
continued development of new drugs
use of bacteriophages to treat bacterial disease
How do microbes fight drugs
Microbes are constantly evolving
widespread clinical use of antibiotics has also provided selective pressure for microbes to evolve
overuse and misuse of antimicrobials,
inappropriate use of antimicrobials,
Sub-therapeutic dosing, and
patient noncompliance with the recommended course of treatment
once resistance originates in a population it can be transmitted to other bacteria
a particular type of resistance mechanism is not confirmed to a single class of drugs
Microbes in abscesses or biofilms may be growing slowly and not be susceptible
Resistance mutants arise spontaneously and are then selected
How do you test the effectiveness of an antimicrobial drug
Kirky-baur disc diffusion test: Paper disks with a chemotherapeutic agent are placed on agar containing the test organism
Zone of inhibition around the disk determines the sensitivity of the organism to the antibiotic
Dilution test measures the minimal bactericidal concentration of a drug
E test: gradient diffusion method that determines antibiotic sensitivity and estimates minimal inhibitory concentration
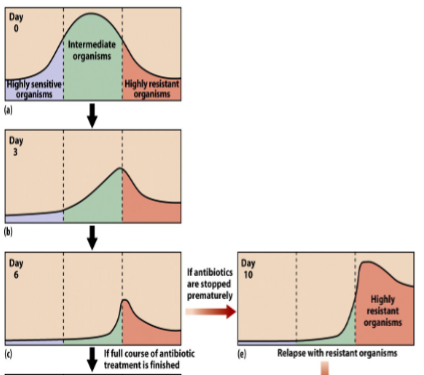
What happens if you stop an antibiotic prescription early?
It will leave the toughest microbes that were harder to kill remaining, and those microbes will come back with a resistance to the antibiotic and be tougher to kill
Disease
Any condition in which normal structure or functions of the body are damaged or impaired
infection by a pathogen,
genetics (as in many cancers or deficiencies),
noninfectious environmental causes, or
inappropriate immune responses
Infection
successful colonization of a host by a microorganism
Pathogenicity
potential ability to cause disease
it’s qualitative; either an organism can or cannot cause a disease
Virulence
disease producing power of an organism or the degree of pathogenicity when compared within a group
quantifies pathogenicity: Median infectious dose (ID) is the number of pathogen cells needed to cause active infection in 50% of inoculated animals.
Median lethal dose is number of pathogenic cells or amount needed to kill 50% of the infected animals
What features protect cells against phagocytosis (come back to)
Capsule production and proteases to protect against phagocytosis and destruction of antibodies
Use a variety of virulence factors.
What’re the 2 types of infectious diseases
disease caused by direct effect of a pathogen
Communicable: contagious disease easily spread from person to person
Non-communicable: infectious disease not spread from person to person
iatrogenic infectious diseases
contracted as result of medical procedure
wounds/flesh-eating Clostridium
Nosocomial infectious diseases
disease acquired in hospital settings
MRSA
Zoonotic infectious diseases
transmitted from animals → humans
rabies, yellow fever, flu
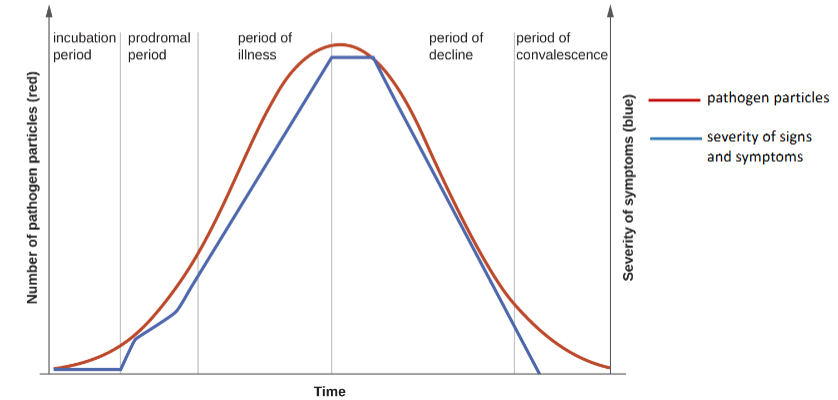
What’re the stages of acute infectious disease in terms of number of pathogens present and severity of signs and symptoms.
Incubation period: low pathogen #, very few symptoms
prodromal period: mid pathogen #s, average symptoms
Illness period: sick and getting sicker. Pathogen #s rise and symptoms worsen to a peak
Decline period: pathogen #s start to decline and symptoms lessen
convalescence period: symptoms stop quickly before the pathogen #s eventually do
Koch’s postulates
1. Suspected agent must be gone in all healthy organisms, but present in the diseased organisms
2. Isolate causative agent from infected organism and grow it in culture
3. Cultured agent must cause same disease when put into healthy regular patient
4. Same agent must be reisolated from inoculated diseased organism
What are the limitations of kochs postulates
many pathogens aren’t culturable
some only cause disease in certain hosts
Some diseases are asymptomatic
Some diseases involve multiple pathogens
What determines outcome of host parasite relationship (come back to)
if they’re primary or opportunistic (attack when immune system compromised)
How do virulence factors contribute to signs and symptoms of infectious diseases
pathogens may make exoenzymes or toxins that work as virulence factors to let them colonize the host
Production of toxins contributes to signs and symptoms of diseases
Virulence factors used for adhesion to attach to receptors on the host cell. They use adhesin (protein or glycoprotein)
Bacteria use virulence factors to evade phagocytosis
Explain toxins
Poisonous substances produced by microorganisms.
Produce fever, cardiovascular problems, diarrhea and shock
Endotoxins
Exotoxins
Toxigenicity: ability of microorganism to make toxin
toxemia: presence of toxin in hosts blood
intoxication: presence of toxins without microbial host
Endotoxins
Built into the structure of a bacterium and released when the cell is damaged.
Often cause fever, inflammation, sometimes serious septic shock
Exotoxins
Proteins made inside pathogenic bacteria as part of their growth and metabolism. The exotoxins are then secreted into the surrounding medium during the log phase.
Virulence factors of eukaryotic pathogens
Fungal virulence: production of adhesins and proteases (Candida), capsule production (Cryptococcus), exotoxins (Claviceps purpurea – ergotism)
Protozoan virulence: unique features for attaching to host cells like flagella, antigenic variation, formation of capsules.
Helminth virulence: proteases to penetrate skin, large size to evade the immune system, and destruction of antibodies.
Epidemiology
Study of where and when diseases occur, and how they’re transmitted in populations
Notifiable disease (noteworthy)
diseases that physicians are required to report occurrences of
considered to be important for public health on national scale
Mortality
deaths from a notifiable disease
Morbidity
incidence (number of new cases) of a specific notifiable disease.
incidence measures risk of developing disease
Sporadic diseases
uncommon, occurs only occasionally and affect relative number of persons.
Endemic diseases
constantly present in certain percentage of population
Epidemic diseases
Disease acquired by many people in each area, causing damage to living organisms in short period of time
Pandemic diseases
affect large geographical area, often global scale
What’re the types of epidemiological studies?
observational: data gathered from study participants through questions or measurements
Experimental: Uses lab or clinical studies to manipulate the subject and study connections between diseases and potential causative agents or assess treatments
Which factors would increase the spread of a communicable disease? (comeback to?)
High population density
quick global travel
poor hygiene and sanitation
How can diseases be transmitted
Direct contact transmission: requires close association
Indirect contact transmission; spreads to a host by a nonliving object called a fomite
Droplet transmission: transmission via airborne droplets less than 1 meter
Vehicle transmission; transmission by inanimate reservoir. (waterborne, foodborne, or airborne)
2 general methods of disease transmission:
Mechanical transmission: arthropod carries pathogen on its feet
Biological transmission: pathogen reproduces in vector; transmitted through bites or feces
What’re nosocomial infections
A healthcare associated infection that was acquired while receiving treatment in a health care facility; got sick by being in hospital
Mostly upper respiratory tract infection
1/25 in hospital patients are infected
2 mil people per year infected, leads to 20,000 deaths
What’s a compromised host
An individual whose resistance to infection is impaired by disease, therapy, or burns
What factors contribute to re-emerging and emerging diseases
If they’re zoonotic, need a vector, or are viral
Population growth, migration, international air travel
Poverty wars and destructive ecological changes
What’s innate immunity
Ability to fight infection or toxins. If not immune then they’re susceptible to disease
Present at birth, protect against any pathogen, doesn’t discriminate between what’s you and what’s not you. Has no memory.
What is adaptive immunity
specific; responds to specific pathogens, discriminates between self/non-self memory.
T cells and B cells
What’re the main anatomical features of the lymphatic system
lymph: carries microbes to lymph nodes where lymphocytes and macrophages destroy the pathogen
lymphatic vessels
lymphoid tissue
red bone marrow
what’re some of the functions of antimicrobial peptides
a special class of broad-spectrum antimicrobial mediators
nonspecific (can interfere with different pathogens)
Some AMPs are secreted by microbiota, macrophages, or other parts to inhibit pathogens such as bacteria, viruses, or fungi by disrupting their cell membranes
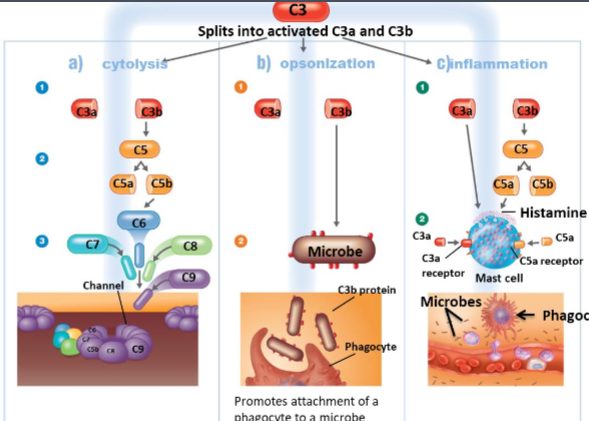
What’s the complement system
Complement is a group of over 30 proteins circulating in serum that are activated in a cascade: one complement protein triggers the next
Complement activation is the process by which circulating complement precursors become functional
Process triggered by classical, alternative, or lectin pathways
What’re the outcomes of complement activation
Cytolysis: Proteins create a membrane attack complex (MAC). Microbes burst as extracellular fluid flows in through transmembrane channel formed by membrane attack complex.
opsonization: Coating microbes with C3b promotes attachment of a phagocyte to a microbe
inflammation: Blood vessels become more permeable, and chemotactic agents attract phagocytes to area.
What’re interferons
cytokines (proteins) made by cells that have antiviral activity
made in response to viral infections, cause neighboring cells to make antiviral proteins to inhibit viral replication
some cause neutrophils and macrophages to kill bacteria
induce apoptosis in viral cells
activate and stimulate immune cells in response to viruses
What’re the signs of acute inflammation and fever?
symptoms: redness, swelling, pain, and heat
destroys harmful agents or limits their effect on the body
replaces and repairs tissue damaged by harmful agent
Inflammation activates acute-phase proteins by the liver that cause vasodilation and increased permeability of blood vessels
What’re the cellular and molecular mediators of inflammation
histamine: stimulates vessels to open further.
Blood plasma and platelets are released into area.
Kinins: Bradykinin – causes heat, promotes extravasation (diapedesis), stimulates mast cells to degranulate, and release histamine
Prostaglandin: promotes histamine release, contribute to fever
Leukotrienes: pro-inflammatory mediators produced by leukocytes
Cytokines: small proteins important in cell signaling
What’re the signs of chronic inflammation
Some pathogens resist host defenses
Remain in body stimulating inflammatory response
Body “walls off” site of inflammation forming a granuloma
Fibrotic lesion around bacteria
Identify and describe components of blood
Cells and cell fragments suspended in plasma
Erythrocytes (red blood cells): transport oxygen from lungs to body tissue, and bring CO2 back to lungs
Leukocytes (white blood cells): immune system response cells that travel through bloodstream to attack foreign pathogens
Platelets: stop bleeding by adhering to blood vessel walls
Created in red bone marrow stem cells via hematopoiesis
What’re the possible outcomes of the inflammation process (come back to?)
Explain the process of phagocytosis and the mechanisms by which phagocytes destroy and degrade pathogens.
Phagocytosis: cells capacity to seek, ingest, and kill pathogens
phagocytes squeeze between endothelial cells of blood vessels through diapedesis
trans endothelial migration: sticking of phagocytes to blood vessels in response to cytokines at site of inflammation