GEN CHEM Ch. 4 Periodic Properties
1/32
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
33 Terms
group
elements in the same column with same number of valence electrons
period
elements in the same row with same number of electron shells
atomic number
number of protons in the atom
mass number
protons and neutrons in a nucleus
group 1
alkaline metals
very reactive with H2O
group 2
alkaline earth metals
reactive with H2O
group 3-12
transition metals
colorful except row 4
multiple oxidation states
most common (+2)
high melting and boiling point
valence electrons in outermost d-orbital

group 13-17
metalloids
combination of metal and non-metal properties

group 17
halogens
group 18
noble gases

period 6-7
inner transition metals
lanthanides (6) and actinides (7)
multiple oxidation states
most common (+3)
valence electrons in f-orbital
oxidation states
number of electrons that an atom loses, gains, or appears to use when bonding with another atom
diatomic atoms
atoms that are usually found paired due to their unstable nature
Have No Fear Of Ice Cold Beer
Hydrogen, Nitrogen, Fluorine, Oxygen, Iodine, Chlorine, Bromine
metals
malleable, lustrous
good conductors of electricity/heat
form basic oxides
lose electrons to form cations
usually solid at room temp, with exception of mercury (Hg) which is liquid
generally high MP and BP
non-metals
brittle, dull
poor conductors of electricty/heat
form acidic oxides
gain electrons to form anions
gas or solid at room temp, with the exception of bromine (Br) which is liquid
general low MP and BP
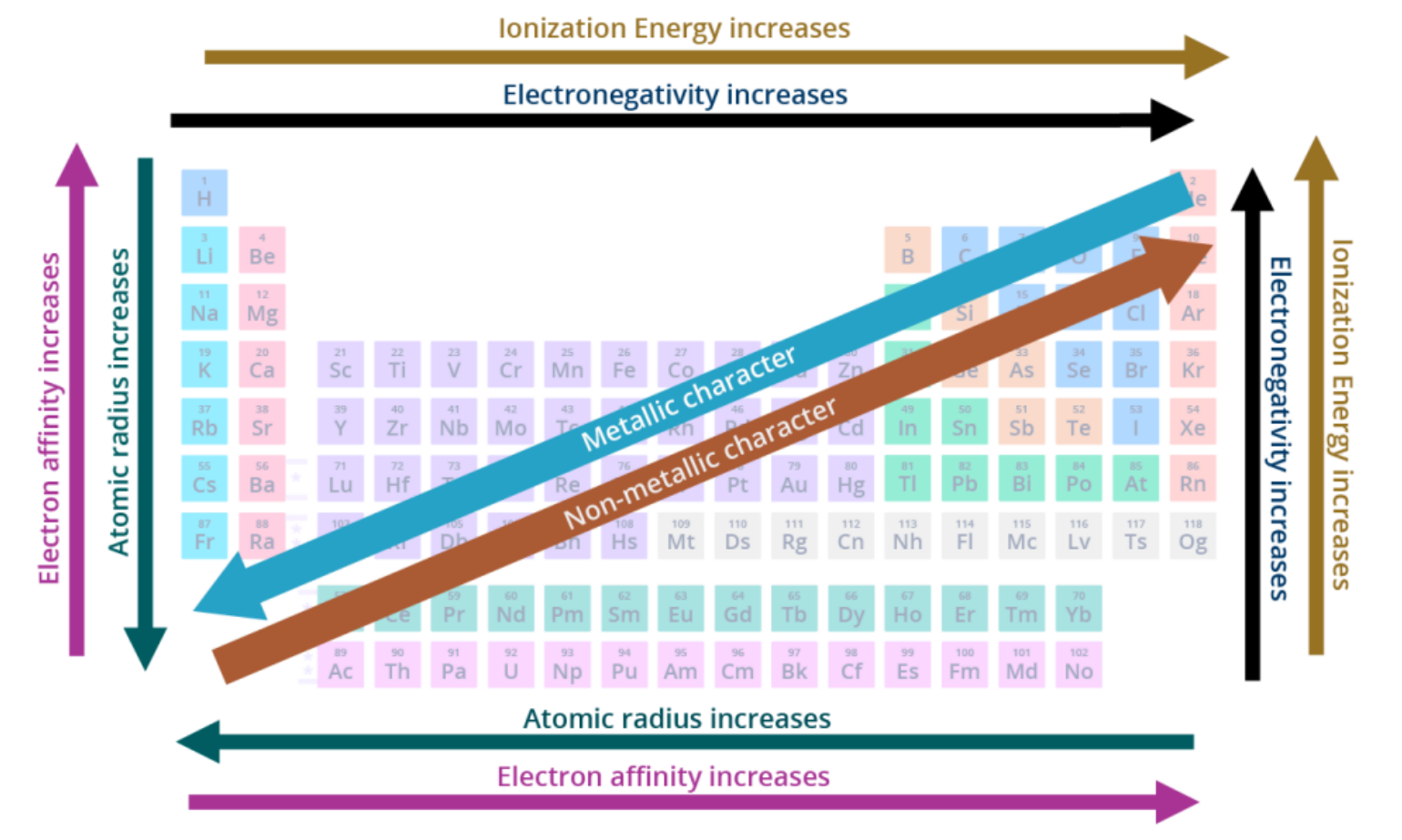
atomic radius
half the distance between the nuclei of two identical atoms between the nuclei of two identical atoms bonded = d/2
increases from R to L across a period and increases going down a group
anions have larger radii compared to cations (anions have more e- in the outer shell)
isoelectronic series → have the same number of e- but a different number of protons
ex: Ca+2 < K+1 < Ar < Cl- < S-2
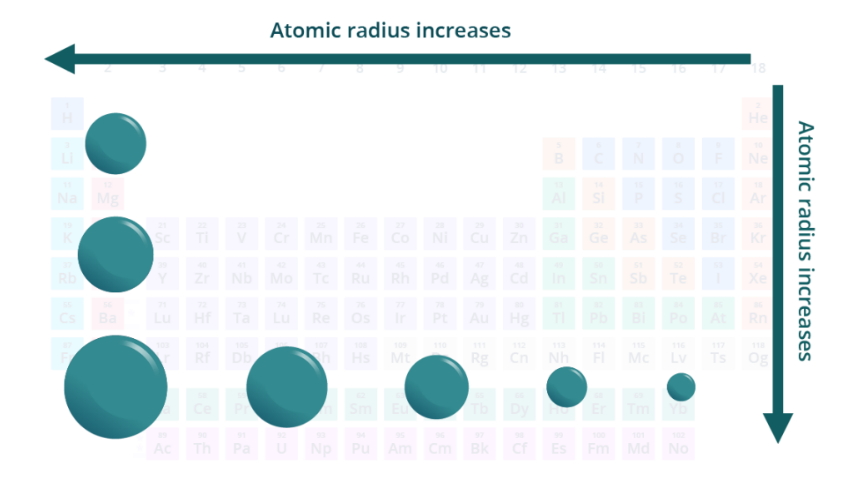
atomic radius increases from ___ to ___ across a period and increases going ____ a group
R to L, down
anions have larger ____ compared to cations (anions have more e- in the outer shell)
radii
isoelectronic series
have the same number of e- but a different number of protons
anions = ions that have gained e- and have more e- than protons → negatively charged
cations = ions that have lost e- and have more protons than e- → positively charged
anions
ions that have gained e- and have more e- than protons → negatively charged
larger radius
typically for
cations
ions that have lost e- and have more protons than e- → positively charged
smaller radius
typically formed from metals
effective nuclear charge (Zeff)
amount of positive charge experienced by an electron in the outermost orbital (how tightly an e- is held by nucleus)
increases from L to R across a period and increases going down a group
Zeff = Z - S
Z = # protons
S = # non-valence (shielding) e-
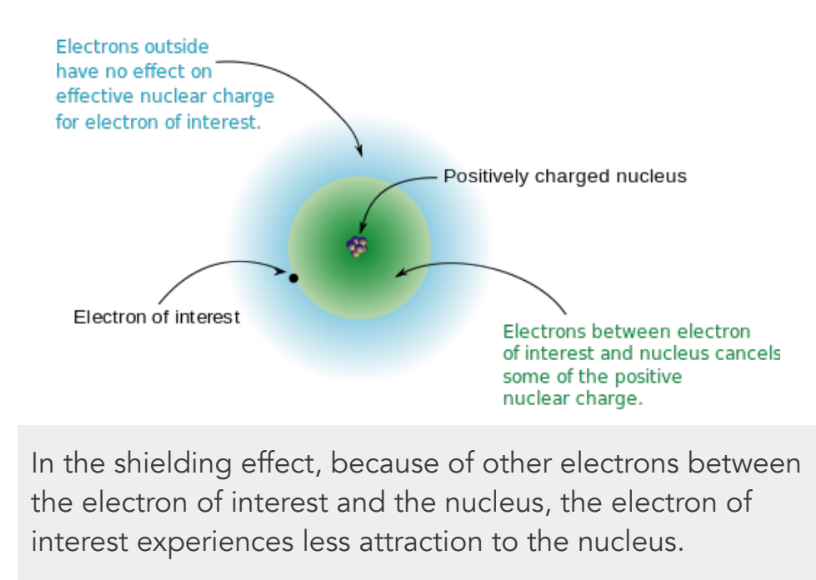
effective nuclear charge increases from __ to __ across a period and increases going ___ a group
L to R, down
Zeff = Z - S
number of protons - number of shielding (non-valence electrons)
shielding electrons
non-valence electrons = atomic number of preceding noble gas
ex: Al - Ne is the preceding noble gas and has atomic number 10 → Al has 10 shielding electrons
ionization energy (IE)
energy needed to remove an electron from an atom
increases from L to R across a period and increases going up a group
includes Noble gases
exceptions
half-filled orbital and filled obitals have greater IE
group 2 > group 13 (ex: Mg > B)
alkaline earth metals have filled orbitals
group 15 > group 16 (ex: N > O)
group 15 have half-filled orbitals
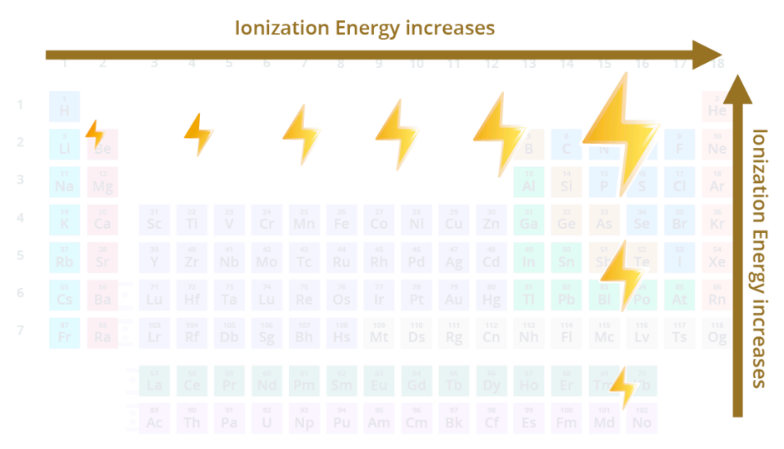
ionization energy increases from __ to __ across a period and increases going ____ a group - includes ______ gases
L to R, up, Noble
multiple ionization energy
1st ionization energy: energy required to move the outermost e-
2nd, 3rd, 4th: energy required to remove 2nd, 3rd, 4th e-, etc.
if the subsequent e- is not in the valence shell, it’s more difficult to remove → the value is always LARGER
ex: Lithium’s second IE is higher than the first IE
electron affinity (EA)
amount of energy released when e- is added to an atom
increases from L to R across a period and increases going up a group
exceptions
half-filled / filled orgitals have lower EA
group 2 < group 1 (ex: Ca < K)
group 2 has filled s-orbitals
group 15 < group 14 (ex: P < Si)
group 15 has half-filled p-orbitals
Noble gases have very low EA
fillled electron shells
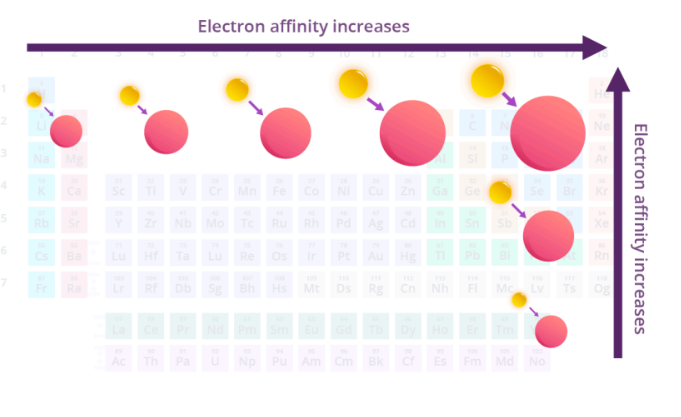
electron affinity increases from __ to __ across a period and increases going _____ a group
L to R, up
electronegativity (EN)
abiltiy to attract e-
increases from L to R across a period and increases going up a group
Most electronegative is Fluorine
exception
noble gases have very low EN bc full valence shells
electronegativity increases from __ to __ across a period and increases going ___ a group
L to R, up
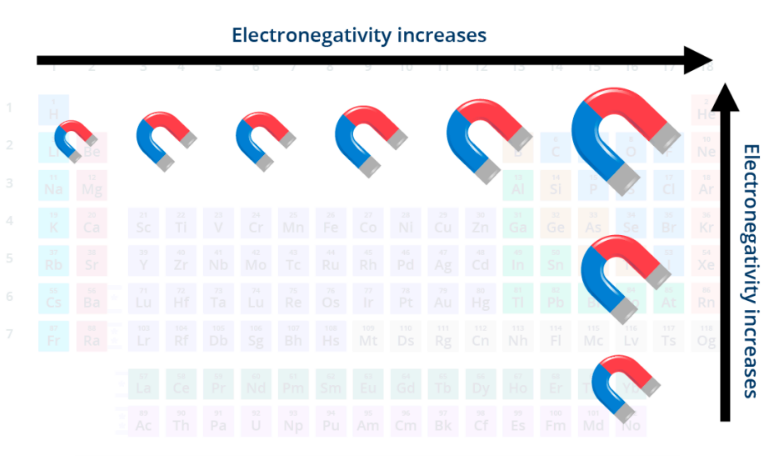
summary of periodic trends