🧬 Bonding
1/93
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
94 Terms
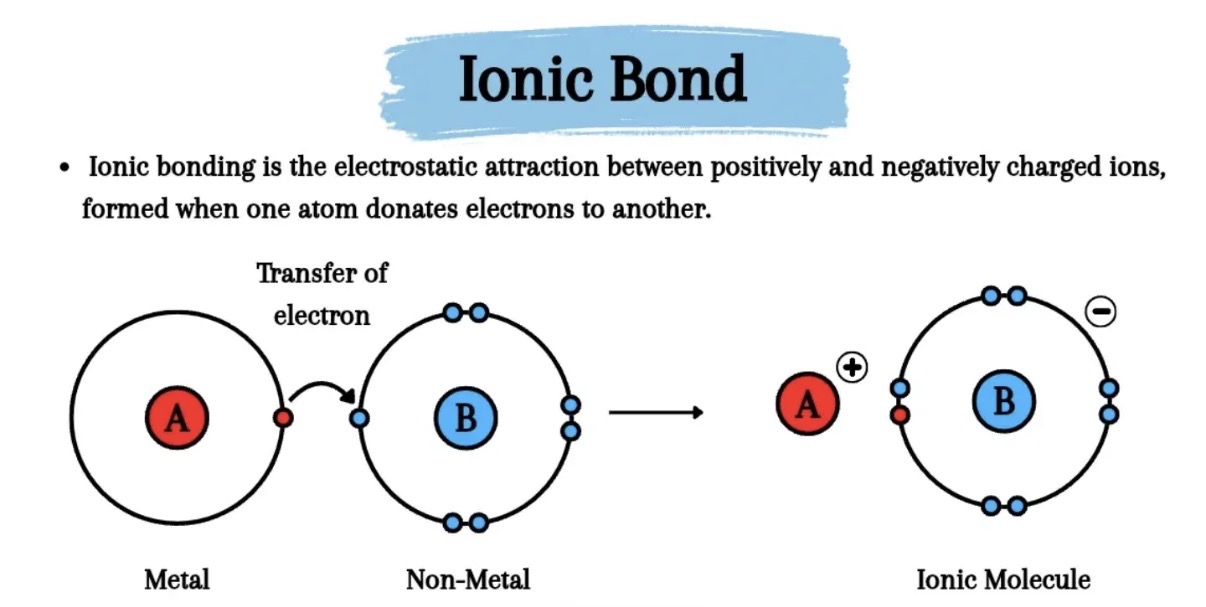
What is ionic bonding?
Bonding between metal and nonmetal where electrons are transferred from one atom to another
What ions do group 1 form?
+1
What ions do group 2 form?
+2
What ions do group 6 form?
-2
What ions do group 7 form?
-1
What does ionic bonding form?
Ions with full electron outer shells
What is the ionic formula of carbonate?
CO32−
What is the ionic formula of ammonia?
NH4+
What is the ionic formula of hydroxide?
OH−
What is the ionic formula of nitrate?
NO3−
What is the ionic formula of sulfate?
SO42−
What are ionic ions held by?
Electrostatic attraction which hold + and - ions together, it is very strong and forms ionic compounds
When done in a lattice it is called ionic bonding
In ionic bond do charges cancel out
Yes they sum to 0
What are ionic crystals?
They are a giant ionic lattices of ions. ‘Giant’ refers to a normal ionic lattice being repeated
What is an example of a giant ionic lattice?
Sodium chloride

What is the electrical conductivity of an ionic compound and why?
They conduct electricity when molten or dissolved in a solution.
The ions in a liquid are free to move (and they carry a charge).
Why can’t ionic compound conduct electricity as a solid?
In a solid the ions are held in fixed positions by strong electrostatic forces (ionic bond)
What is the melting point of an ionic compound and why?
They have high melting points
The giant ionic lattice are held together by strong electrostatic forces of attraction
It takes a lot of energy to overcome these forces
What is the solubility of an ionic compound and why?
They tend to be soluble in water as
Water is polar (part of molecule is partially positive and other is partially negative)
The water molecules pull the ions away from lattice and cause it to dissolve
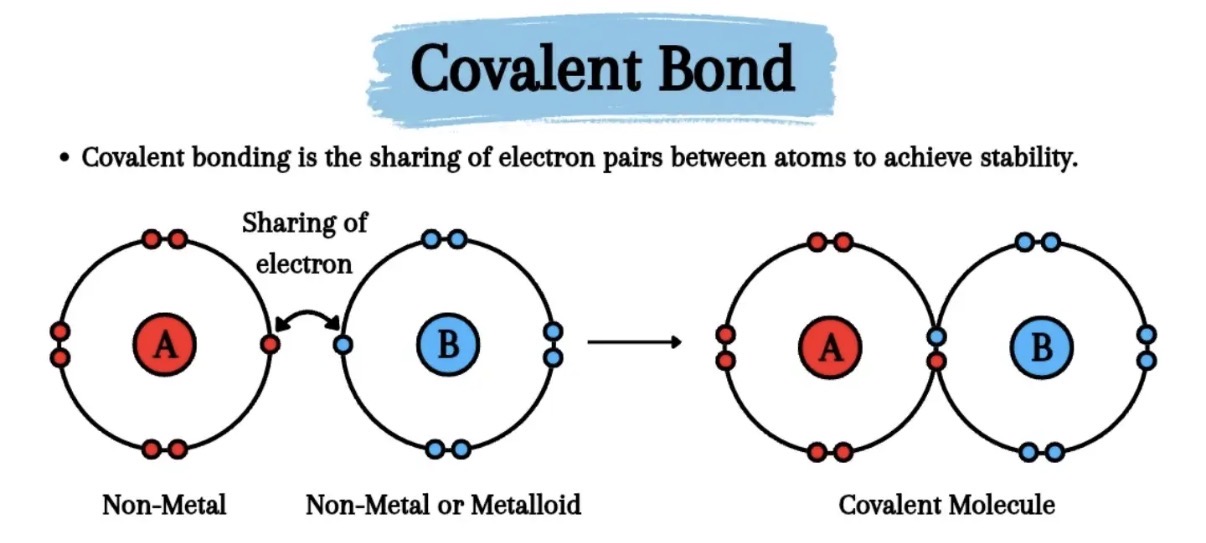
What is an covalent bond?
A covalent bond (which can be single, double or triple) between a metal and nonmetal with intermolecular forces also present. Electrons are shared
What are single covalent bonds?
Where 2 atoms share two electrons, so they both get fuller outer shells.
Both the positive nuclei are attracted electrostatically to the shared electrons
What are double covalent bonds?
When atoms share 4 electrons to get a full outer shell
What are triple covalent bonds?
When atoms share 6 electrons to get a full outer shell
What are simple covalent compounds?
The atoms in the molecules are held together by strong covalent bonds but the molecules within covalent compound are held by intermolecular forces
What are the atoms in molecules held by?
Strong covalent bonds
What are the molecules in covalent compounds held by?
Intermolecular forces
What are giant covalent structures?
They are a types of crystal structure- a huge network of covalently bonded electrons
Why do carbon atoms form a giant structure?
Because they can each form 4 strong covalent bonds
How are the carbon sheets in graphite bonded?
They are arranged in flat hexagons with three carbons covalently bonded to each other. The 4th outer electron of each carbon is delocalised
How many delocalised electrons does graphite have per carbons bonding have?
1
What are sheets of graphene held by in graphite.
Weak van der Waals forces
What are Van der waals forces?
These are instantaneous dipoles:
They occur because the electron charge cloud is constantly moving around atom/molecule
When it is more on one side of atom/molecule it forms a dipole and δ+ charge and δ− charge on either side of atom
If one end lines up with end of other atom/molecule of opposite charge it forms an instantaneous temporary dipole on other atom
This creates a domino effect
What makes some Van der waals forces stronger than others?
Larger forces: have larger charge clouds therefore stronger forces
Shape of molecules: If molecule is longer and straighter it means molecules can get closer so intermolecular forces can be stronger
How do intermolecular forces affect boiling points?
As strength of intermolecular force increases the energy needed to overcome increases so boiling point increases
What are dipole- dipole forces ?
They are weak intermolecular forces caused by the attraction of opposite partial charges
They are permanetent and caused by the uneven electron distribution
Are non polar molecules affected by a charge?
No as no change in distribution of charge/electron throughout molecule
If a polar Liquid is placed next to a charge rod wha happens?
The liquid will bend so the oppositely charged end of molecule will face the rod
The more polar the molecule the more it will bend
What are hydrogen bonds?
A weak intermolecular forces between hydrogen and NOF particle. Where hydrogen is δ+ and NOF particle is δ−
NOF particles are very electronegative so draw bonding electrons from H2atoms (which have high charge density-very small)
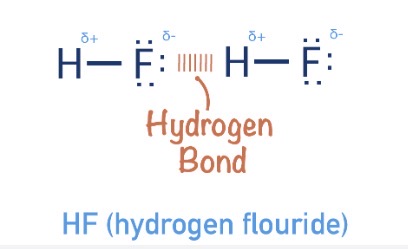
Strength order of weak intermolecular forces?
WEAKER than covalent and ionic bonds
Hydrogen
Dipole-Dipole
Van der Waals
Why is graphite slippery and a lubricant?
The weak van der Waals forces in between the layers are easily broken so the layers can slide over each other
Why can graphite conduct electricity?
The delocalised electrons in graphite (carbon only bond to 3 other C) are free to move along sheet so conduct electrical charge
Why is graphite low density and what is used for due to this?
It is low density due to distance between layer of covalent bonds and other layers, so low density and used to make light weight sports equipment
Why does graphite have such a high melting point?
Due to strong covalent bonds in hexagon sheets. Very difficult to break
Why is graphite insoluble?
Due to strong covalent bonds in hexagon sheet which are very difficult to break
How is diamond bonded?
Each carbon in diamond is covalently bonded to 4 other carbons in a tetrahedral structure

Why does diamond have high melting point and is extremely hard?
Due to its strong covalent bond which are very hard to break
Why a good thermal conductor?
Because due to its strong covalent bonds vibrations travel easily through its stiff lattice so good thermal conductor
Why can’t diamond conduct electricity?
No delocalised electrons, all localised electrons are held in bonds
Why won’t diamond dissolve in a solvent?
Due to its strong covalent bond which are very hard to break
What is a coordinate bond/ Dative covalent bond?
When one atom provides both shared electrons represented by →

Why does coordinate bond form?
When one atom has lone pair of electron and other has no electrons to share
What does a shape of molecule depend on?
It depends on the number of bonding pairs or lone pairs
What are unshared electrons/non-bonding pairs called?
Lone pairs
What are charge clouds?
Electrons exist are charge clouds
Charge clouds: an area where you have a big chance of finding an electron.
Do electrons stay still?
No they whizz around inside the charge cloud
Do charge clouds repel each other?
Yes as much as they can
How much do lone pairs and bonding pairs repel?
Lone pair charge clouds repel more than Bonding pair charge clouds so bond angles bond angles are often reduced because bonding pairs are pushed together by lone pair repulsion.
Why are bonding pair angles?
Because bonding pairs are pushed together by lone-pair repulsion
How to draw molecules?
Look image

How to find number of electron pairs?
Find the central atom - it's the one all the other atoms are bonded to.
Work out how many electrons are in the outer shell of the central atom.
This will be the same as its group number in the periodic table.Add 1 electron for every atom that the central atom is bonded to.
(You can work this out from the formula of the molecule or ion.)If you're looking at an ion, you need to take its charge into account — add
1 electron for each negative charge or subtract 1 for each positive charge.Add up all the electrons. Divide by 2 to find the number of electron pairs.
Compare the number of electron pairs to the number of bonds to find the number of lone pairs and the number of bonding pairs on the central atom.
How to find electron pairs of CF4
Carbon tetrafluoride- CF,
The central atom in this molecule is carbon.
Carbon's in group 4. It has 4 electrons in its outer shell.
The carbon atom is bonded to 4 fluorine atoms.
CF, isn't an ion.
There are 4 + 4 = 8 electrons in the outer shell of the carbon atom, which is 8÷2=4 electron pairs.
4 pairs of electrons are involved in bonding the fluorine atoms to the carbon so there must be 4 bonding pairs of electrons.
That's all the electrons, so there are no lone pairs.
How to draw molecules with double or triple bonds?
When drawing you can treat these molecules double or triple bonds like 1 bond
What it the name, drawn structure and angle of a compound with 2Bp:0Lp?
Linear - 180
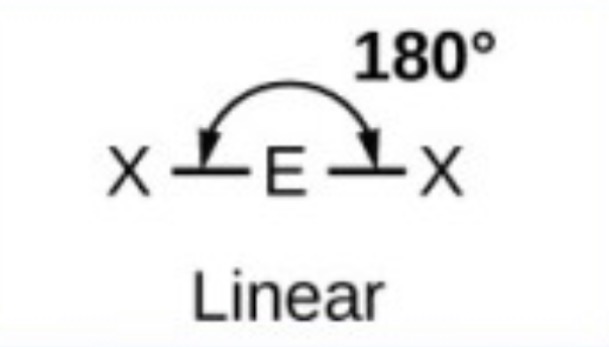
What it the name, drawn structure and angle of a compound with 3Bp:0Lp?
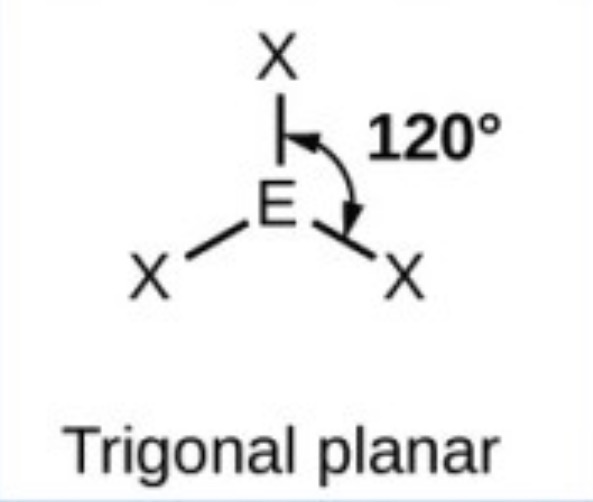
Trigonal planar - 120
What it the name, drawn structure and angle of a compound with 2Bp:1Lp?
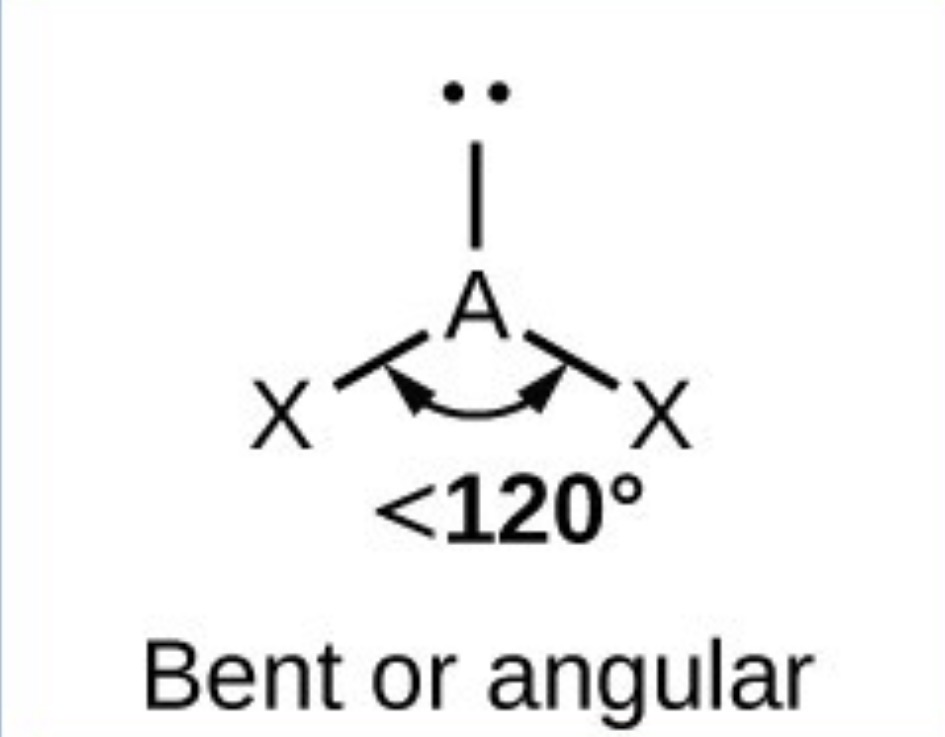
Bent - 117.5
What it the name, drawn structure and angle of a compound with 4Bp:0Lp?
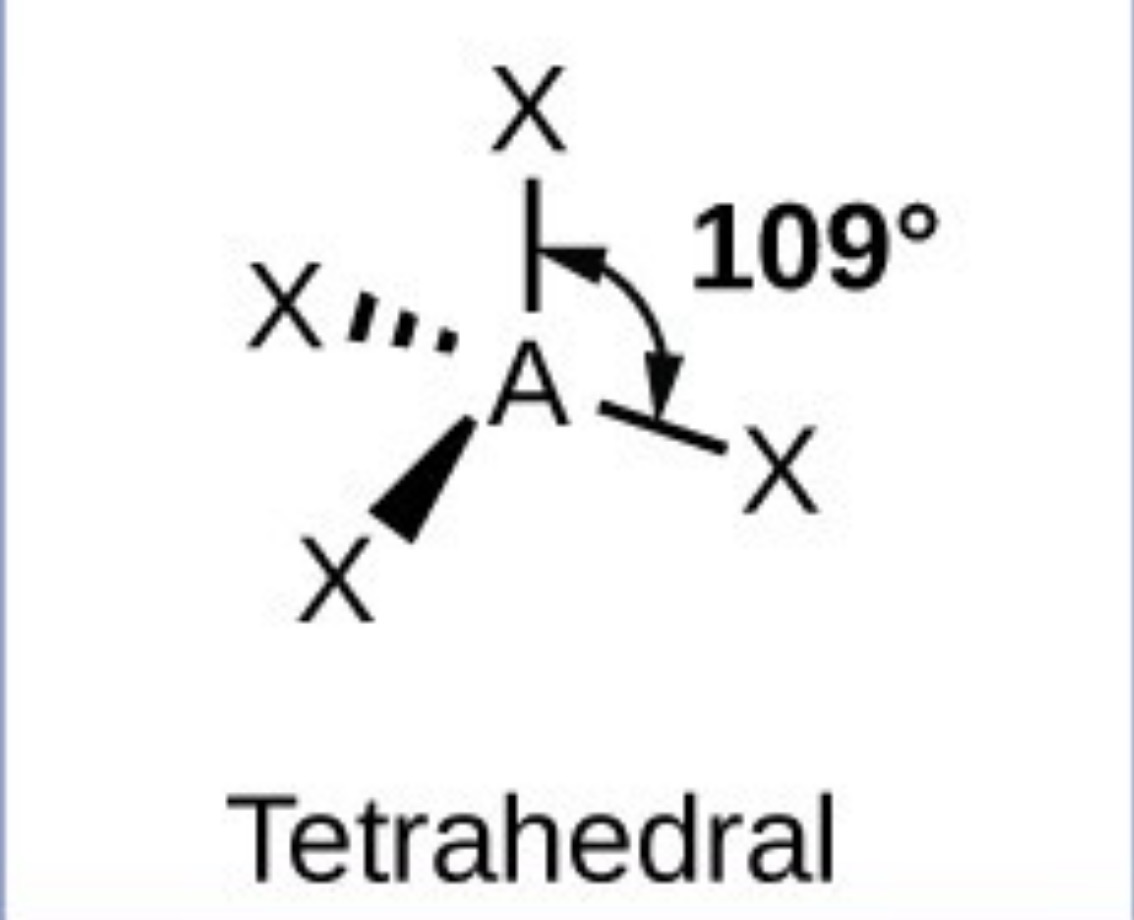
Tetrahedral - 109
What it the name, drawn structure and angle of a compound with 3Bp:1Lp?
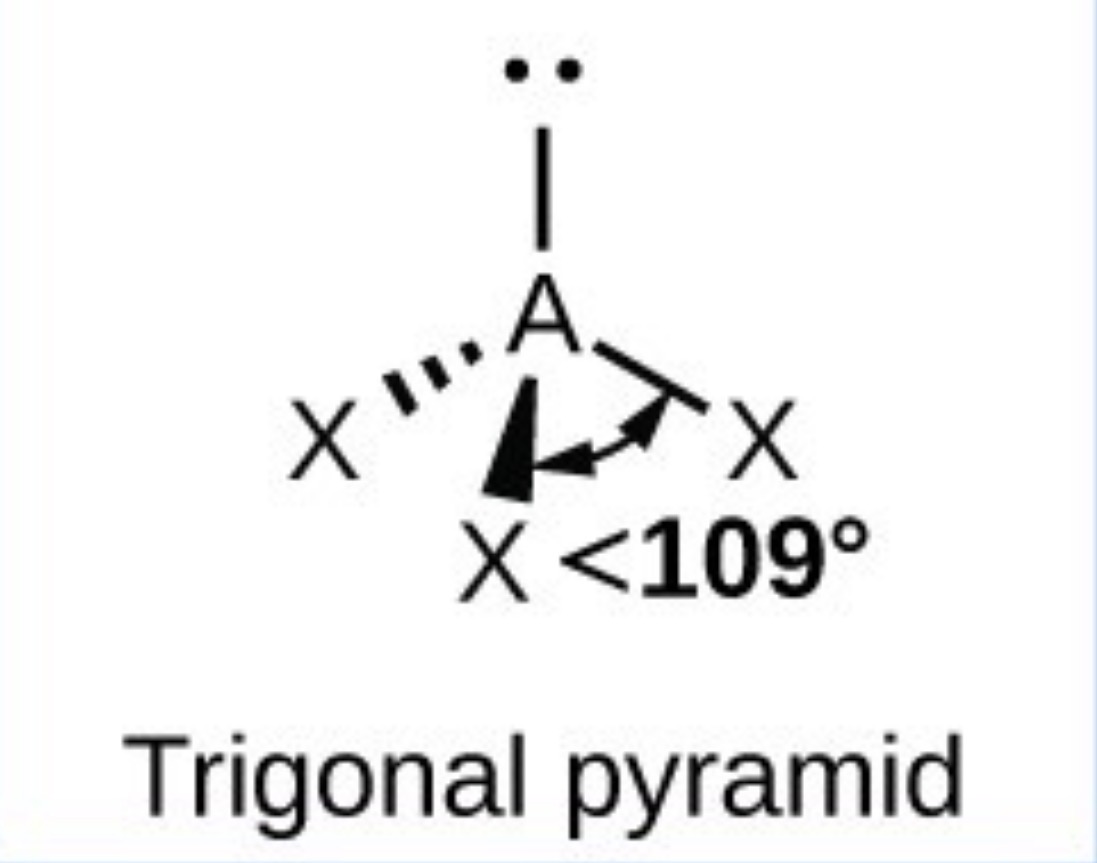
Trigonal pyramid - 107.5
What it the name, drawn structure and angle of a compound with 2Bp:2Lp?
Bent - 104
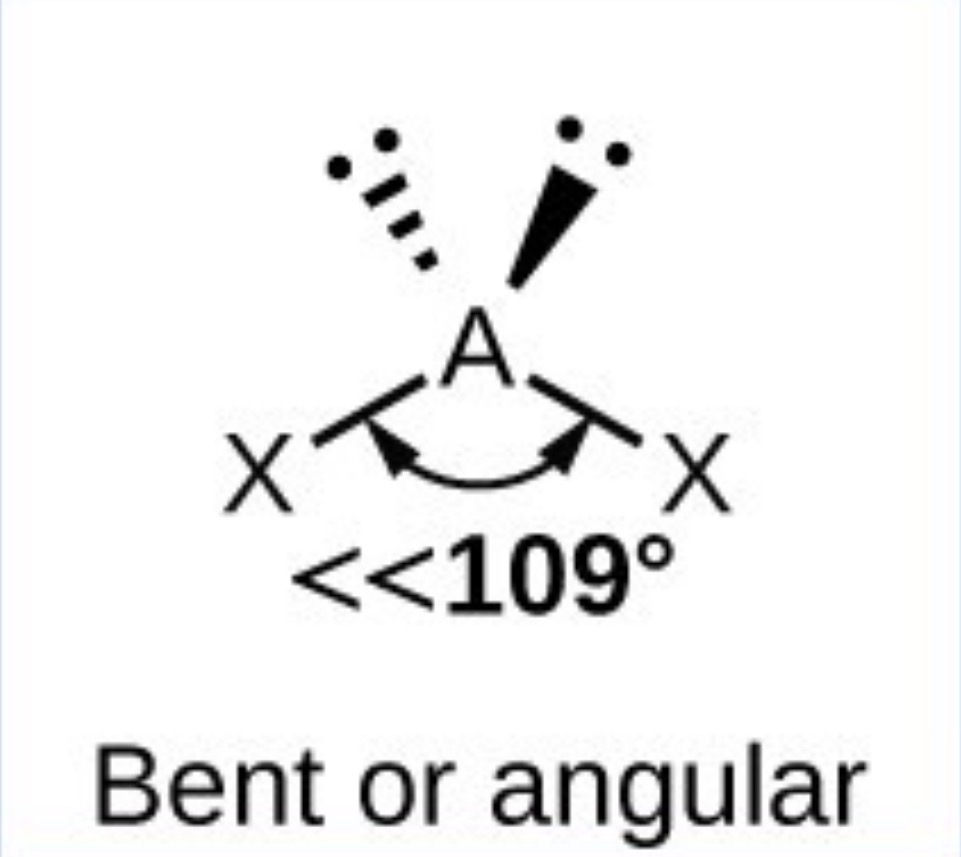
What it the name, drawn structure and angle of a compound with 5Bp:0Lp?
Trigonal bipyramidal - 120(Trigonal planer) and 90 (to other 2)
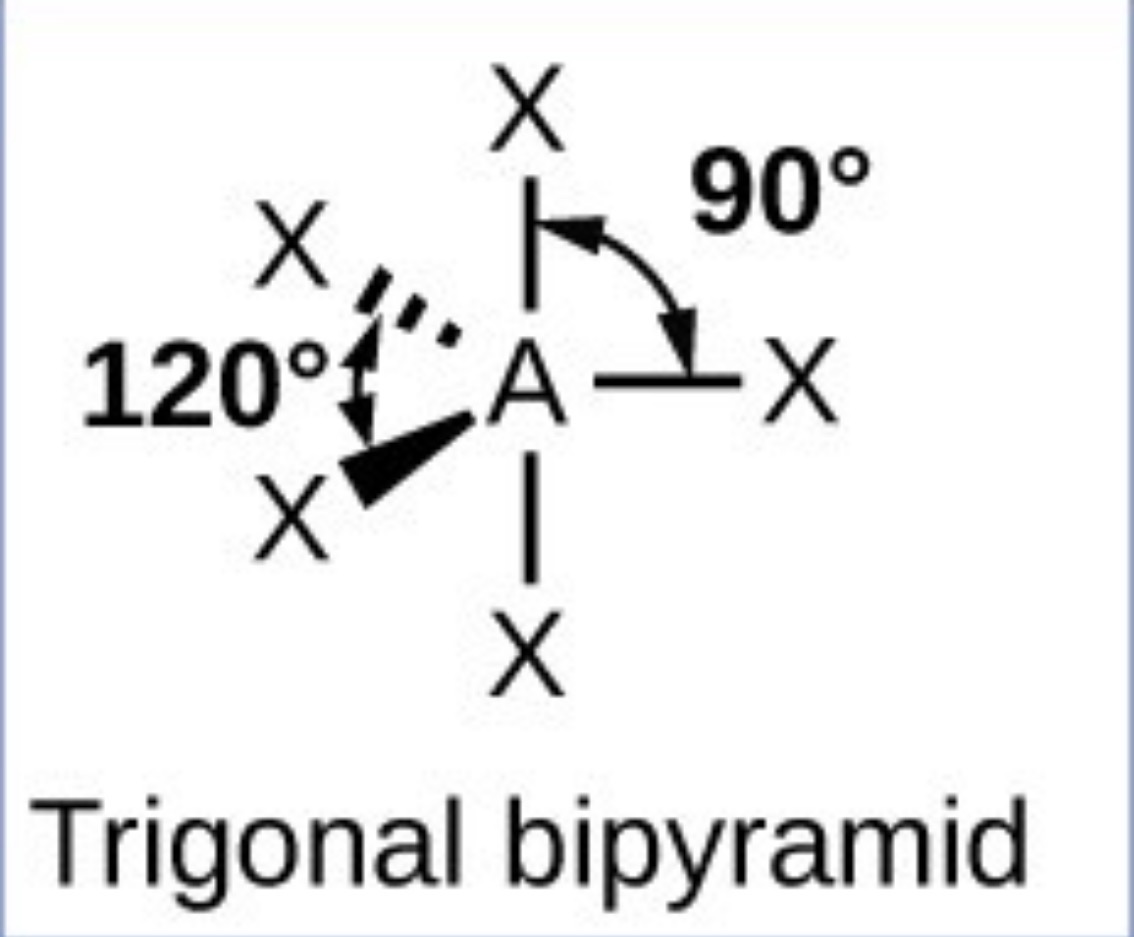
What it the name, drawn structure and angle of a compound with 4Bp:1Lp?
Seesaw - 102 (base of seesaw) and 86.5
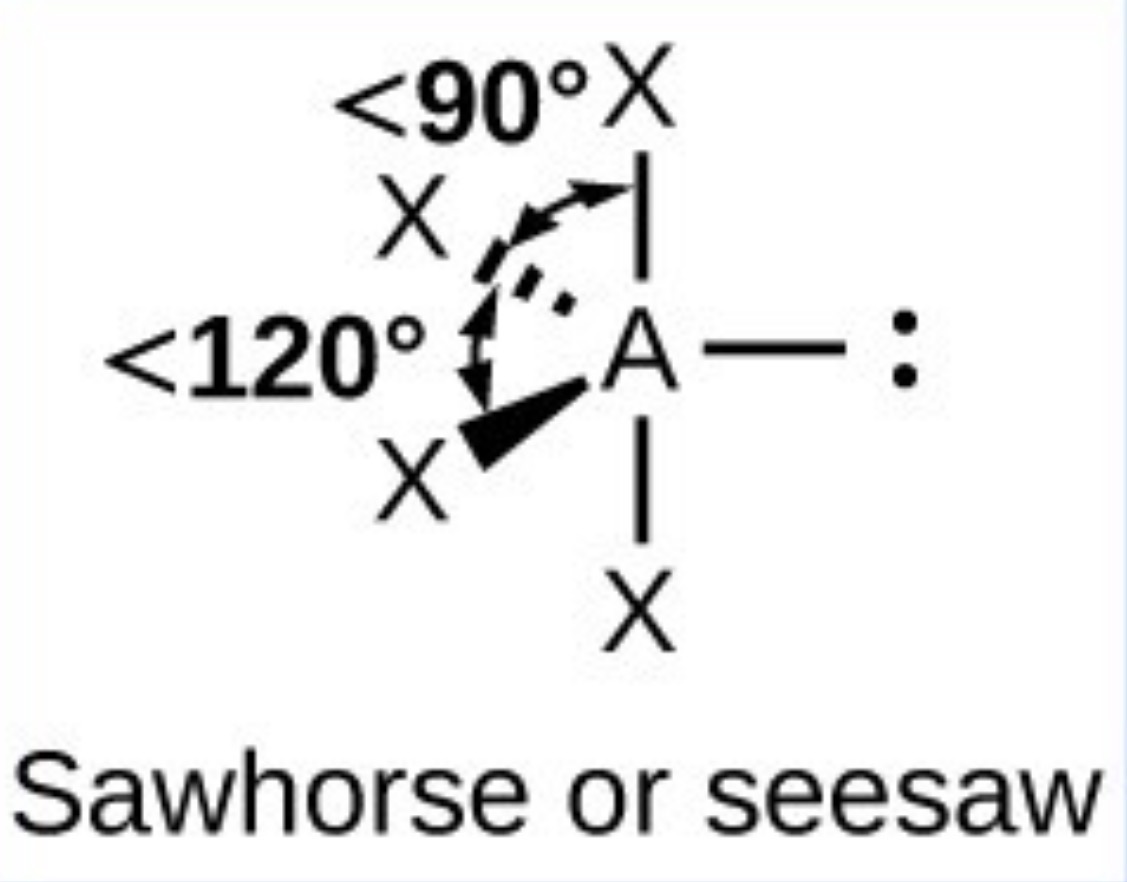
What it the name, drawn structure and angle of a compound with 3Bp:2Lp?
T shaped- 87.5
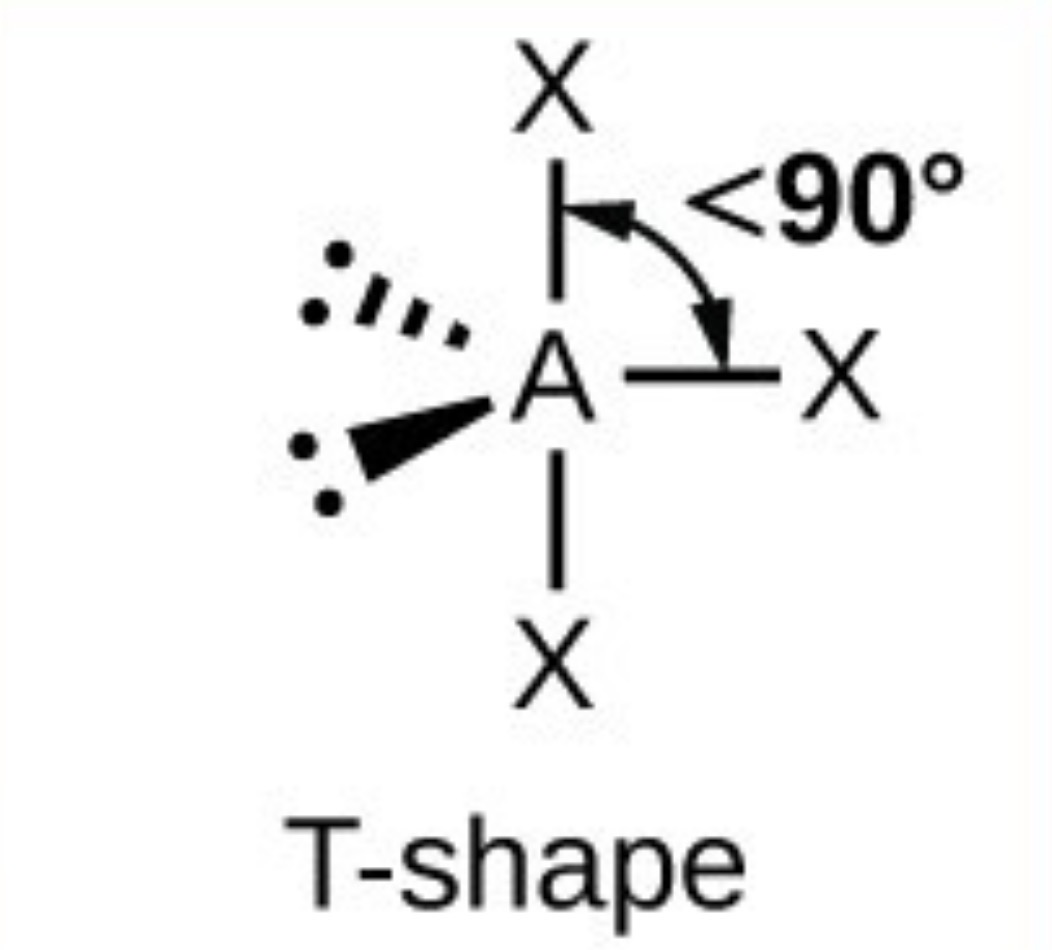
What it the name, drawn structure and angle of a compound with 2Bp:3Lp?
Linear - 180
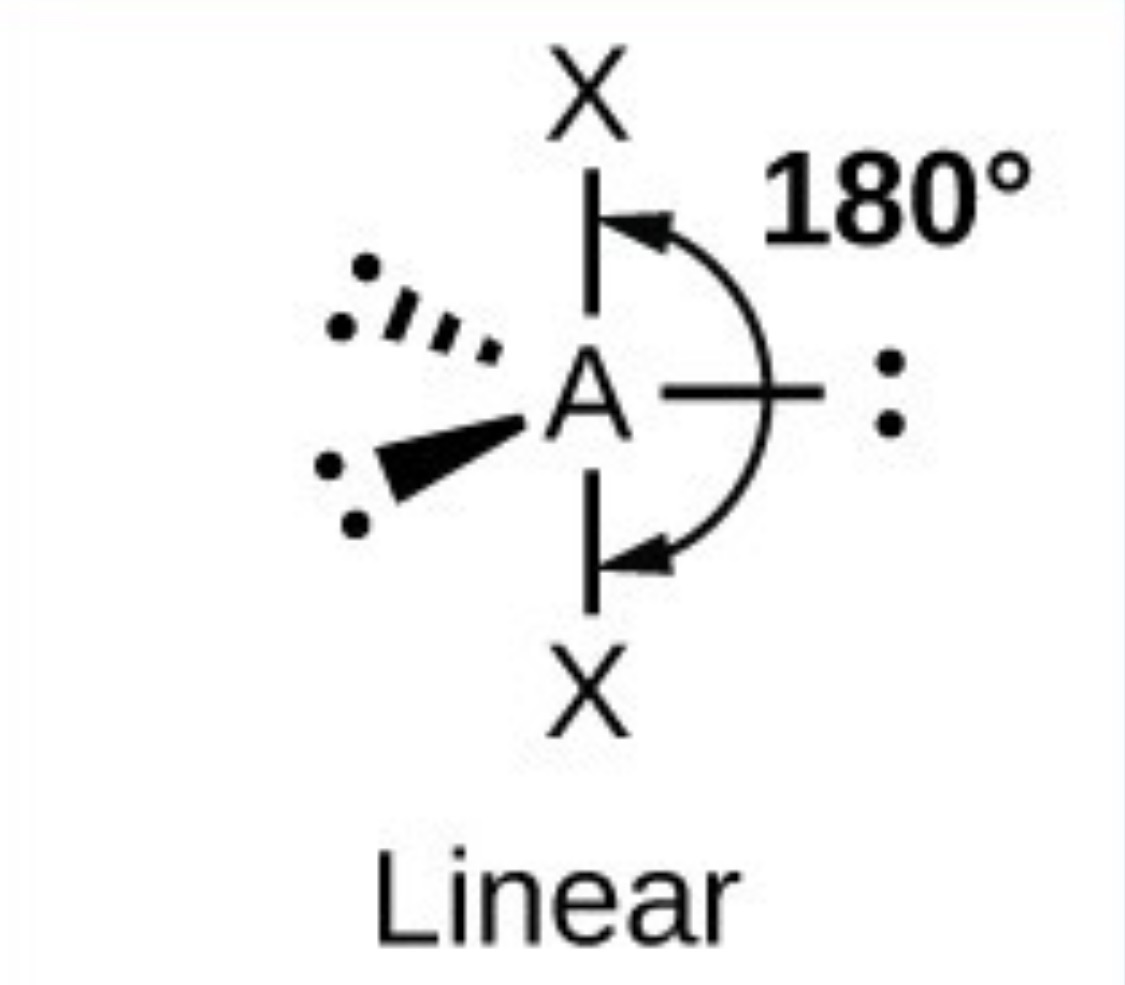
What it the name, drawn structure and angle of a compound with 6Bp:0Lp?
Octahedral - 90
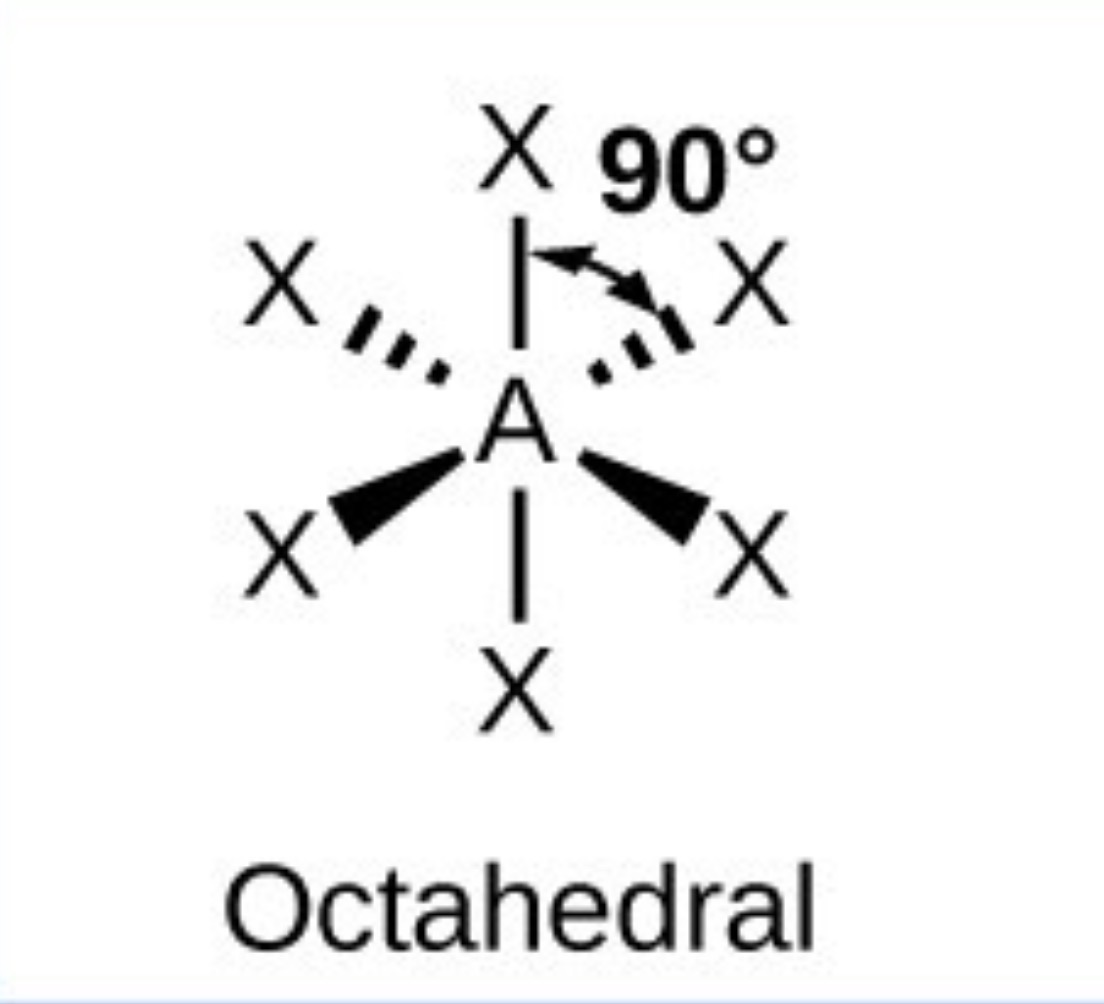
What it the name, drawn structure and angle of a compound with 5Bp:1Lp?
Square pyramidal - 90
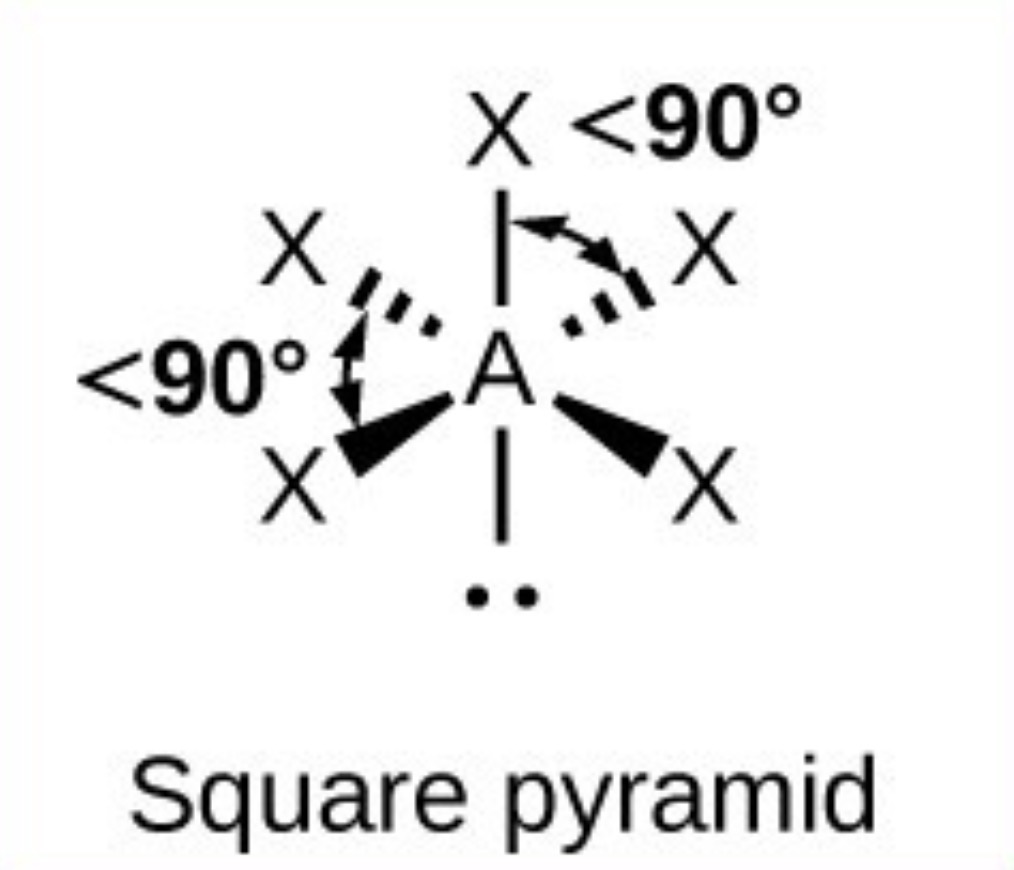
What it the name, drawn structure and angle of a compound with 4Bp:2Lp?
Square planar - 90
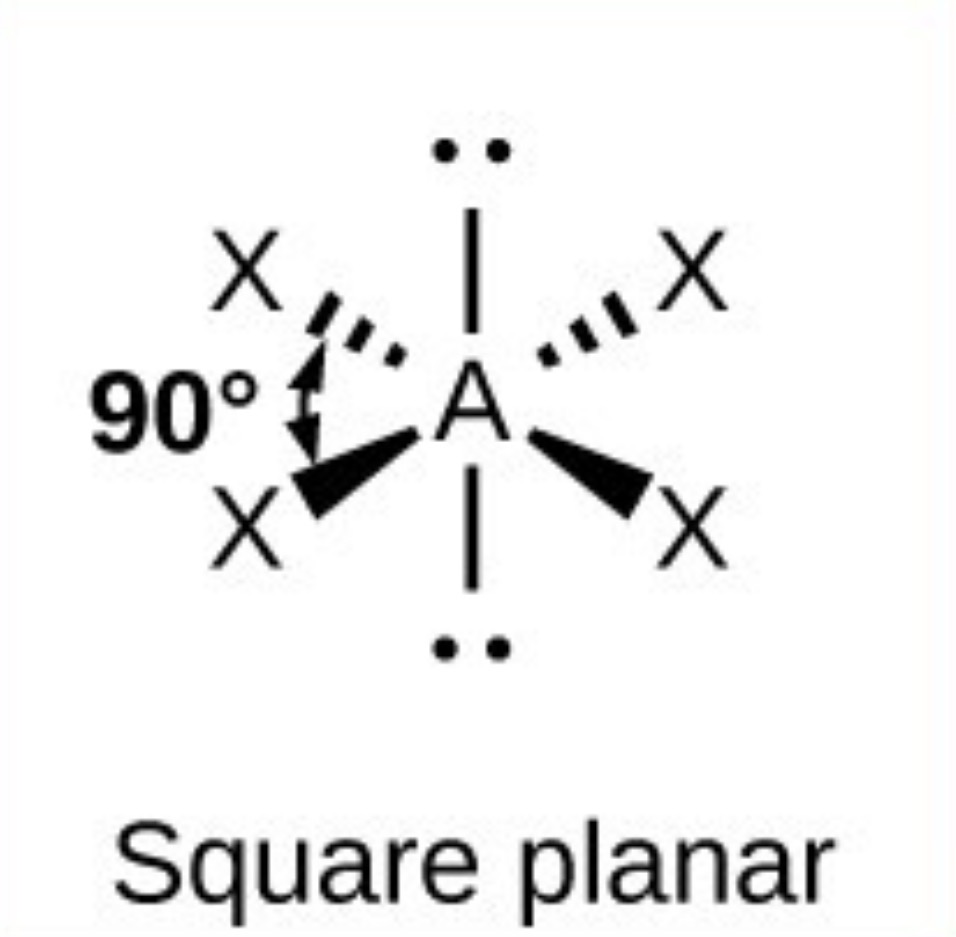
What it the name, drawn structure and angle of a compound with 3Bp:3Lp?
T shape - <90
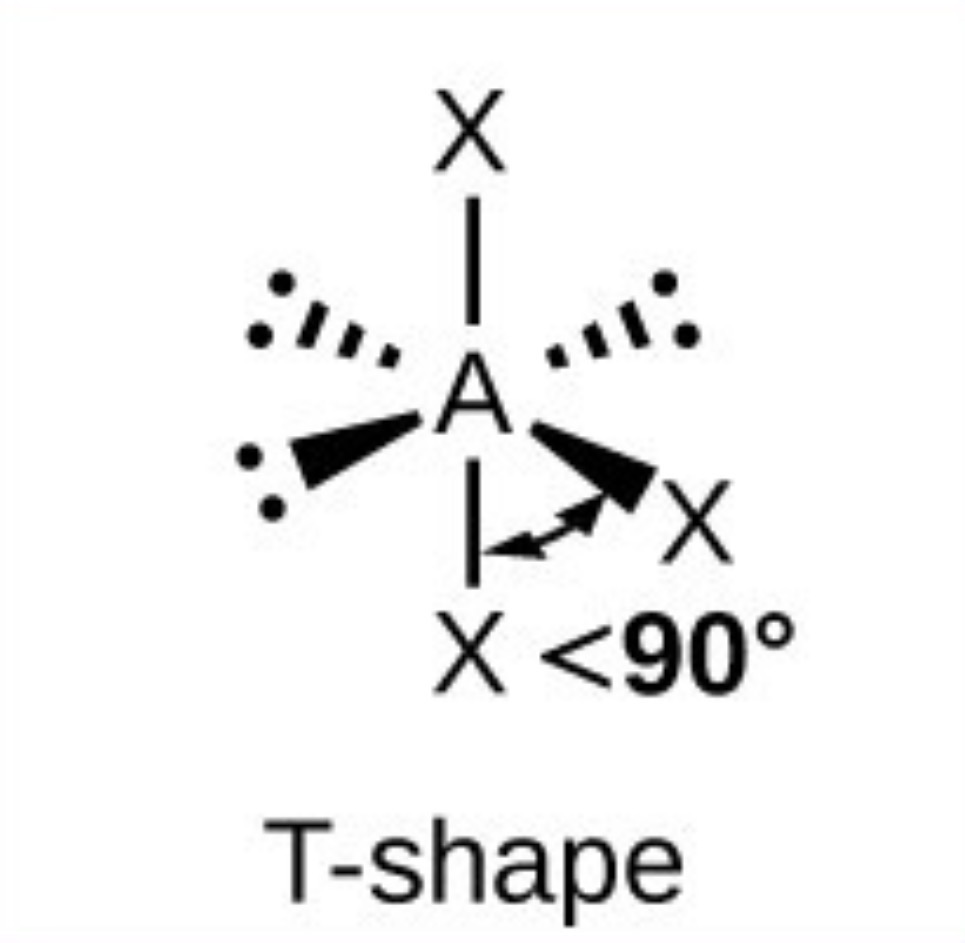
What it the name, drawn structure and angle of a compound with 2Bp:4Lp?
Linear - 180
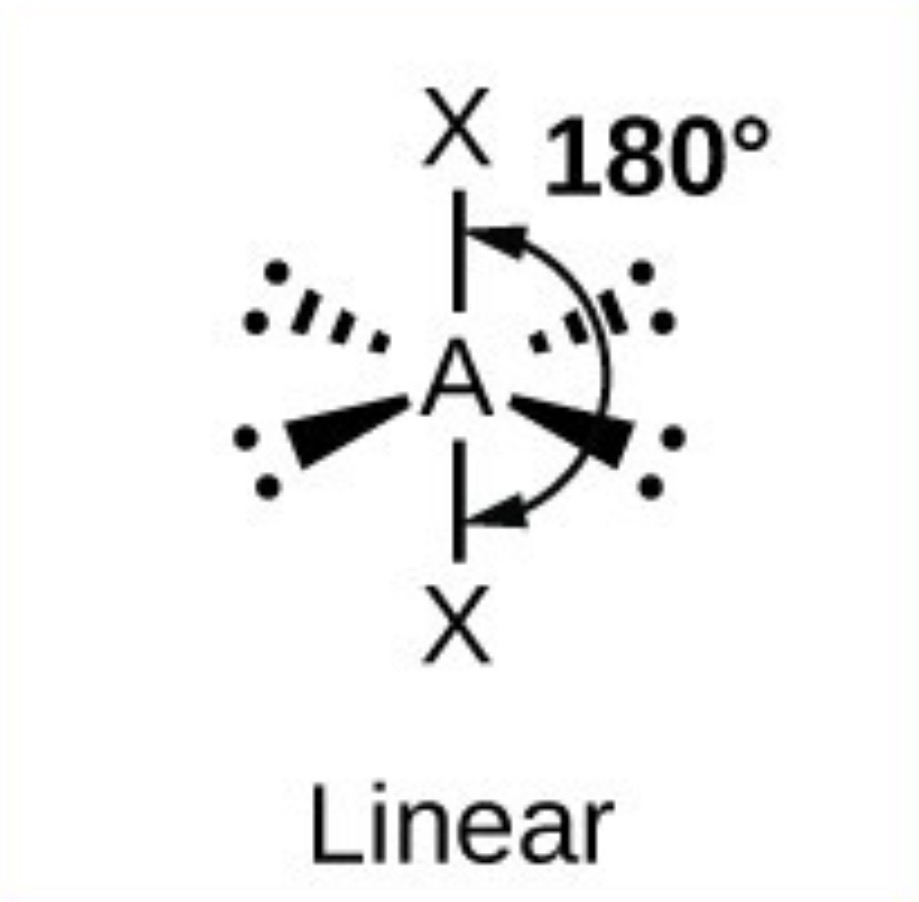
What is electronegativity?
The ability to attract bonding electrons in covalent bond
What is electronegativity measured in?
The Pauling scale
What elements ate most electronegative?
Fluorine (top right of periodic table)
What are non-polar molecules?
Molecules in which atoms in molecules are equally electronegative E,g. Diatomic particles
What are polar bonds?
When electrons are more attractive to one side due to atoms electronegativity. The more electronegative the more polar
What is formed in polar bonds?
Dipole- a difference in charge between two atoms caused by shift in electron density
How to determine direction of polarity?
Linear: In direction of uneven distribution of electrons (polar bond)
Several Bonds:
They can cancel each other out if in same direction so no polarity
If all polar bond point roughly in same direction and don’t cancel then charge will be arranged unevenly in that general direction

What are intermolecular forces?
Intermolecular forces are forces between molecules
How does hydrogen bonding affect Boiling and Melting point?
It increases the melting point and boiling point as hydrogen bonds require extra energy to overcome
How hydrogen bonding affect density?
The molecules in water are held by intermolecular forces and when it turns to ice the molecules form a regular lattice structure
As hydrogen bonds are longer than normal bonds, ice is less dense than water
What is the electrical conductivity of simple covalent compounds?
They don’t conduct electricity as there are no free ions or electrons to carry charge
What is the melting point of simple covalent compounds?
Low melting point as the weak forces between molecules are easily broken
What is the solubility of simple covalent compounds?
Some are soluble depending on the solubility of the compound and solvent
What cause percent dipole (polar molecule)?
If charge is distributed unevenly in whole molecule
Polar bonds
Shape