Chem 214: Nucleophilic additions
1/14
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
15 Terms
What are the sections of this topic?
Hydrolysis reactions : 1.1 Esters, 1.2 Amides, 1.3 Nitriles
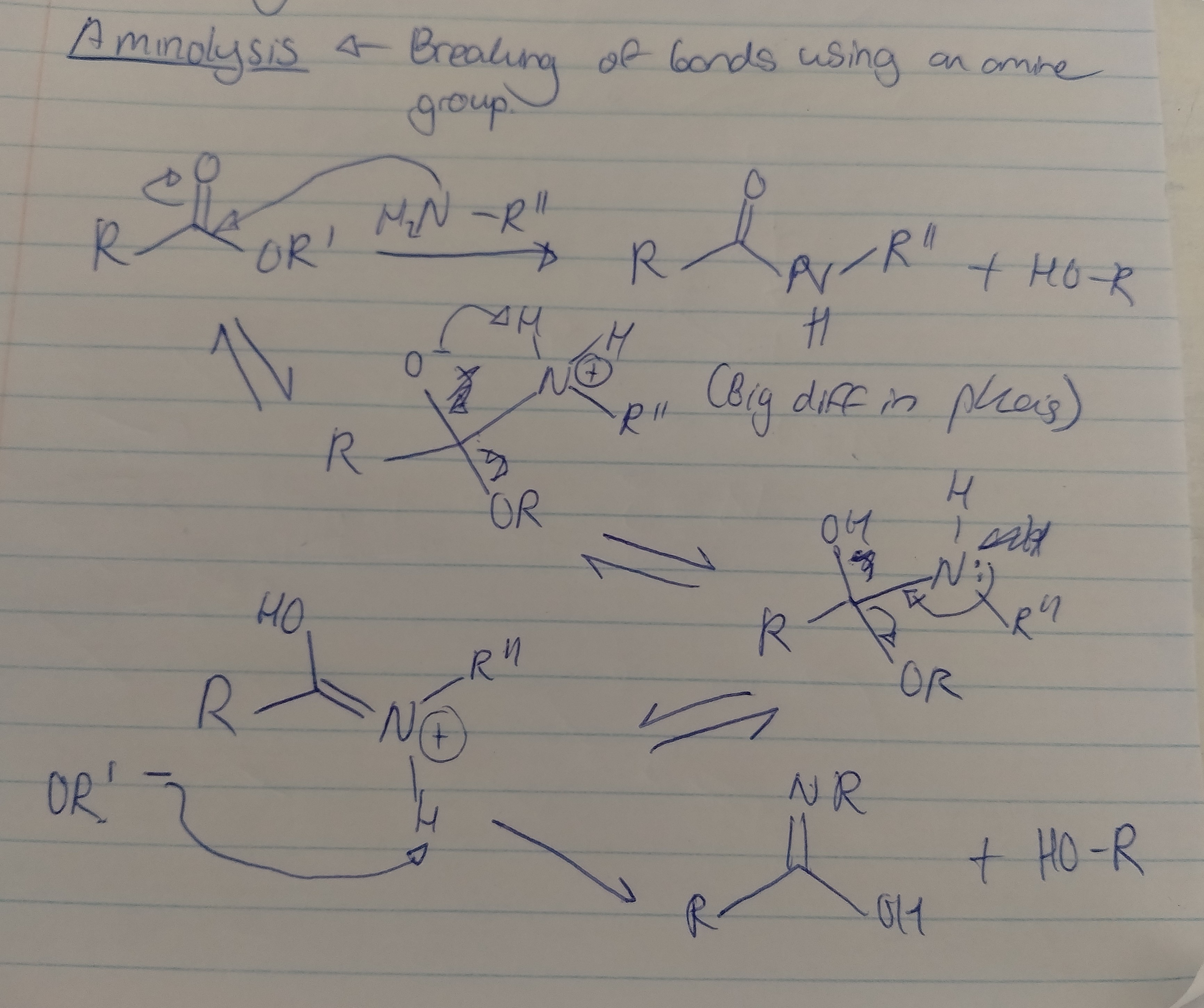
Aminolysis
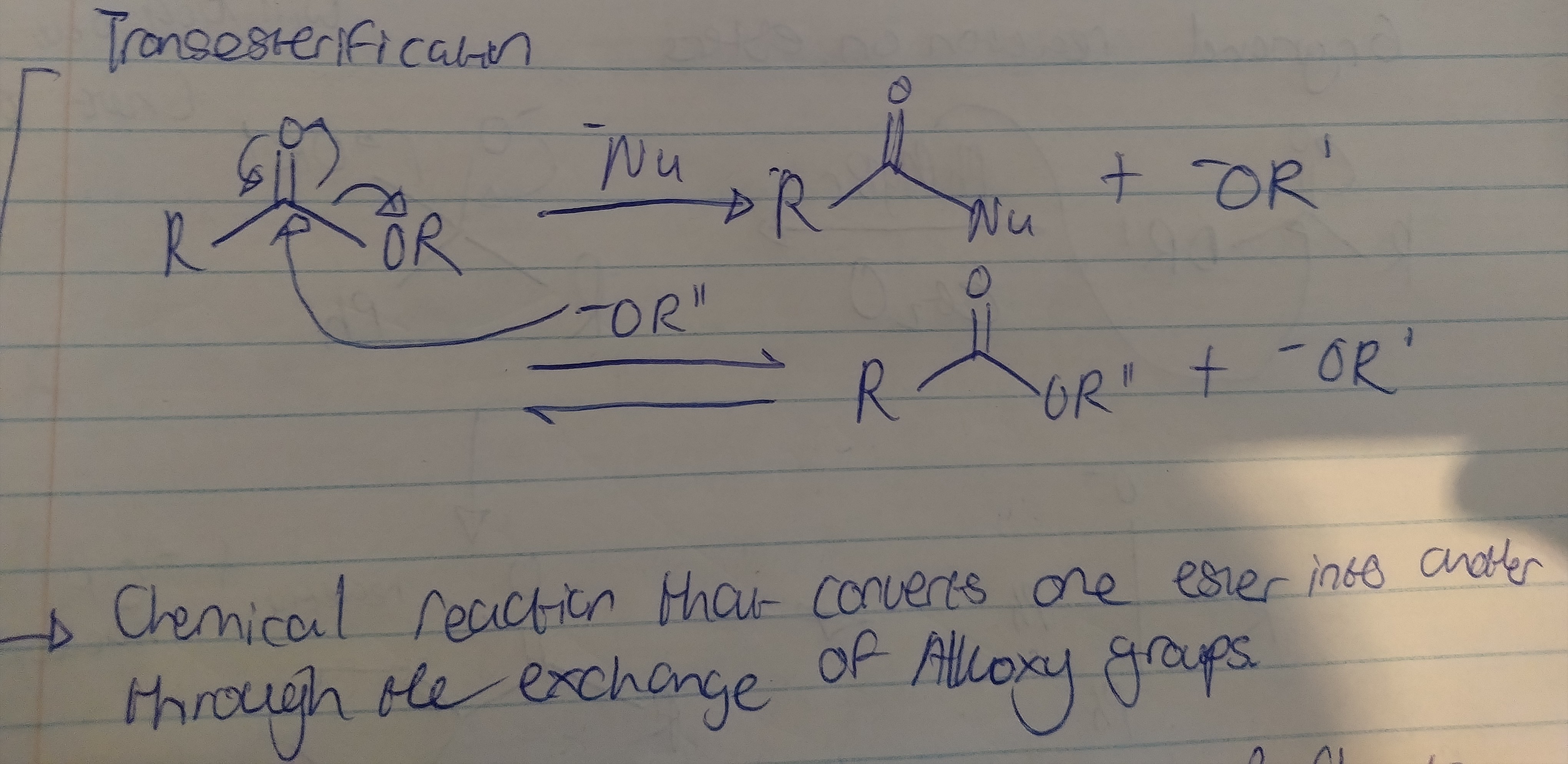
Transesterification
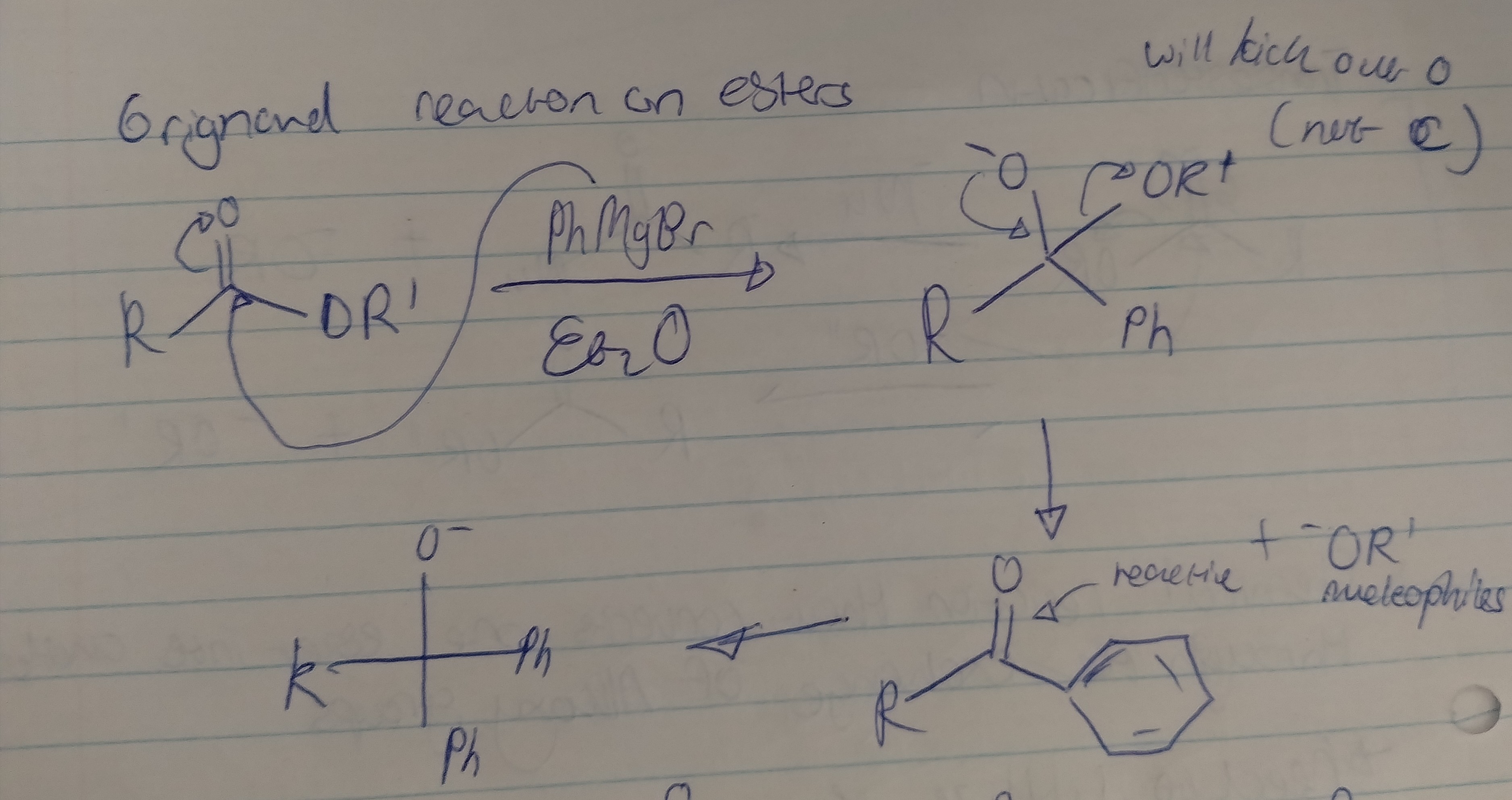
Grignard reactions on esters
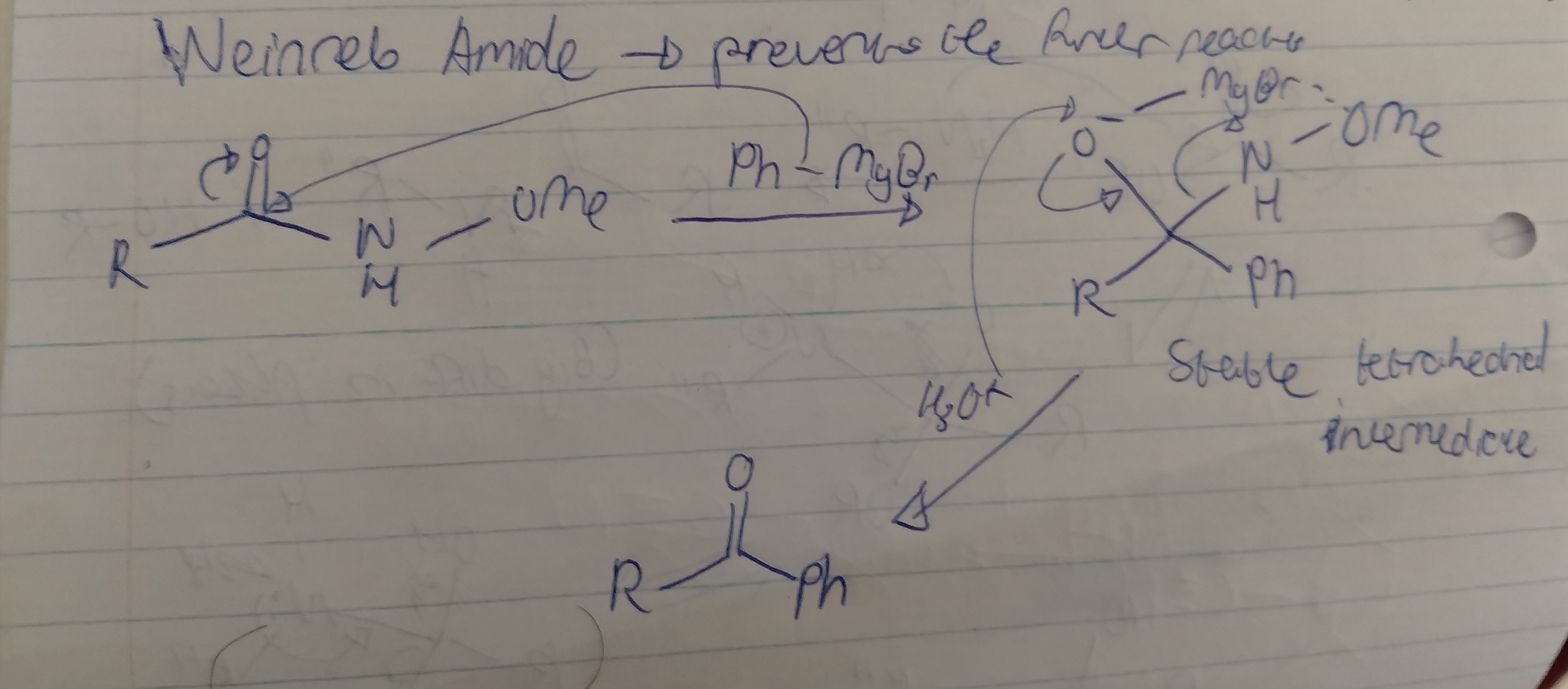
Weinreb Amide reaction
using acid chloride to make esters
making an acid chloride
What is a hydrolysis reaction?
It is the splitting of a molecule into its constituents using water (OH)
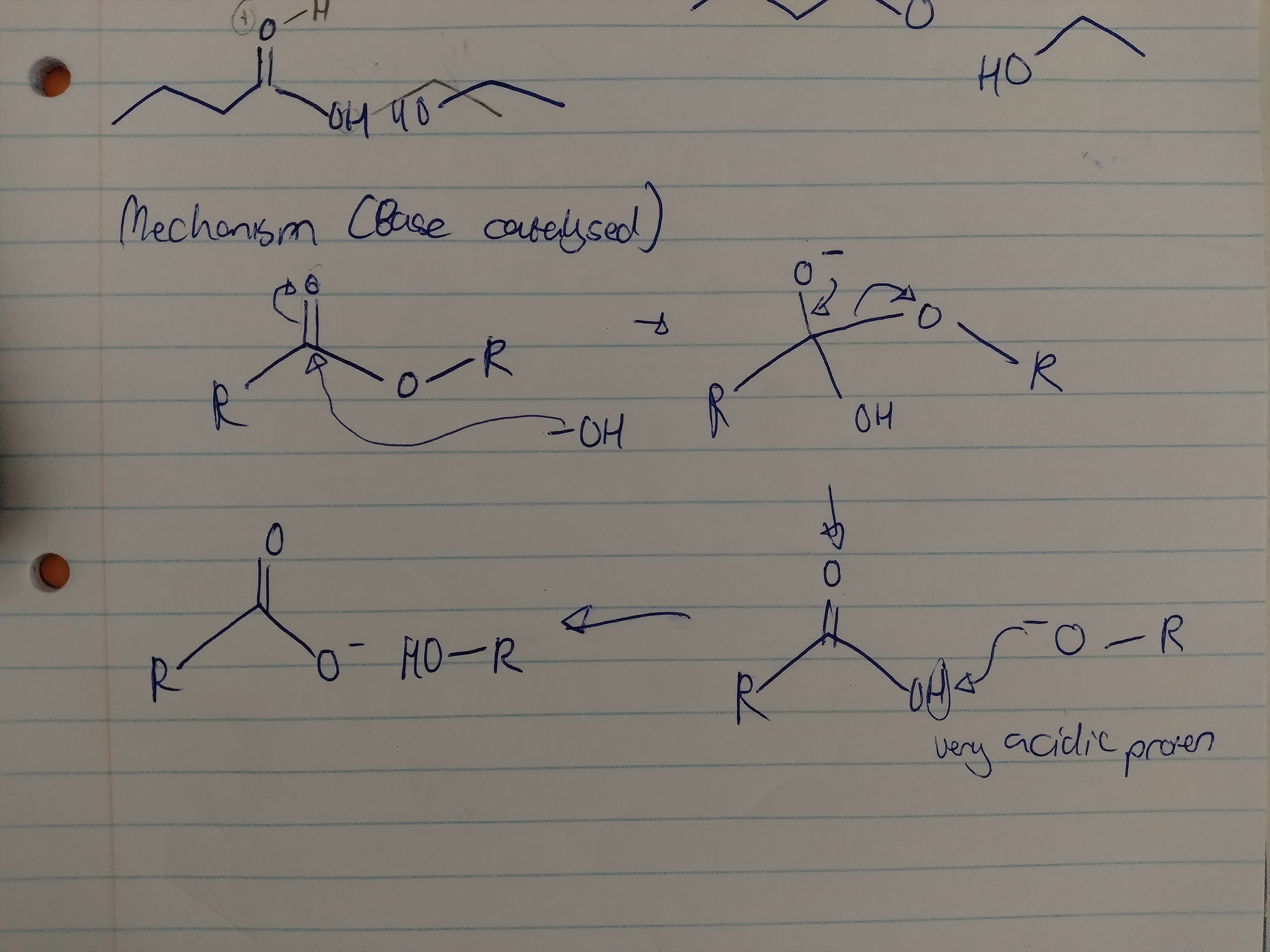
Show the mechanism for the base-catalysed hydrolysis of an ester with all solvents and reagents?
heated, solvent - Methanol or ethanol, strong base like NaOH for catalysis
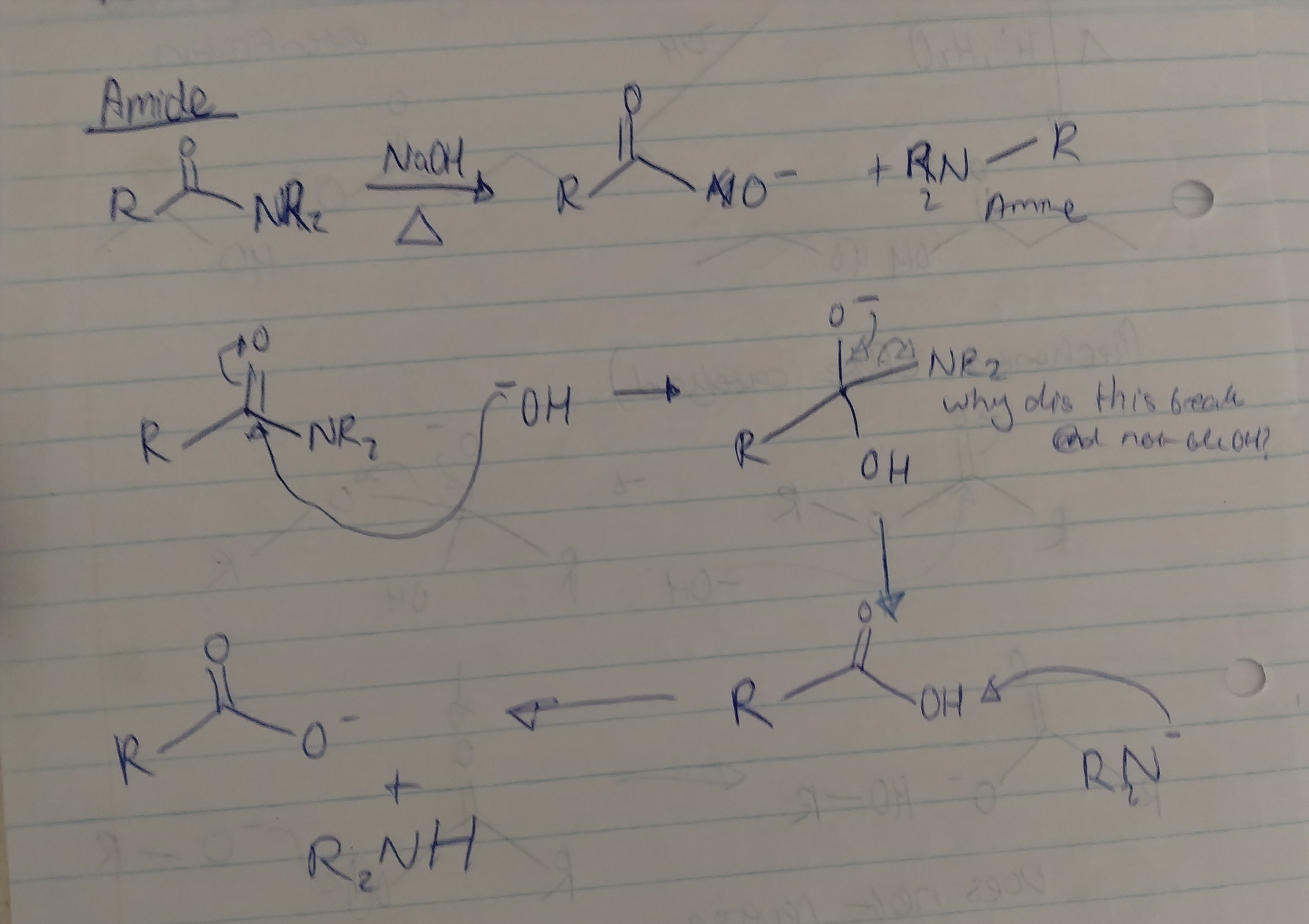
Give the mechanism for hydrolysis of an amide with all necessary conditions, solvents, and reagents.
Strong acid (H2SO4 or HCl), water as a solvent, heating
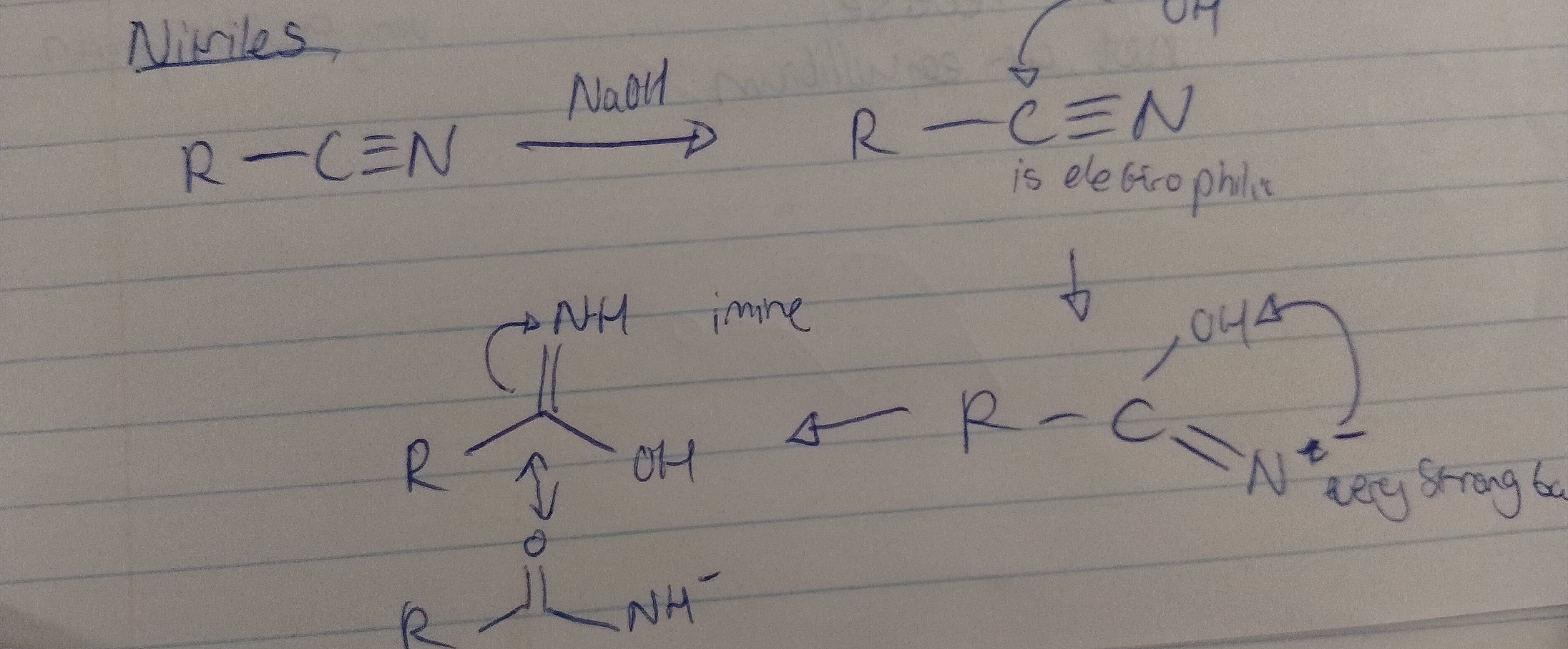
Give the mechanism for the hydrolysis of a nitrile with all necessary solvents, reagents and conditions.
Reagents - strong acid like H3O+ and heat
What are the products of aminolysis?
An amide and an alcohol
Draw the mechanism for aminolysis with all the necessary reagents, conditions and solvents
room temperature, anhydrous solvents (DCM, THF)
What is transestirifcation?
A reaction where an ester reacts with an alcohol to form a new ester and new alcohol.
Draw the transesterification mechanism including all necessary reagents, solvents and conditions
temp - heated, base or acid catalyst
What is the reactivity series?
Acid chloride » Anhydride > aldehydes> ketones> esters » amines
Please draw the mechanism for an ester and Grignard reagent and describe the problem with this reaction?
The product of this reaction is a ketone, which is highly reactive and will continue to react with the Grignard reagent, resulting in an alkoxide. A ketone can be formed using Grignard reagent when reacting it with a Weinreb amide.
Draw the mechanism for the use of a Weinreb amide
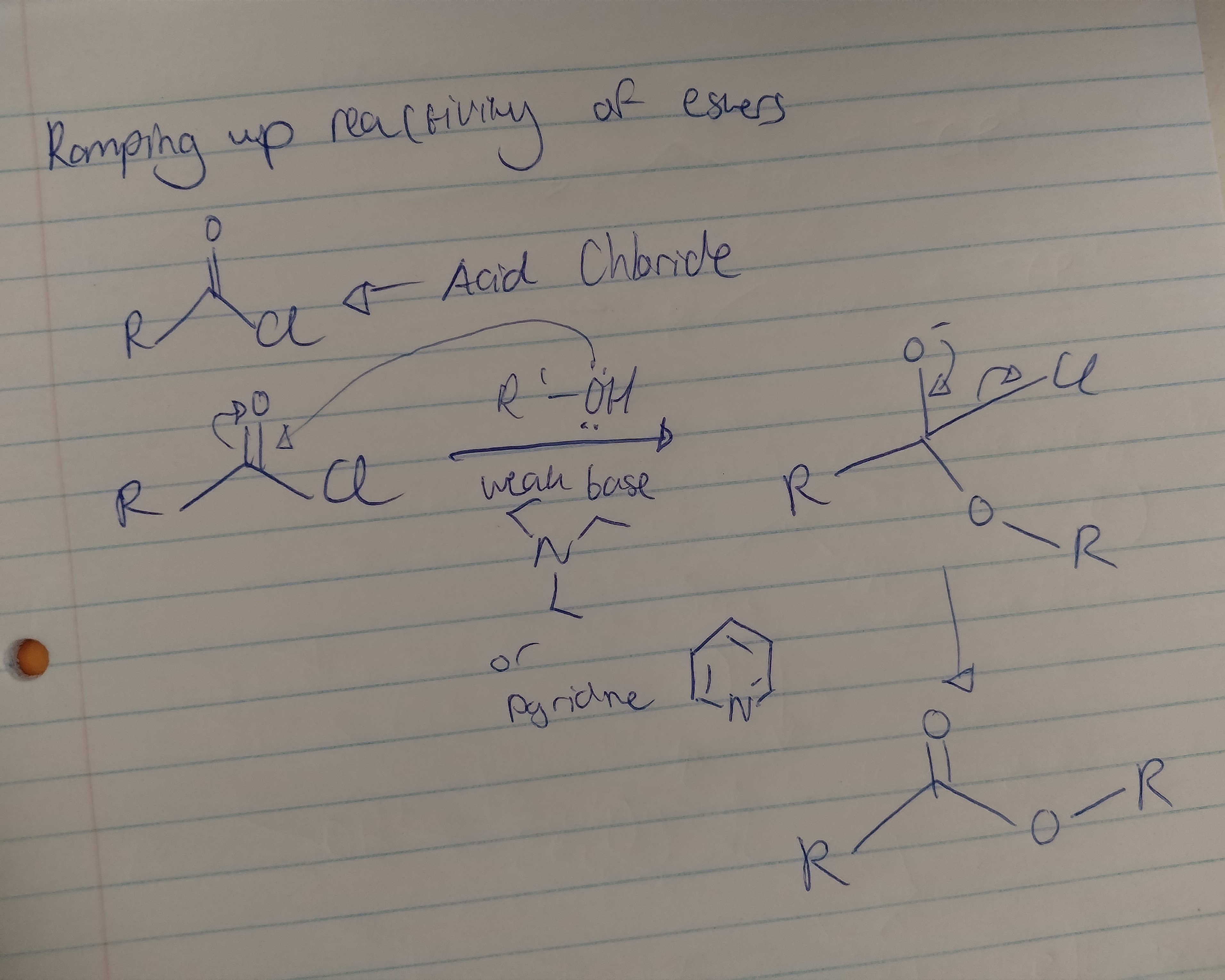
What is the purpose of acid chlorides?
They are highly reactive acylating agents that introduce a RO- to other molecules, facilitating the formation of esters, ketones and amides.
Draw out the mechanism for the reaction of an acid chloride with a primary alcohol
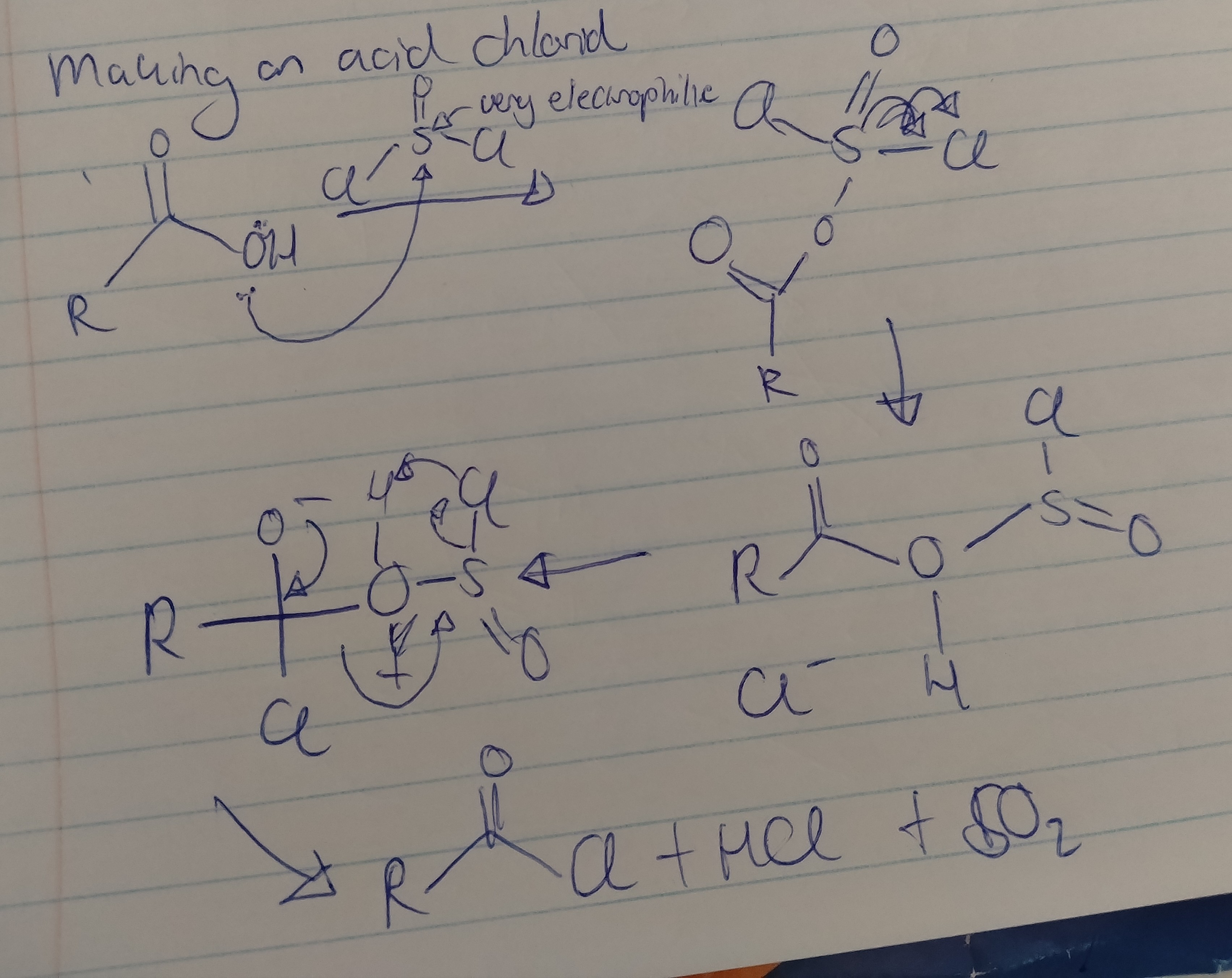
Draw the mechanism for the production of an acid chloride and write down the needed reactants.
A carboxylic acid, and thional chloride.