food chem exam 2 polysaccharides
1/67
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
68 Terms
Define and Classify polysaccharides
Polysaccharides - 10 -20 monosaccharides bonded by glycosidic bond
starch, cellulose, gums
polysaccharide uses in food industry
modify viscosity
form gels
stabilize emulsions
control water
texture
thickening
anti-staling
film formation
foam application
binding
basic structure of starch
is a polysaccaride composed of repeating glucose monomers and has organized units called granules
α 1-4 for linear amylose
α 1-6 for branched chain amylopectin
predominant food reserve in plants and one of our main carb sources
amylose decreased ___
viscosity
amylose has ___ glycosidic bonds
a-1-4
amylose structure
____ but forms helical structure with weak hydrogen bond
linear
amylose is smaller____
than amylopectin
amylose good for ____ because retrogradation and high packing ability
gelling
amylose
____ in cold water but slightly at high temps
insoluble
amylopectin
good for ____ but not gelling because branched chains take too long to reasscociate
thickening and forming paste
amylopectin ____ chains and ___ molecule size
branched, large
amylopectin ____ glycosidic bonds
α 1-6
amylopectin is____ than amylose
bigger
amylopectin glucose phospate derivatives carry ____
negative charges which repell each other and increase starch viscosity
homo-polymer
two examples are ___ and ___
composed of ___ monosaccharide unit with one or more type of glycosidic linkages (repeating glucose units)
can have ___ and un-___
starch (amylose) and cellulose
starch is repeating glucose monomeric units
single
branched
hetero-polysaccarides
composed of ___ monosaccharides
the four types are ___, ___, ___,____
examples include:
more than one type of
copolymers (a-b-a-b)
carrageenan gum
blocked polymers (a-a-a-b-b-b)
alginate gum
branched polymers
guar gum
random polymers (random and branched)
locust bean gum, dietary fiber, pectins
Type A: ___ grains
cereal
corn, wheat, rice
low weight amylose and short chain amylopectin
high degree of packing/crystallinity because short chains
low swelling power
opaque gels upon cooling
Type B: ____ and ____ starches
root and tuber
potato and cassava
high molecular weight amylose and long branched chain amylopectin also more disorganized
low degree of packing/crystallinity
lower gelatinization/pasting temp
high swelling power
highly viscous/paste
weak gel bc fewer amylose molecules can escape into environment
waxy starches
GMO to contain almost all amylopectin
less order and packing in a granule
high swelling power
no or very poor gelatinization bc no amylose to leak out and gel
heavy body and stringy paste
INSTANT PUDDING
iodine stain
amylose - dark blue (iodine gets into the amylose)
amylopectin - red
waxy starch
0-8% amylose basically all amylopectin
paste rather than gel
normal starch
20-30% amylose
some gelling ability
aka native starches and cereal grains
high amylose starch
40-70% amylose (remember 100% amylose is not possible)
very high gelling ability (high temp required tho)
maltese cross
demonstrates crystalline property and the tight packing and extensive H bonding of amylose/lopectin
maltese cross disappears when starch gelatinization occurs
type A starch granules are small, type B are large
why would starch require higher gelatinization temps
more H bonds
higher degree of packing in a granule
longer chains
higher phosphate groups?
gelatinization
when starch is heated sufficiently enough that hydrophobic and H bonds are broken and in the presence of water, the starch granule swells and amylose leaks out
amylose reassociates outside of granule upon cooling and forms gel
pasting
second stage of swelling where starch is heated above gelatinization temp and reaches maximum viscosity, leaches all amylose out into environment and eventually the starch granule completely disrupts
barbender graph
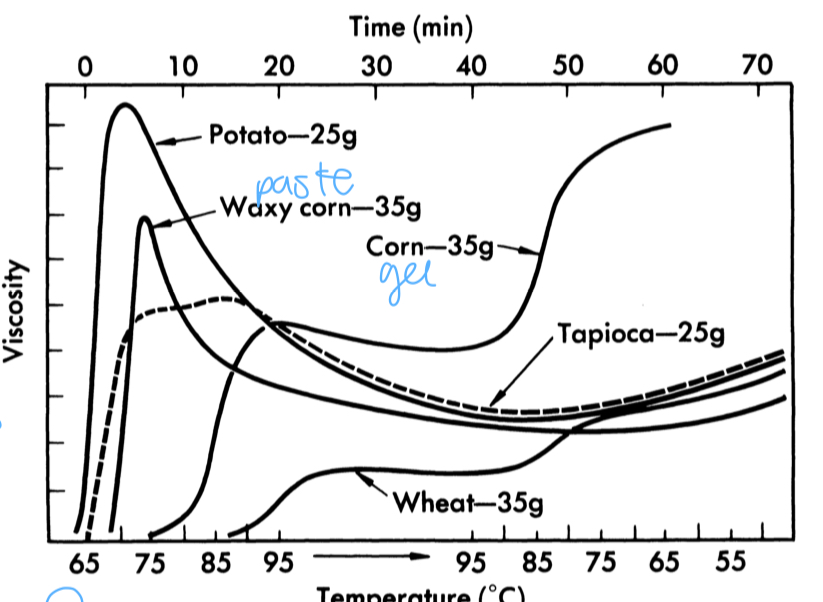
starch retrogradation
if you do not force gel quickly at cooling, amylose is insoluble at cold temp and precipitates out as a solid bc it reforms as an organized structure
more amylose faster retro bc smaller
heating reverses
anti staling agents: lipids and emulsifiers (CMM is example)
stale bread
freezing slows down staling, fridge speeds up
happens when amylopectin branches slowly reassociate upon cooling (bread hardens and gets crumby)
amylose also leeches out and retrogrades almost fully upon cooling which gives fresh bread its elastic and tender crumb
Ingredient interactions with starch
sugar competes w starch which raises gelatinization temp, retards gelatinization, and sucrose has higher effect than fructose bc sucrose is more soluble
proteins and gums also compete for water
lactose increases gel temp but less than sucrose bc poor solubility
acids break glycosidic linkages
limits swelling and lowers viscosity
acid breaks glucose off of starch and glucose is more soluble
ph below 4 acid hydrolysis is complete unless cross linked starch
lipids
coat surface of starch and create waterproof granule
water
if too low, no gelatinization
dextrinization instead
uses of low amylose starch
salad dressing emulsifier
uses of high amylose starch
donut film to prevent oil absorption!!! (cellulose)
quick candy hardening
coating battered products
why do we modify starches
withstand high heat, acids, storage and transport, improve characteristics, freeze/thaw stability, fat replacer, binder
physical modifications of starch
pre gelatinized
instant pudding
chemical modifications of starch: cross link
cross link
acid hydrolysis
acids hydrolize starch into smaller units (oligosacc and D-glucose)
changes from shortened chains include increased sweetness, increased reactivity as reducing sugars, decreased viscositt, increased gelling, increased solubility, increased fermentability, less swelling bc smller units cannot swell as much
amorphous amylopectin regions more hydrolyzed than amylose
simple and cheap but off pigments and caramelization
increased DE (dextrose equiv)
oxidation
chemical mods: enzyme hydrolysis
dextrinization (enzyme hydrolysis)
alpha amylase
endo-enzyme
cleaves at non crystalline 1-4 linkage within starch
more specific than acid
products are dextrins (smaller starch units) mainly maltose and matotriose
beta amylase can cleave at:
exo-enzyme (hydrolyzes starch at non reducing end)
product is maltose and limit b- dextrins
glucoamylase (amyloglucosidase)
exo-enzyme at non reducing end of a-1-4 and 1-6 links
removes b-D-glucose and a-limit dextrins
enzymed used to produce light beers
isoamylase and pullulanase
hydrolyze at a-1-6 bonds (remove branched chains)
chemical modifications of starch: degradation and subs
degradation
esters and ethers
substitutions
esters
products of enzymatic hydrolysis
dextrins
used in splenda! (small amt of sugar molecules but negligible)
maltodextrin
added to splenda to make 1:1 sugar sub
bland no flavor or sweetness but addes bult and good humectant also has fat llike properties
corn syrups
corn starch plus acid and heat or
a-amylase, glucoamylase, fructase
cross linked starch
chemically link starch molecules together with cross bridges
less water is able to enter molecule so minimized swelling
strong due to extensive H bonding
phorylchloride or sodium trimetaphosphate
used in canned soup, spaghetti sauce, pie fillings
phosphorylated starches
negative charge = molecule repulsion = decreased gelling
lower gel but increased viscosity
can drop viscosity with salt to mask phosphate neg charge
good thickening agent for frozen or thawed foods
oxidized starch
bleach flour, less retrodegradation, adheres well to proteins through charge interactions and cross linking
breakfast corn dogs bc neg charged batter binds to meat protein
Hydroxypropylated Starches
Treat starch with propylene oxide
Substitution occurs on the –OH at C2
alkaline ph to avoid dextrinization
Improves starch swelling and solubility
Great cold-storage and freeze-thaw stability = clear paste with no retrogradation
Used in frozen foods and desserts as thickening agent
Also, smooth texture and often used to improve viscosity under acidic conditions
resistant starch
Is a starch and its degradation products that resist digestion in the small intestine
Ferments in large intestine creating butyrate
Feeds gut bacteria • Naturally in foods • Brown rice • Beans • Whole grain bread and pasta • Lentils
types of resistant starch
Type 1 – physically inaccessible starch
Surrounded and entrapped by a cell wall or protein matrix (e.g. legume seeds, coarsely ground grains)
Type 2 – Ungelatinized
Indigestible because of their chemical configuration (e.g. uncooked potato, green banana flour, high-amylose cornstarch or grains)
Type 3 – Retrograded amylose
The heating and cooling of starches during processing render them inaccessible to enzymatic hydrolysis (e.g. cooked and cooled potato, bread, cornflakes)
Type 4 – Chemically modified starch
Derivatives interfere with the binding of the starch to hydrolytic enzymes (e.g. commercial starch products used in processed foods (salad dressing cheese sauces, gravies, soups, etc.))
Type 5 – Amylose-lipid starch
Rigid helical conformation interferes with the binding of the starch to hydrolytic enzymes
homopolymer examples
starch and cellulose
heteropolymers examples
pectin and most gums
gums
a viscous secretion of some trees and shrubs (or from microorganisms) that hardens on drying but is soluble in water (at STP) from which adhesives and other products are made.
cellulose
like starch, insoluble in cold water
cotton, wood
BETA 1-4 glycosidic bonds
linear and rigid (straight chain crystalline regions = hella H bonds)
humans cant digest so passes right through G.I. and no cals
can be added as fiber to breads, no cal bulk for fat free foods
can slightly improve upon heated bc H bonds break and slight swelling but not a ton
modified cellulose
microcrystalline cellulose MCC
Methylcellulose (MC)
Carboxymethyl cellulose (CMC)
MCC
microcrystalline cellulose
prepared via acid hydrolysis
basically amporphous regions removed so just left with crystalline
Emulsifier, Stabilizes emulsions & foams
Fat substitute and extender, Add “body” to foods
can serve as a suspension aid
Absorbs oils & syrups
Anti-caking agent: dry mixes
Keeping them free-flowing (e.g., keep dried honey granules free flowing
MC
methylcellulose
created with very alkaline solution causing fibers to swell
methyl chloride used bc reacts with sodium hydroxide and hydroxyl groups subbed with methyl
Solubility decrease as temperature increases
DONUTS no oil soaked during frying bc gels at higher temperatures and return to solution at lower temperatures
fried meats
CMC
Carboxymethyl cellulose
Cellulose treated with alkali (usually NaOH) to swell fibers and then chloroacetic acid is introduced:
Some of the hydroxyl groups (-OH) are substituted with carboxymethyl ether (CH2CO2H) group
non-digestible fiber in dietetic foods
Hot and cold water soluble
Common stabilizer in ice cream
Foam stabilizer meringues
Tends to interact with proteins due to charge, increasing their viscosity, solubility, & stability
Used to stabilize milk proteins in milk against casein precipitation • Used to stabilize egg whites
cellulose gum on labels
pectin
Complex polysaccharide derived from soft tissue of plants
jams and jelliesss
Linear chain of 1-4 -D-galacturonpryansyl units
high degree of esterification in young unripe fruits
from immature fruit and no gel, to ripe fruit that gels, to overripe fruit and no gel: protopectin, pectin (pectinic acid), and pectic acid
DE decreases as aging process happens
low PH
high sugar
low DE methoxyl pectin gels need divalent cation such as calcium to gel (good for diet jams bc u dont need sucrose)
pectin esterase removes cloudiness but u have to heat product to stop enzyme
gums origins
Seaweeds (agar, carrageenan)
Seeds (guar and locust bean gums)
Microbes (xanthan and gellan gums)
Modified starch and cellulose
gum properties
very hydrophilic
good gel formers
(a) – stiff extended chain with a large radius that interacts with many water molecules and chains thus producing a large increase in viscosity
(b) – chain that folds into a compact shape – only has a small effect on viscosity
(c) – branching increases stiffness but reduces the radius. Lesser effect on viscosity than (a)
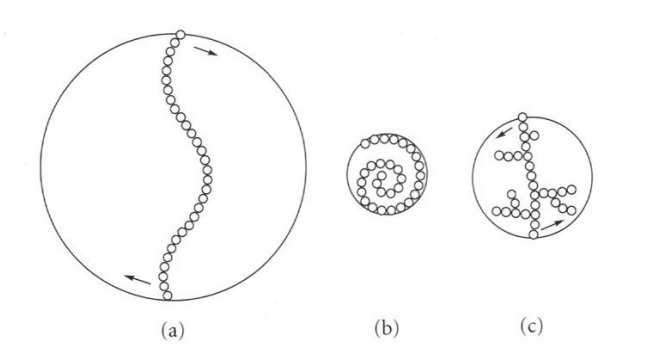
gum interactions
Non-ionized gums = little effect of pH and salts
ionized Negatively charged gums
Low pH = deionization = aggregation = precipitation
Can modify by placing a strong acidic group on gum so it remains ionized at low pH (important in fruit juices)
High pH = highly ionized = soluble = viscous
Ions (e.g. Na+ , K+ , Ca2+) = salt bridges = gels
Proteins •
CMC Carboxymethylcellulose inhibits in-solubilization of proteins in fruit flavored milk drinks
Flours with poor bread making properties can be increased (xanthan or carrageenan)
Sugars
Salt
chewing gum
Modern chewing gum is composed of:
Gum base (25-35%),
Sweeteners (40-50% if caloric sweetener,
0.05-0.5% if artificial sweetener),
plasticizer (1-2%),
flavor (1.5-3%)
color (optional)
Coating (starch, sorbitol, mannitol)
Plasticizer softens the gum and increases the flexibility (lecithin, hydrogenated vegetable oil, glycerol etc.)
ionic gums
alginate, carrageenan
non-ionic gums
guar gum, locust bean gum, gum arabic
alginate
brown algae/kelp
used in pudding for gel texture, ice cream, “caviar”, foam stabilizer, salad dressing stabilizer
forms gels at room temp if introduced to di or trivalent cations
carrageenan
red seaweed
KIL kappa iota lambda polymers
cold water soluble and does not gel
stabilizes proteins
MILK and DAIRY PRODUCTS
guar and locust bean gum
No effect of pH and ions (salts) since they are uncharged
soluble in hot and cold water
salad dressings (thixotropic = thickens up at rest but thins out if agitated)
guar gum 2:1 mannose to galactose
locust bean gum 4:1 mannose to galactose but un-uniform distribution
locust + carrageenan or xanthan = lunch meat binder
carob tree is source of guar gum (carat name origin)
gum arabic
One of the oldest known gums mummy wrappings
Produced naturally as a teardrop shaped globule extruding from the bark of the tree when it has been injured
Very large complex polymer
Readily dissolves in water
Pseudoplastic behavior
Notable b/c does not significantly increase viscosity until high concentration
starburst jellybeans
lozenge
retards sugar crystallization
beverage powders
xanthan gum
branched ionic
Produced by Xanthomonas species a microbe that lives on leaves of cabbage plants
Soluble in hot and cold water
Cellulose backbone with charged trisaccharide branches
Branching prevents gelation
Very viscous due to charged branches
Expensive ingredient
Very high viscosity at low concentrations
remain stable when sitting, but flow when poured
Chocolate sauces – constant viscosity over wide temperature ranges
Easily flowable at refrigeration temperature