Chemistry- All about Alkenes
1/51
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
52 Terms
What is the product of propene treated with H₂ / Pd (or Pt, Ni)?
Propane (alkene → alkane via hydrogenation)
What is the major product of propene + HBr (no peroxides)?
2-bromopropane (Markovnikov addition of H and Br)
What is the product of propene + HBr / ROOR (peroxides)?
1-bromopropane (anti-Markovnikov addition)
What is the product of propene + H₂O / H₂SO₄?
2-propanol (Markovnikov alcohol addition)
What is the product of propene + 1. Hg(OAc)₂, H₂O 2. NaBH₄?
2-propanol (Markovnikov hydration, no rearrangement)
What is the product of propene + 1. BH₃·THF 2. H₂O₂, OH⁻?
1-propanol (anti-Markovnikov alcohol, syn addition)
What is the product of ethene + Br₂ (in CCl₄)?
1,2-dibromoethane (vicinal dibromide, anti addition)
What is the product of propene + Br₂ / H₂O?
1-bromo-2-propanol (OH on more substituted carbon)
What is the product of ethene + mCPBA?
Ethylene oxide (epoxide ring)
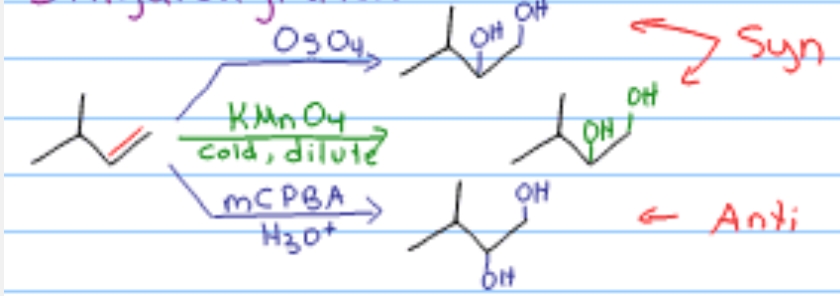
What is the product of cyclohexene + KMnO₄ (cold, dilute) or OsO₄?
cis-1,2-cyclohexanediol (syn diol)
What happens to 2-butene + ozone (O₃), then Zn/H₂O?
Two molecules of acetaldehyde (CH₃CHO)
What happens to terminal alkene CH₂=CHCH₃ + hot KMnO₄?
CO₂ + acetic acid (complete oxidative cleavage)
What is the general product of alkene combustion in excess oxygen?
CO₂ + H₂O
Which alkene reactions give anti addition?
Halogenation (Br₂/Cl₂) and halohydrin formation (Br₂/H₂O)
Which alkene reactions give syn addition?
Hydrogenation (H₂/Pd), hydroboration-oxidation, and syn dihydroxylation (OsO₄/KMnO₄ cold)
Which hydration method avoids carbocation rearrangements?
Hydroboration-oxidation OR oxymercuration-demercuration
Name the reagent: Hydrogenation of an alkene to an alkane.
H₂ / Pd, Pt, or Ni
Name the reagent: Anti-Markovnikov addition of HBr to an alkene.
HBr / ROOR (peroxides)
Name the reagent: Markovnikov hydrohalogenation
HBr (no peroxides)
Name the reagent: Hydroboration-oxidation
BH₃·THF then H₂O₂, OH⁻
Name the reagent: oxymercuration-demercuration
Hg(OAc)₂, H₂O
NaBH₄
Name the reagent: Halogenation of alkene
Br₂ in CCl₄
Name the reagent: Formation of a halohydrin from an alkene
Br₂ / H₂O (or Cl₂ / H₂O)
Name the reagent: Epoxidation of alkene → epoxide
mCPBA or RCO3H
Name the reagent: Syn dihydroxylation (cis diol formation)
OsO₄ OR cold, dilute KMnO₄
Name the reagent: Ozonolysis
O₃ then Zn/H₂O
Name the reagent: Oxidative cleavage producing carboxylic acids / CO₂
Hot, concentrated KMnO₄
Name the reagent: Acid-catalyzed hydration (Markovnikov alcohol formation)
H₂SO₄, H₂O
Name the reagent: Halohydrin formation
Br₂ / H₂O
Name the reagent: Anti-Markovnikov hydration of an alkene (to form alcohol).
BH₃·THF
H₂O₂, OH⁻
Which reactions give anti-Markovnikov alcohol formation?
Hydroboration-oxidation (BH₃·THF then H₂O₂, OH⁻)
Which of the alkene reactions has syn addition?
Hydrogenation → H₂ / Pd, Pt, Ni
Hydroboration-oxidation → BH₃·THF, then H₂O₂ / OH⁻
Syn dihydroxylation → OsO₄ OR cold dilute KMnO₄
Which of the alkene reactions has anti-addition?
Halogenation → Br₂ or Cl₂ (in CCl₄)
Halohydrin formation → Br₂ / H₂O or Cl₂ / H₂O
Ozonolysis (conceptually anti before cleavage) → O₃, then workup
which of the alkene reactions has anti-mark rule?
Hydroboration-oxidation
BH₃·THF, then H₂O₂ / OH⁻
→ OH goes to less substituted carbonHBr / peroxides (ROOR)
→ Br goes to less substituted carbon
Which of the alkene reactions has stereochemistry?
Hydrogenation (syn)
Hydroboration-oxidation (syn, anti-Markovnikov)
Halogenation (anti)
Halohydrin formation (anti)
Syn dihydroxylation (syn)
Which alkene reactions involve cleavage?
Ozonolysis → O₃, then Zn/H₂O or DMS
→ aldehydes and ketonesHot, concentrated KMnO₄
→ carboxylic acids / ketones / CO₂ (for terminal alkenes)
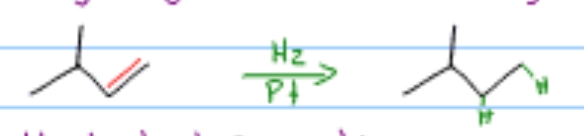
Which alkene reaction is this?
Hydrogenation
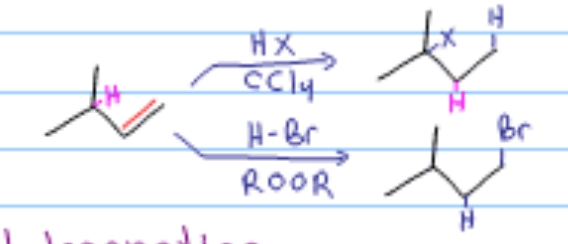
Which alkene reaction is this?
Hydrohalogenation
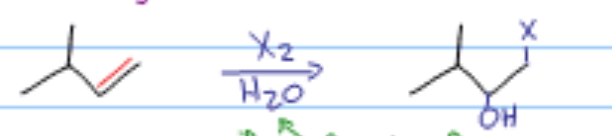
Which alkene reaction is this?
halohydrin
Which alkene reaction is this?
Halogenation
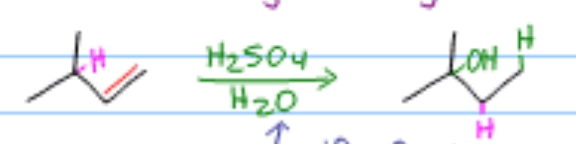
Which alkene reaction is this?
Acid-Catalyzed Hydration
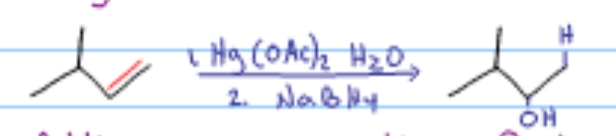
Which alkene reaction is this?
Oxymercuration-Reduction

What reaction is this?
Hydroboration-Oxidation
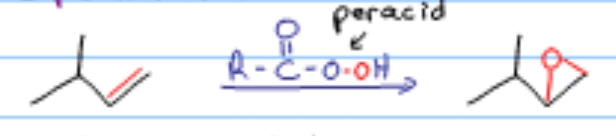
What reaction is this?
Epoxidation
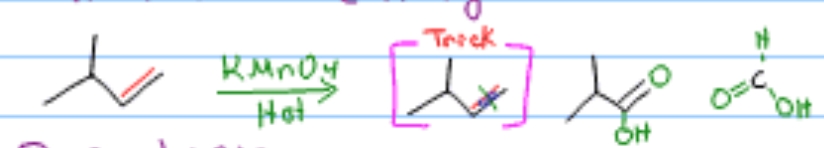
What reaction is this?
Oxidative Cleavage
What reaction is this?
Dihydroxylation
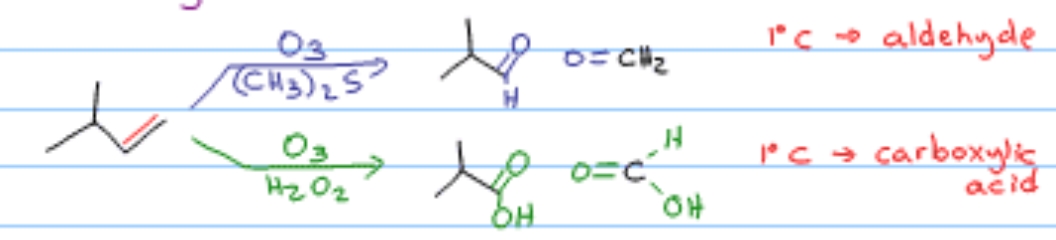
What reaction is this?
Ozonolysis
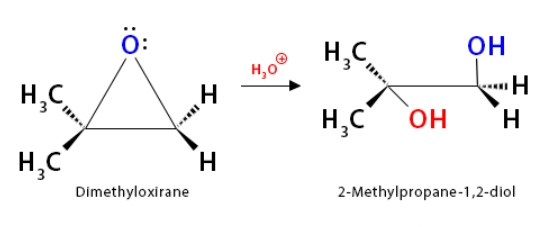
What reaction is this?
Hydroxylation by acid-catalyzed epoxide hydrolysis
Which has the highest boiling point: ethene, ethanol, or benzene, and why?
Ethanol
Why do alkenes generally have much lower boiling points than alcohols of similar molecular weight?
Alkenes only have London dispersion forces, while alcohols can form hydrogen bonds, which are much stronger intermolecular forces.
Why does benzene usually have a higher boiling point than a similar-sized alkene?
Benzene has stronger electron cloud interactions due to aromaticity and π-electron delocalization, leading to stronger dispersion forces and better stacking than simple alkenes.
Rank the solubility in water: alkene, alcohol, benzene. Explain briefly.
Alcohol > alkene > benzene
Alcohols: hydrogen bonding → high solubility (especially small ones)
Alkenes: nonpolar → very low solubility
Benzene: nonpolar and hydrophobic aromatic ring → extremely low solubility