5.3 Limiting Reactant
1/5
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
6 Terms
Limiting Reactant
The reactant that yields the smallest amount of product, “limiting” the amount of product made as a whole
Theoretical Yield
The amount of product that could be made given a perfect environment (no impurities, procedure errors, etc.)
Actual Yield
The amount of product that is actually made. Usually given as experiment results (not calculatable)
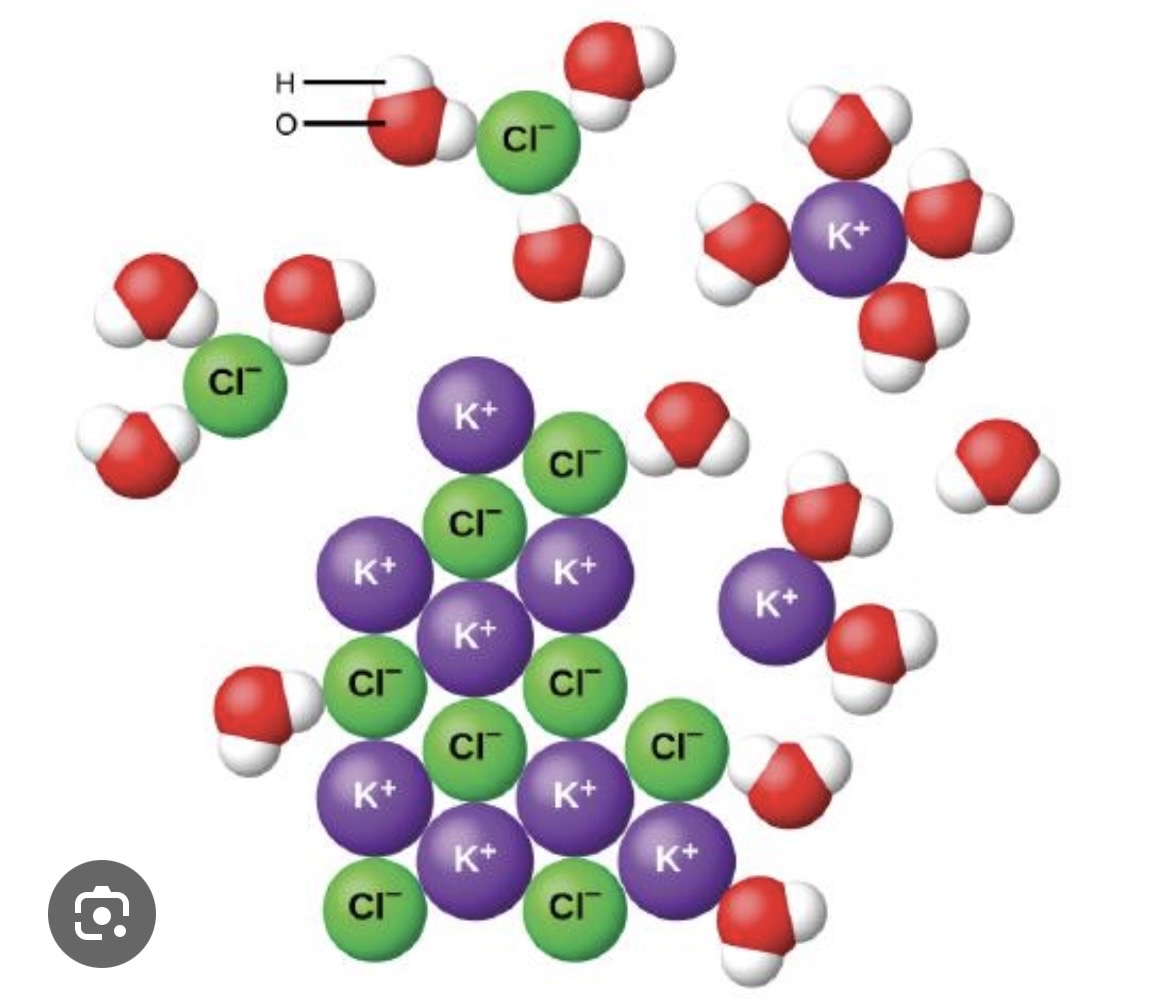
What do the separated ions represent?
Aqueous-state ions
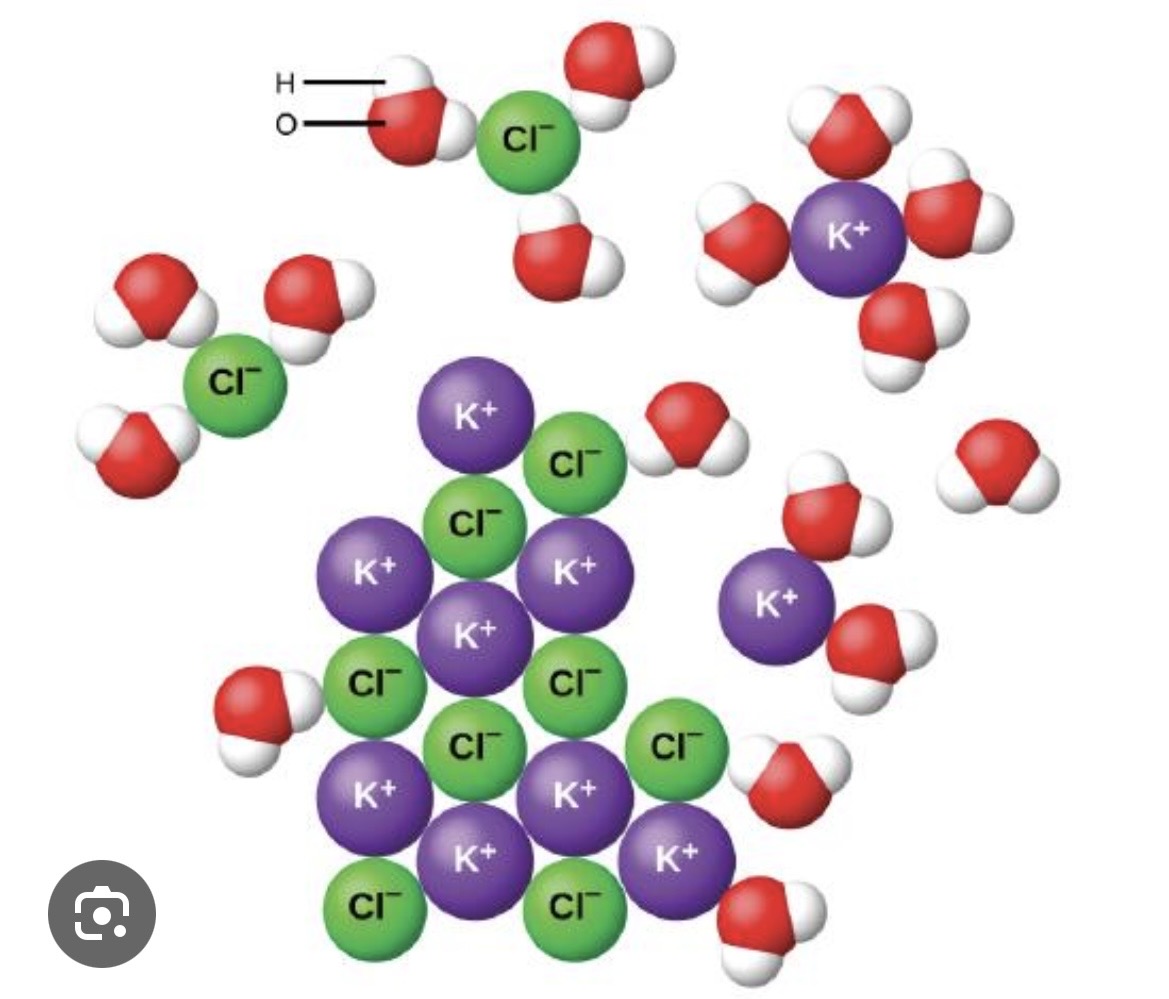
Ionic Compounds and Water
Water weakens and overwhelms the ionic bonds in an ionic compound, separating the ions and aquefying them.
Percent Yield
Calculated by (Actual Yield)/(Theoretical Yield). Finds how much reactants actually became product.