Functional Groups
1/27
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
28 Terms
How many carbon bonds does a Alkane molecule have?
Has a single Carbon Bond
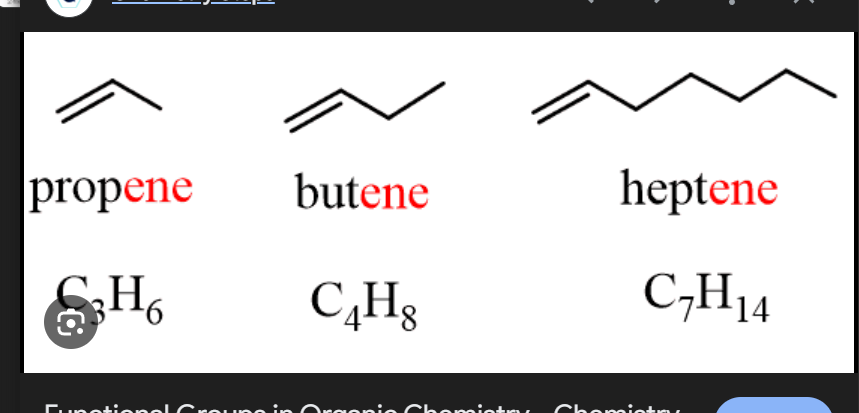
How many carbon bonds does a Alkene molecule have?
Double Carbon Bonds
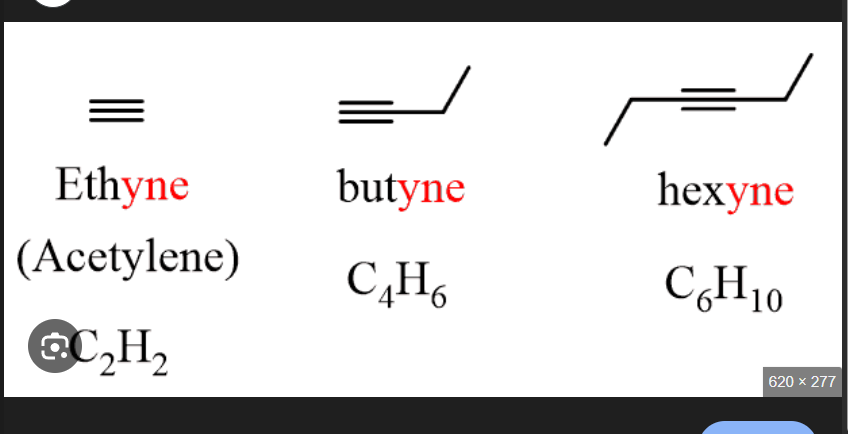
How many carbon bonds does an Alkyne molecule have?
Triple Bonds
How many carbon atoms are there in the hydrocarbon prefix “Meth.” Ex: CH4
1 Carbon Atom. Hence no skeletal structure
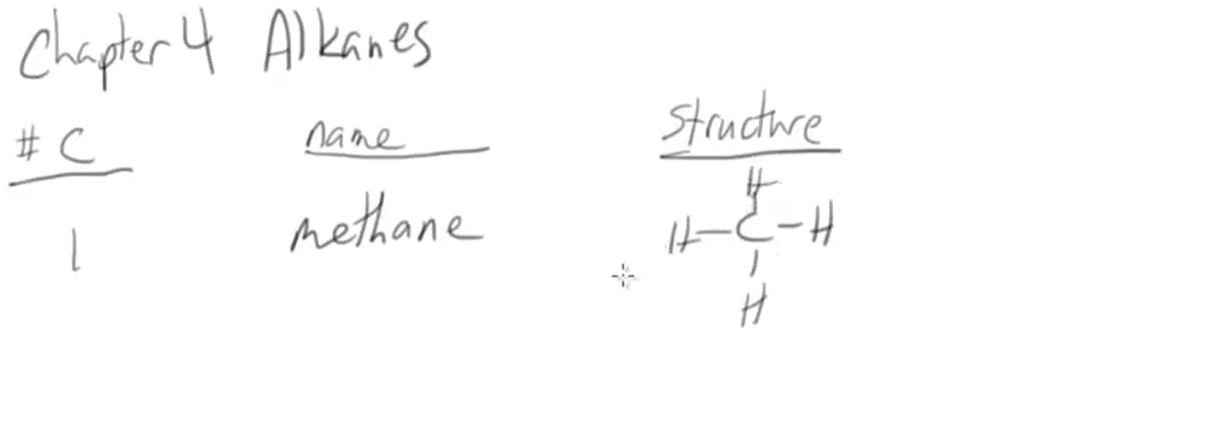
How many carbon atoms are in Ethane?
2 Carbon Atoms
Propane. How many carbon atoms?
3 Carbon atoms
Butane
4 carbon atoms
Pentane
5 carbon atoms
Hexane
6 carbon atoms
Heptane
7 Carbon atoms
What does R represent in a carbon containing group?
CH3 (Methyl)
CH3CH2 (Ethyl)
Long Carbon Chain
Benzene Ring
What’s the general formula of aldehyde?
R-CHO
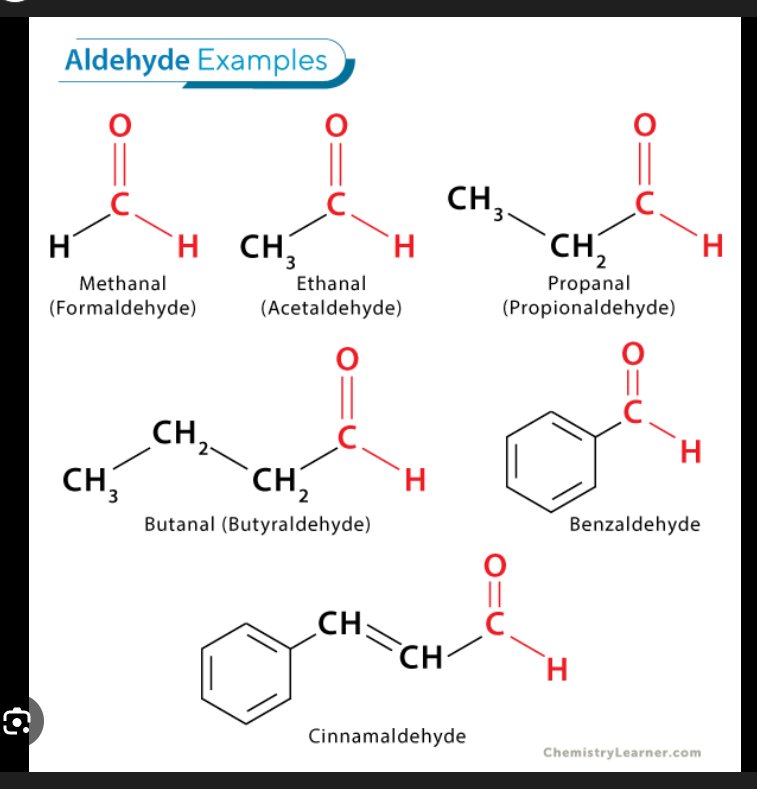
Structural nature of Aldehyde group
CHO group is always at the end of the carbon chain. There’s a double bond with Oxygen. Never in the middle.
General Formula for Ketone
2 carbon groups (R–CO–R')
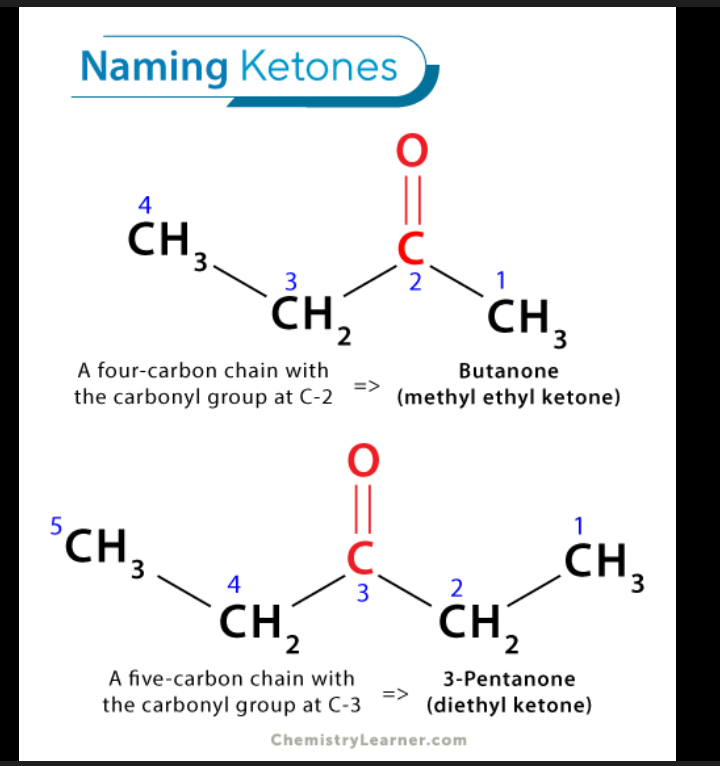
Structural nature of ketone
If the C=O is in the middle of the chain, it's a ketone.
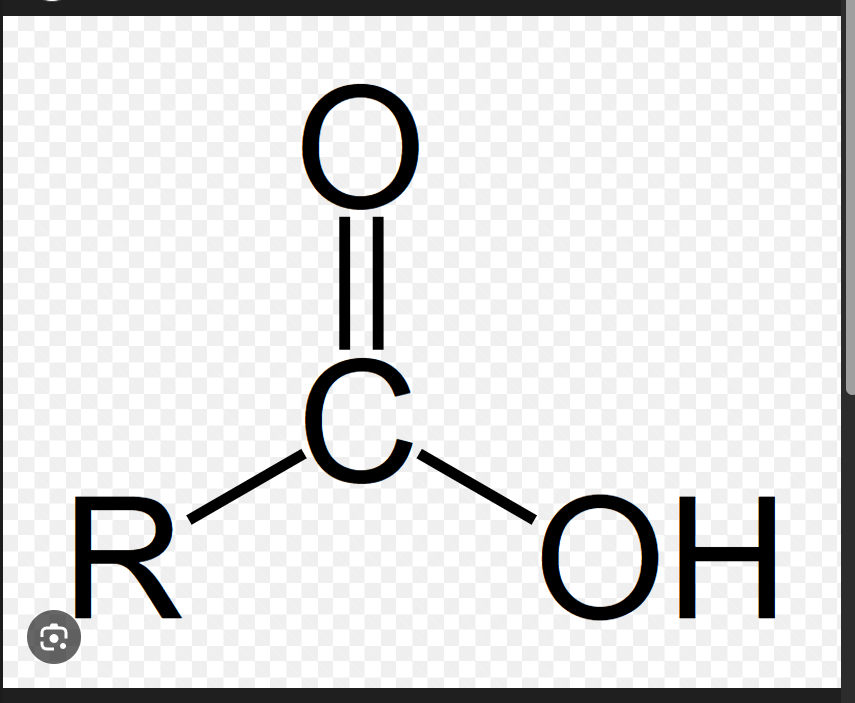
Carboxylic acid General Formula
RCOOH
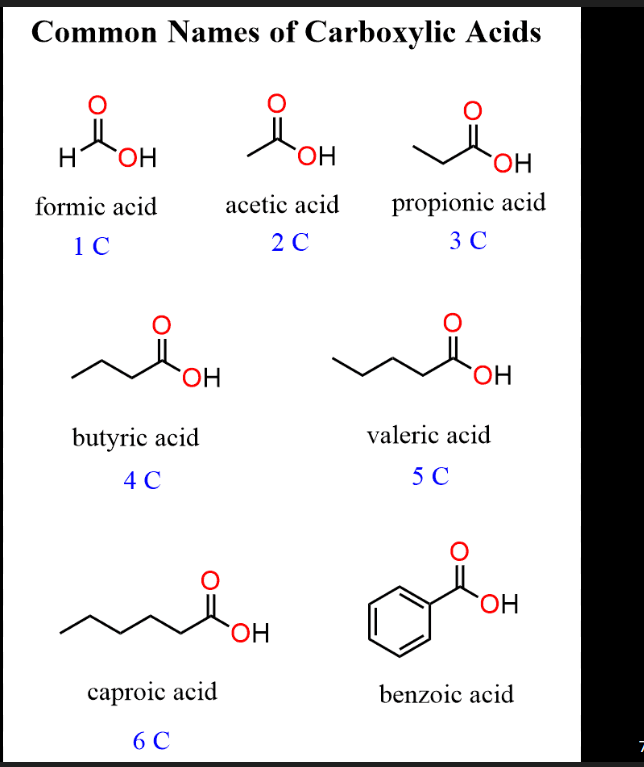
Carboxylic acid structural nature
Also has –CHO-like notation, but there's an –OH attached to the carbonyl carbon (–COOH). No H directly on the carbonyl carbon.
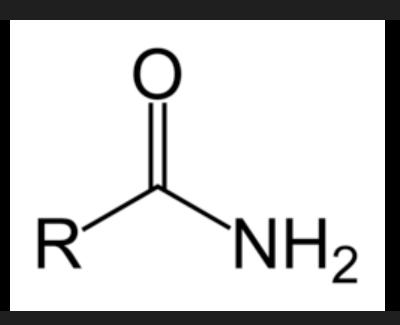
Ester/Amide structural nature
Esters have the general structure R-COO-R', where the carbonyl carbon is single-bonded to an oxygen which is then connected to another carbon group.
Naming an ester molecule
-yl -oate
Alcohol General Formula
R-OH
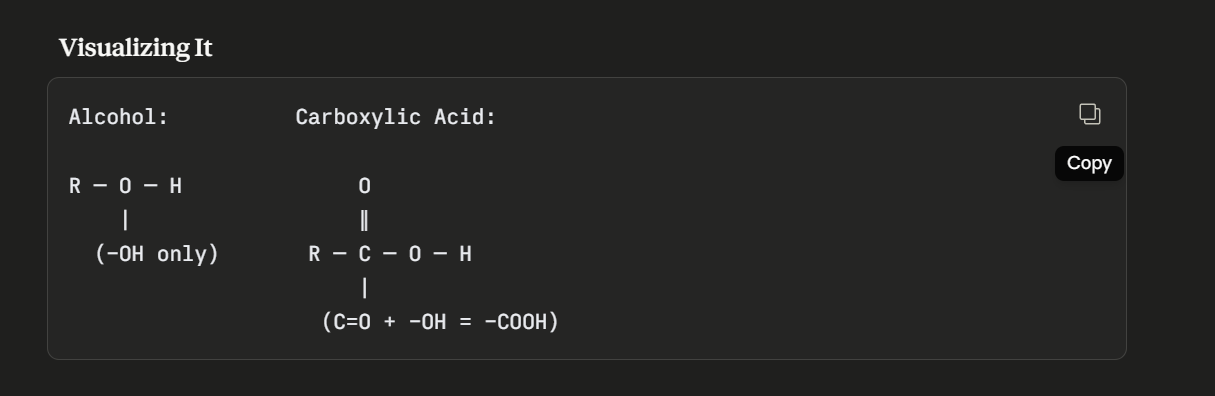
Briefly explain the difference between an alcohol and a carboxylic acid.
Alcohol only contains hydroxyl group (-OH), but no carbonyl group (C=O). Carboxylic acid contains both of these groups in the same carbon function group (-COOH).
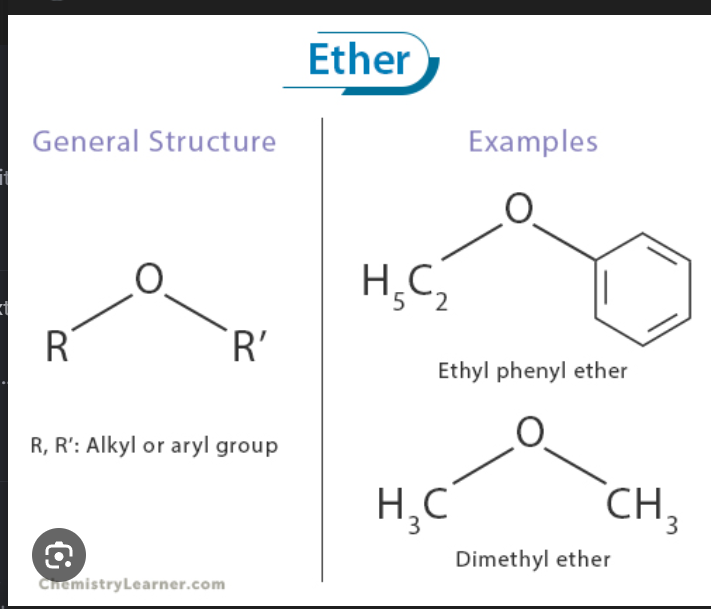
Ether
R-O-R^1. There’s only single bonds
List all of the Strong Acids
HClO4
HCl
HBr
HI
HNO3
H2SO4
List all of the Strong Bases
LiOH
NaOH
KOH
Ca(OH)2
Sr(OH)2
Ba(OH)2
What makes a molecule a CIS fatty acid?
Hydrogen atoms are on the same side of the double carbon bonds
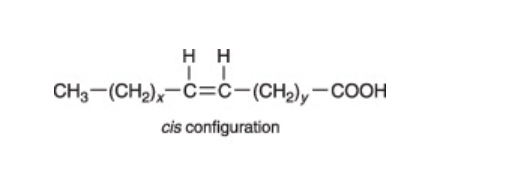
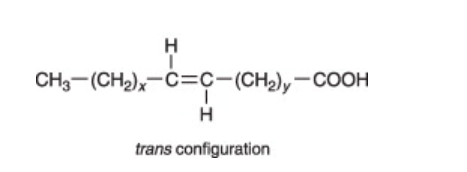
What makes a molecule a trans fatty acid?
Hydrogen atoms are on the opposite side of the double carbon atoms
Hydrophobic Group(s)
R-CH3
Hydrolpillic Groups
-OH, -COOH, -NH2