Biophysics Lecture 7 Revision
1/25
Earn XP
Description and Tags
Use this.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
26 Terms
Concrete mechanisms of facilitated passive transport
Endogenic carriers (for proteins translated in ribosomes) → Glucose carrier (GLUT-1), aquaporins, and potassium (K+), sodium (Na+ ), and calcium (Ca2+) channels
Exogamic carriers (non-ribosomal translated proteins) → Valinomycin and Gramicidin A
Transport of glucose in erythrocytes
Glucose is mainly transported into the erythrocyte membrane through the protein GLUT-1, a carrier protein that utilizes facilitated diffusion to move glucose molecules from an area of high concentration of glucose (in the ECM) to an area of low concentration of glucose (cytoplasm of the erythrocyte). In order to achieve this, the carrier protein changes conformation, going from facing the outer leaflet of the bilayer to facing the lower leaflet once glucose molecules bind to it.
Gluconeogenesis
When the body does not receive enough glucose (when glycogen stores are depleted - after around 6-8 hours of prolonged fasting) from the diet, it begins to produce glucose from non-carbohydrate precursors (like lactate, glycerol, and glucogenic amino acids). This is necessary for the survival of the brain and red blood cells, and is performed mostly by the liver (and kidneys).
GLUT-2 (the membrane protein primarily found in hepatocytes, renal tubular cells, and pancreatic beta cells) is activated during and after prolonged fasting. It allows glucose to pass in 2 directions (both in and out).
GLUT-3
Found mostly in neurons and the placenta.
GLUT-4
Found primarily in the adipose tissues of the hypodermis and striated muscles (cardiac and skeletal).
The rate of substrate (molecular) transport, apart from the concentration gradient, is dependent upon:
1) Quantity of carrier proteins and glucose molecules → The more present, the faster transport occurs.
2) Rate → Vmax provides the maximal rate and limits how much glucose can be transported into cells (uptake).
3) Affinity (specificity) → The affinity of the transport protein towards the substrate molecule (considering conformational changes of the carrier protein - if open on one side, corresponding glucose molecules on that side have a higher affinity than on the opposite side). The interaction coefficient of the substrate and carrier molecules determines this. The affinity between these is the inverse of Km.
The effect of substrate concentration on enzyme activity in the case of passive diffusion
The more substrate is present, the more it binds to enzymes, increasing rate of reaction. This happens in a linear fashion on a typical line graph.
Enzyme-catalyed reactions and the “saturation effect.”
As substrate concentration increases, initially, the rate of reaction increases significantly. Eventually, however, with higher substrate concentration, the rate of reaction stagnates (plateaus), because most or all of the enzymes in a given area have their active sites completely occupied by substrates, becoming “saturated.”
This yields a hyperbolic curve.
Km (Michaelis Constant)
The concentration of the substrate that allows the rate of reaction to reach half of Vmax.
It is an inverse measure of affinity. This means that an anzyme with a high Km has a lower affinity for its substrate and requires a greater concentration of substrate to reach Vmax.
Enzymes with low and high Km
Enzymes with low Km reach saturation much faster (meaning high affinity for substrate), so the enzyme more or less acts at a constant rate regardless of substrate concentration.
Enzymes with high Km reach saturation much slower (meaning low affinity for substrate), so the enzyme’s rate of reaction is dependent on substrate concentration.
Remember these are all relative to the physiological concentration of these substrates (how much is in the body).
GLUT-1
The main carrier protein found primarily in erythrocytes that allows the import and export of glucose.
GLUT-2
Plays major roles in import and export of glucose during gluconeogenesis. Transports glucose in 2 directions (in and out). Found primarily in renal tubular cells, hepatocytes, and pancreatic beta cells.
1) Determining number of enzymes in a given area (of tissue) and 2) number of substrate in a given area
1) To determine the number of enzymes, the limiting factor is the activity of the enzyme itself and not the concentration of substrate. The concentration of substrate must be high enough so that the enzyme is acting at Vmax.
2) Enzymes are used to determine the amount of substrate in a sample. For example, glucose oxidase helps measure the amount of plasma glucose. The limiting factor is the substrate. The concentration of substrate must be below Km so that the rate of formation of a product increases steeply.
Michaeles-menten Equation
Where Km represents the Michaelis constant, and S represents the concentration of the substrate.
On a traditional graph, note that V represents the rate of reaction, while S represents the substrate concentration.
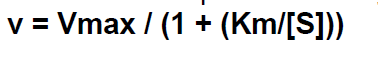
The Lineweaver-Burk double reciprocal plot
A rearrangement of the Michaelis-Menten equation that aims to obtain a linear relationship from the hyperbolic curve produced from the graph of rate of reaction and substrate concentration.

Aquaporins
Transport proteins embedded on the surface of the plasma membrane that regulate the flow of water in and out of cells.
Aquaglyceroporins are types of aquaporins that help transport glycerol, CO2, ammonia, and urea across the membrane.
Diseases associated with aquaporin dysfunction
Nephrogenic diabetes insipidus, where the kidneys fail to respond to antidiuretic hormone, causing water-diluted urine and extreme thirst, and neuromyelitis optica spectrum disorder (NMOSD), an autoimmune disease in which the immune system attacks the central nervous system, particularly the optic nerves and spinal cord.
Selective permeability of protein channels (in relation to sodium and potassium ions)
The protein channels have different properties in accordance with the molecule they transport. These properties include hydrophobic and hydrophilic properties, and specific chemical bonds that shape the behavior of the channel protein. For instance, the sodium ion channel is around 0.3 by 0.5 nanometers in diameter and is negatively charged. This attracts the positively charged dehydrated sodium ions to the channel. They are dehydrated because the attractive force pulls sodium away from the hydrating water molecules.
In contrast, the potassium protein channel (approx. 0.3 by 0.3 nm in diameter) lacks any significant charges and possesses different chemical bonds; therefore, no strong attractive force pulls potassium ions in. This is mainly because the hydration form of potassium is much smaller than that of sodium, so it takes less “motivation” to pull potassium into the protein channel.
The differences described above are what make channels selectively permeable to specific ions and molecules.
Two major classes of sodium channels in mammals
1) Voltage-gated sodium channels (VGSC)
Present around the entire body in various cell types → propagate action potentials.
Open when the membrane potential in their vicinity becomes depolarized1, allowing for the flow of sodium from areas of high concentration (usually the exterior of the cell) to areas of low concentration (usually the interior of the cell)
2) Epithelial sodium channel (ESC)
Present mainly in the skin and kidneys.
1 → Depolarization occurs when the cell’s resting potential becomes less negative, moves towards zero, or becomes positive. Depolarization is really important in excitable cells like neurons and muscle cells, where this electrical signal can be used to induce action potentials that cause the transport of neurotransmitters from neuron to neuron, allowing us to experience certain emotions.
2 Voltage-gated sodium channel gates
1) Activating (voltage-dependent) gate → opening allows for the influx of sodium and cell depolarization.
2) Inactivating (time-dependent) gate → stops the flow of sodium regardless of the activating gate or persistent sodium ions.
After the inactivating gate closes (slower than the opening of the activating gate, btw) and sodium ions stop pouring in, repolarization, where the cell goes back to the resting membrane state, occurs.
Two major classes of potassium channels
1) Voltage-gated → closed when the cell's resting potential is -70 mV and becomes excited during depolarization (less negative cell potential), causing them to open and allow the outward flow of potassium ions.
2) Non-gated → always open, so it allows for a constant flow of potassium across the cell, depending on concentration gradients.
Selectivity filter for potassium channel
This is what does not allow sodium ions to pass. There are 3 main reasons for this:
1) The sodium ion is too small to interact with the carbonyl oxygens of the selectivity filter and water.
2) The hydration energy of sodium is harder than that of potassium, meaning it is harder to dehydrate sodium ions, so they aren’t able to be fully dehydrated in the case that they do pass.
3) It is thermodynamically unfavorable.
The way potassium ions become dehydrated in the selectivity filter is by engaging in interactions between the OH groups of the hydration shell and the carbonyl oxygens of the filter.
Do note that sodium ions may still diffuse through potassium ions, but the channel prefers bigger ions that are capable of interacting with the carbonyl oxygens. So, sodium ions will face significant challenges, as the sodium ion is smaller and therefore cannot interact as much with the carbonyl oxygens, causing incomplete dehydration.
Ca2+ Channels
In skeletal strited muscle fibers and on the surface of the sarcoplasmic reticulum are the calcium channels. They are voltage-gated channels, meaning that they only open during excitation (depolarization). When the gate opens, this allows for the passive diffusion of calcium ions from the sarcoplasmic cysterns in the sacroplasma.
Exogamic carriers
Non-ribosomal translated proteins that are not natural components of the membrane. Instead they are found in certain antibiotics and bacteria that allow for the absorption of ions. They are divided into 2 groups:
Mobile carriers → diffuse into the membrane together with ions.
FIxed carriers → (pores, channels) ions diffuse through them and they are fixed in place.
Valinomycin
A type of mobile carrier that facilitates the transport of potassium ions by binding to them and forming a valinomycin-K+ complex that provides hydrophobicity to cross freely across the lipid bilayer of bacteria unimpeded. If the side where the valinomycin-K+ has low enough potassium levels, then the complex dissociates, releasing potassium ions. This process dissipates the bacterium’s transmembrane potassium gradient, leading to the loss of its membrane potential and ultimately, bacterial death.
It is a macrocyclic molecule formed by 12 alternating amino acids and esters that are synthesized by specific Streptomyces. It is a macrocyclic molecule composed of 12 alternating amino acids and esters, synthesized by specific Streptomyces species.
It is called an “ionophore” because it helps transport ions.
Valinomycin exhibits a 10,000-fold selectivity for K+ over Na+.
Valinomycin is a potent antibiotic.
It is commonly used as an agent to induce apoptosis (programmed cell death).
Gramicidin A
A fixed exogamic carrier that embeds itself into the cellular membrane of bacteria, allowing for the flow of monovalent ions (like Na+, K+, and H+) but not divalent ions (like Ca2+). It is a linear polypeptide that contains 15 consecutive D and L amino acid residues. Specifically, gramicidin facilitates the transport of potassium ions outside of the bacterial membrane, disrupting the electrochemical gradient and inducing apoptosis of the bacteria. It is for this reason that bacterial gramicidin A channels are called “killer” channels.
It is important to note that these amino acids are hydrophobic.
In soluted conditions, gramicidin A might allow 100,000,000 ions to pass.
Transport of ions across the bilayer is faster than that of the mobile exogamic carriers.