SOILS 101 Exam 3 Study Aid
1/122
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
123 Terms
chemical equivalency
-1 mole of negative charge attracts 1 mole of positive charge, regardless of valence
-However, only 1/2 of the atomic weights of divalent cations and 1/3 of the weight of trivalent cations are needed to provide 1 mole of charge
Equivalent weight
The weight needed to provide 1 mole of equivalent charge
Determined by dividing atomic weight by valence
Cation Exchange Capacity (CEC)
The sum of the total exchangeable cations that a soil can adsorb
-Expressed as cmolc/kg of soil
-a property of the soil solids
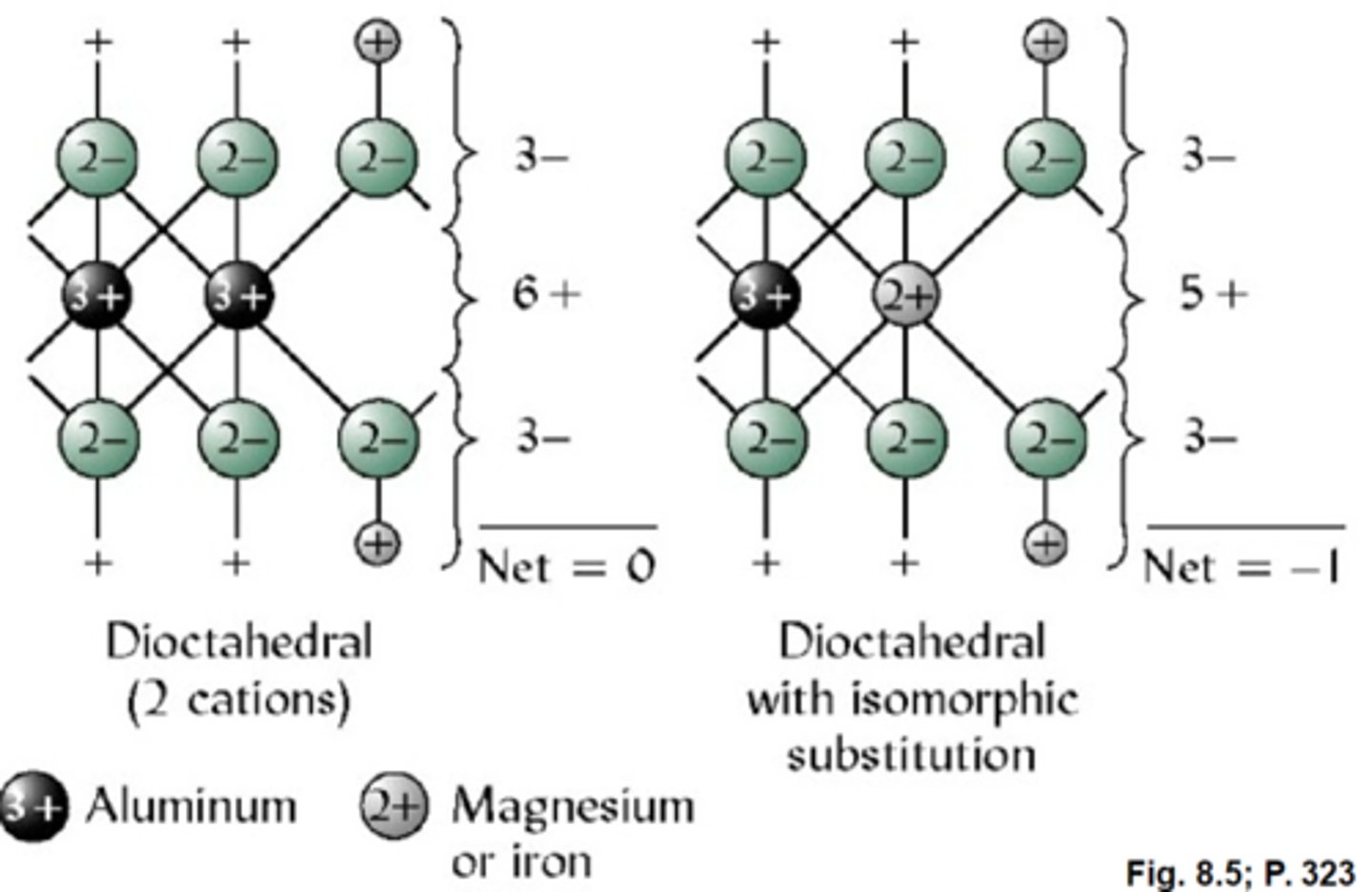
Permanent charge
Charge on colloids arising from isomorphic substitution in 2:1 or 2:1:1 clays.
-occurs on micas, vermiculites, smectites, and chlorites
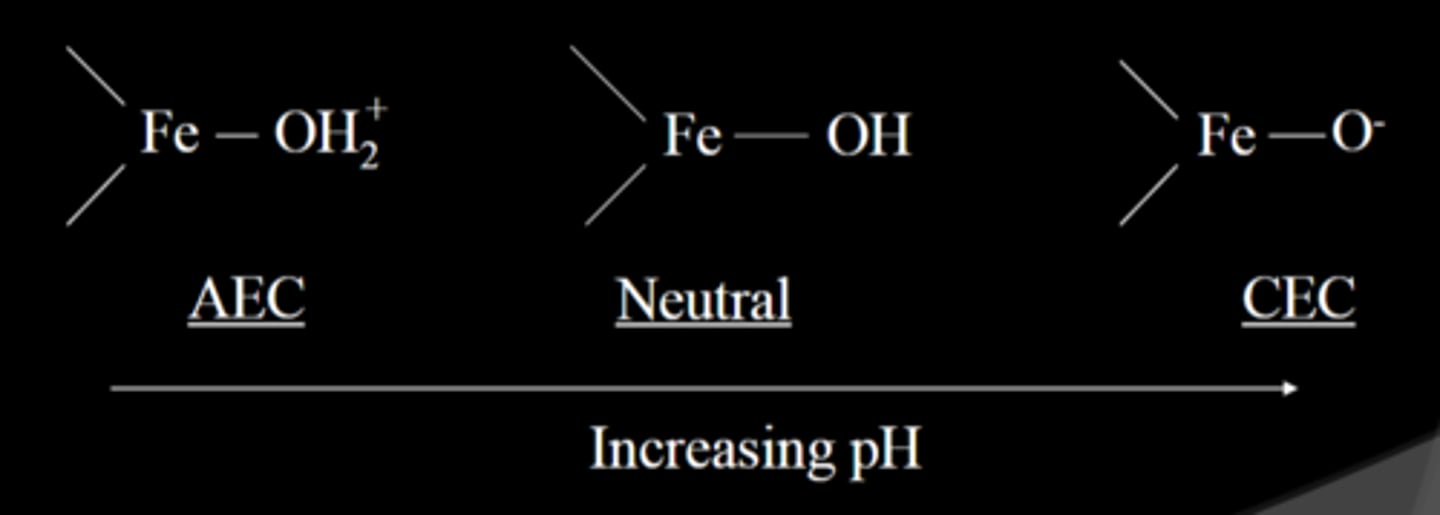
pH dependent charge
Charge on colloids that arises from the protonation or deprotonation of the functional groups on colloid surfaces.
-associated H+ (low pH) neutralizes charge, while lack of H+ (high pH) gives negative charge
-occurs on Fe/Al oxides, humus, allophane, and the edges of 1:1 clays like kaolinite
increases
Higher pH _______ the CEC (creates more negative charge)
broken edges
Where does pH dependent charge originate on 1:1 clays like kaolinite?
adsorption
The ability of an object to attract and hold particles on its surface
2-3
Soil organic carbon consists of ___ times as much carbon as is stored in vegetation.
Soil organic carbon
Comprises 1/2 of the mass of soil organic matter
carbonates
Soil carbon is stored as soil organic matter and inorganic soil ___________
Histosols, Inceptisols, and Gelisols
The 3 soil orders that contain 45% of all the soil organic carbon in the world
biomass
The component of SOM consisting of living organisms
detritus
The component of SOM consisting of identifiable dead tissue
humus
The component of SOM consisting of nonliving nontissue: humic substances and nonhumic substances
-The stable fraction of SOM remaining after plant and animal residues have decomposed.
-dark color, heterogenous, colloidal
-modified lignin and synthesized organic compounds
-stable and resistant
-can bind strongly to clay particles
-not well-defined
-accounts for 50-90% of the CEC for most soils
-chelation of metals (combines with the metal in the soil)
Active Pool
Pool of carbon consisting of biomass, particle OM and nonhumic substances
-labile (very active)
-broken down rapidly (days to years)
-food for soil organisms
-mineralizable N
-easy gains/losses
Slow Pool
Pool of carbon consisting of fine particle OM and lignin
-difficult to break down - takes decades
-some N and other nutrients available
Passive Pool
Pool of carbon consisting of mostly humus
-very stable - breaks down over 100s to 1000s of years
-slow losses/gains
-large CEC contribution
-increases water holding capacity
decomposition
The process of breaking down organic matter
-N, P, S and other plant essential elements are also released
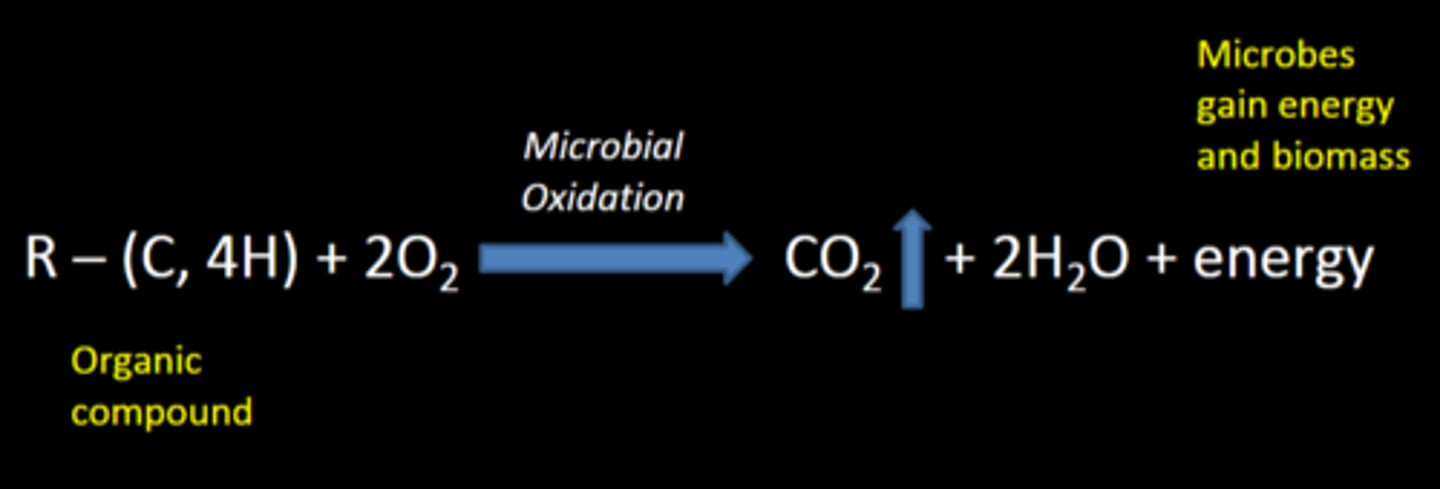
CO2
During decomposition, 60-80% of carbon is converted to ___
biomass
During decomposition, 3-8% of carbon is converted to ______
humus
During decomposition, 15-30% of carbon is converted to ______
environmental conditions and quality as a food source
Decomposition rates depend on?
plant residues
The primary source of soil organic matter
faster
Sugars, starches, and proteins decompose ______ than lignins and phenolic compounds
lignin
The substance in plants that makes cell walls rigid
-very resistant to decomposition
-only decomposed by specialized microbes (such as white rot fungi)
-Fuzzy leaves contain less than waxy leaves and decompose faster
24
Microbes need __ grams of N for every 1 gram of food
high
If the C/N ratio is ____ (above 25:1), nitrogen will be used up by microbes as they consume carbon, and will therefore be less available to plants
slower
Wet, low oxygen soils will have _______ decomposition
compost
Humus-like organic residues that have been piled, moistened, and allowed to undergo aerobic decomposition
-acts as a slow-release fertilizer
Entisols
-soils of very recent origins
-usually the only genetic horizon is an A horizon
-all soils that do not fit in the other 11 orders are included
-characterized by great diversity in environmental setting and land use
-many found in steep, rocky settings
-those found in fluvial and alluvial material can be fertile and productive
Andisols
-Soils formed in volcanic ash or other volcanic ejecta
-Dominated by glassy and poorly crystalline colloidal material
-often high organic matter
-high water holding capacity
-naturally fertile
-capacity to fix large quantities of phosphorous
-least extensive soil order worldwide
-cover significant forest land in Pacific Northwest
pH and Base Saturation
What 2 factors is soil acidity described by?
-relates to the production of acid cations and the leaching of base cations
pH
The negative logarithm of the hydrogen ion concentration
-forms a scale ranging from 1 to 14
-lower values indicate acid conditions, while higher values indicate alkaline conditions
3.5-8.5
What is the typical pH range of soils?
more
Humid region soils tend to be ____ acidic than dry region ones
-due to rainfall leaching away base cations
H+ and Al3+
What are the two acid cations?
Percent base saturation
The percent of the total CEC that is occupied by the base cations Ca2+, Mg2+, K+ and Na+
-as it increases, so does pH
-a value of 100% indicates neutral or alkaline pH
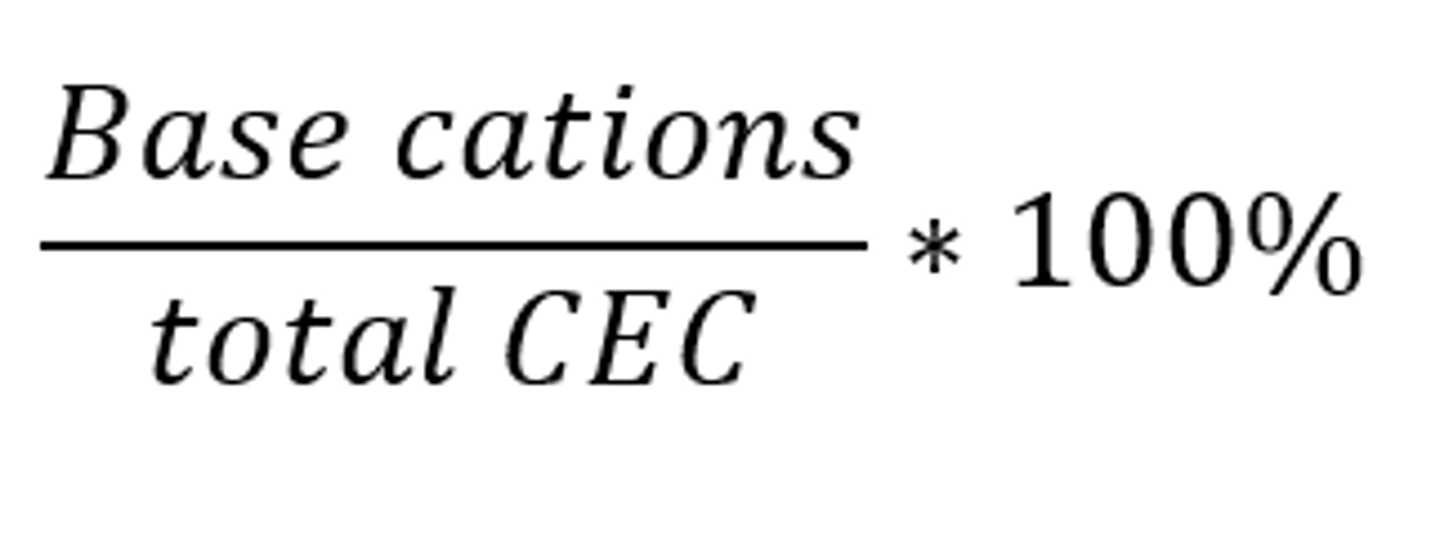
Active acidity
Acidity pool consisting of H+ or Al3+ cations in the soil solution
Exchangable acidity
Acidity pool consisting of H+ or Al3+ cations held near clay and humus surfaces
Residual acidity
Acidity pool consisting of H+ or Al3+ cations bound to clay and humus surfaces
Buffering Capacity
The ability of a material or solution to resist changes to pH
-active acidity can be easily changed, but the change is buffered by exchange and residual acidity
Al compounds
What is soil pH buffered by at low pH?
CEC and pH dependent charge
What is soil pH buffered by at neutral pH?
Carbonate precipitates
What is soil pH buffered by at high pH?
Al toxicity
Generally the most significant problem for plant growth in acid soils
-At pH of 5 and below, Al3+ is in solution, inhibiting root formation and microbial processes
-Al3+ also binds phosphorous, leading to plant deficiencies
bacteria
Are bacteria or fungi the dominant microbes in acid soils?
Organic matter, chemicals, fertilizers
3 types of additions that intensify soil acidity
-used in cases when growing acid-loving plants, reducing availability of toxic heavy metals, or controlling pathogens
Liming
The process of adding calcium carbonate or a similar substance to the soil in order to decrease acidity
-must be added repeatedly over the years as the cations are leached away or taken up by plants and the soil returns to its low pH
Calcium Carbonate Equivalent (CCE)
The neutralizing value of a liming material compared to pure calcium carbonate
Finer
_____ liming particles result in faster reaction time, due to their greater surface area
increases
When lime is added, the base saturation ________
increases
When lime is added, the pH ________
cyclic process
Liming is a _______ __________ (has to be repeated over the years after the soil pH returns to acidic)
nutrient deficiencies
What is a problem that happens with overliming (raising the pH too high)?
5.5-6.5
Ideal pH range for most crops.
Half
More than ____ of Earth's arable land is salt-affected.
-precipitation in these areas is insufficient to leach base cations and soluble salts
Calcareous soil
Soil containing sufficient calcium carbonate, often with magnesium carbonate, to effervesce visibly when treated with weak hydrochloric acid
-semi-arid and arid region soils containing free lime (undissolved CaCO3)
-high soil pH between 7 and 8.4
-P deficiencies (Ca and Mg bind it)
-Micronutrient deficiencies: B, Zn, Fe, Mn, Cu
-Mo toxicity
Sodic soils
Soils where plant growth is constrained by high levels of Na+, OH-, and HCO3-, and poor soil structure
-Na+ causes soil to be dispersed due to its large hydrated radius
-have serious degradation and loss of microporosity
-can cause soil to become puddled due to poor infiltration
Gypsum and water
What 2 things can be applied to a sodic soil for reclamation?
-allows for salt to be leached from the soil
Nitrogen
Macronutrient that is a major part of proteins, chlorophyll, and nucleic acids
-generally most abundant macronutrient in plant tissue
-drives leaf and height growth in plants
Lodging
Effect of oversupplying nitrogen to plants
-common in small grains (wheat, etc.)
Nitrogen Deficiency
Deficiency that is characterized by chlorosis (yellowing of leaves)
-older leaves yellow first
-stunted height, thin, spindly, low shoot:root ratio, faster maturation
Fixation
Step in the nitrogen cycle where atmospheric N2 is converted to elemental or gaseous forms
Legumes
Plants of the bean and pea family, whose roots have nodules for N fixation via N-fixing symbiotic bacteria
Haber-Bosch process
The process used to synthesize N fertilizer on an industrial scale.
-uses large energy inputs
-increased world N fertilizer production enormously
Mineralization
The conversion of organically-bound N to inorganic forms of N such as ammonium and nitrate (which plants can take up)
NH3 Volatilization
The loss of ammonia from the soil as gas
-occurs with pHs above 7 or temperatures above 16 C
Nitrification
The conversion of ammonium to nitrate (which is preferred by plants)
-first step increases soil acidity with the production of nitrite, second step creates nitrate
-performed entirely by autotrophic bacteria
-must be preceded by ammonification of NH4+ from OM
-Bacteria need oxygen, moisture, high Ca and pH
Immobilization
The opposite of mineralization; nitrate is made unavailable by becoming organically-bound.
Leaching
The loss of N by being washed from the soil.
-nitrate is an anion and is repelled by negative soil surfaces, so if supply exceeds plant uptake, it can be washed away
-nitrate = most common contaminant of US groundwater
-can cause human health problems and the eutrophication of surface water
Denitrification
The conversion of nitrate to N2 gas
-nitrite --> nitric oxide --> nitrous oxide --> nitrogen gas
-occurs in wet, oxygen-deficient environments
-microbially driven process
Organic forms
90-95% of total soil N is in __________ ______ (as humus or microbial tissue)
Sulfur
Macronutrient found in amino acids, enzymes, aromatic oils
-legumes, cabbage, onions require large amounts
-levels in plant tissues are 1/10th that of plant tissue N
Sulfur deficiency
Deficiency characterized by spindly plants with light green-to-yellow leaves - BUT occurs on young leaves first and may be striped.
Phosphorus
Macronutrient present in low quantities in soil
-less than 1/10th of the levels of total N
-is very insoluble
-strongly sorbed by mineral surfaces in soil
-also quickly taken up by microbial biomass
-Important for fruiting stages of plants
Phosphorus deficiency
Deficiency characterized by plants having stunted growth and dark green/purpling of older leaves
mycorrhizal fungi
Type of fungi that are found in association with plant roots and improve phosphorus availability to plants
Erosion
What is the major loss pathway of environmental concern for Phosphorus?
-As contrasted to N, which leaches from the soil into water bodies, P that is adsorbed to soil or OM particles is lost
Potassium
Macronutrient that weathers from mineral sources
-not dependent on microbial processes like N, S, or P
-low environmental concerns
-important for cow health
-important for health, resistance to disease, and winter hardiness of plants--needed for plump seeds and stiff stalks
Potassium deficiency
Deficiency characterized by older plant leaves having yellow and burnt-looking edges.
NPK fertilizer
Fertilizer containing nitrogen, phosphorus, and potassium
-may be described with 3 numbers that represent the percent of each nutrient present (e.g. a bag of 10-10-10 contains: 10% available N, 10% available P2O5, 10% available K2O)
Calcium
Macronutrient that is a major component of cell walls
-rarely see deficiencies - usually only in acidic soils (where Al toxicity also becomes an issue)
Calcium deficiency
Deficiency characterized by yellow leaves (except the veins remain green), failure to thrive
-Buds, unfolding leaves, fruits, and root tips are most affected
-Accompanied by and similar to Al and Mn toxicities
-causes blossom-end rot on tomatoes
Magnesium
Macronutrient used for chlorophyll and photosynthesis
-Not commonly a deficiency
Magnesium deficiency
Deficiency characterized by:
-mottled green/yellow leaves in dicots
-striping in monocots
-interveinal chlorosis
Ca:Mg
What is the nutrient ratio that plants need to be between 1:1 to 1:15?
Micronutrients
Nutrients such as Mo, Cu, Zn, Mn, Fe, B, Cl
-essential for plant growth
-high levels can be very toxic
-irrigation waters should be monitored for toxic levels
-relatively immobile in plants, so deficiency signs are seen in younger parts of plant
-more soluble at low pH, less soluble at high pH
Chelation
Occurs when organic compounds have 2 or more atoms capable of bonding cationic micronutrients, increasing their bioavailability
-from Greek word "to claw"
-common in microbe and plant root exudites
-increases the availability of micronutrients
Limiting Factor
The concept that overall plant growth in a soil system will be limited by the factor (nutrient) that is present in the lowest relative amount, regardless of whether or not all other factors are sufficient
-determined using soil fertility testing
Nitrogen
Which macronutrient is not typically tested for in soil fertility testing?
-its levels change too much and it moves through the soil too fast for tests to be useful for farmers
15-20
How many subsamples should be included in each soil sample for soil fertility testing?
-must be mixed to form a composite sample in a non-metal container, then air-dried and bagged.
Yes
Should you always send soil samples to a testing lab that is within your general area of the US? (Yes/No)
Spodosol
Soil Order:
-acidic soils
-low fertility
-wet and sandy
-often found under coniferous forests in cooler temperatures
-diagnostic Spodic horizon: subsurface accumulation of organic matter, iron and aluminum
Ultisol
Soil Order:
-found in wet tropical and sub-tropical forests
-strongly acidic clays
-acid silicates and Fe and Al oxides
-not as weathered as Oxisols
taxonomic system
The 12 soil orders are a ___________ _______
-their organization allows us to study and understand soils and their development, helps us understand geographic distribution of soils, how they will be affected by changing climate, and allows communication between soil scientists
Oxygen
Gas that is present in the atmosphere as 21% but decreases with soil depth to 5%-0% in deep, poorly-drained horizons
-concentration decreases after heavy rainfall
-when concentrations are too low (<10%), respiration is impeded
Carbon Dioxide
Gas that is present in the atmosphere as 0.041% but increases 10-100x in the soil.
-May become toxic to some plants above 10%
water vapor
Gas that is present in soils in greater concentrations than in atmosphere (100% relative humidity)
Anaerobic
__________ conditions can lead to production of reduced nitrogen gases, hydrogen sulfide, methane, and ethylene