Electron configuration 2
1/7
Earn XP
Description and Tags
https://www.youtube.com/watch?v=LxtMp4v8FNQ
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
8 Terms
Why do we fill the 4s subshell before filling the 3d subshell
The energy of the 4s subshell is less than the energy of the 3d subshell
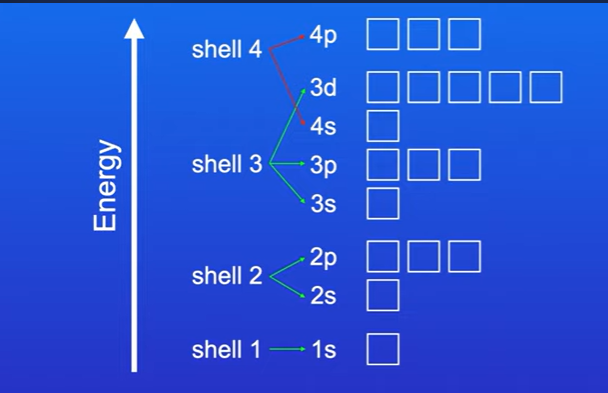
Work out the orbitals for the element iron , 26 electrons.
Electron configuration of iron is 1s2 2s2 2p6 3s2 3p6 3d6 4s2
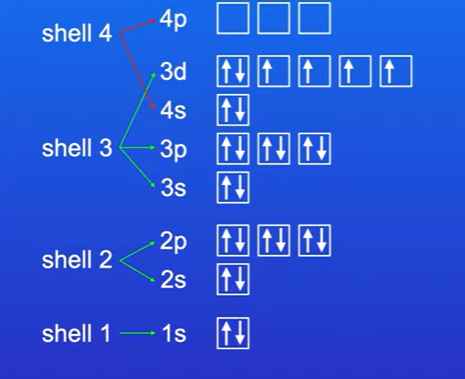
What is electron configuration written as
In the order of electron shells, not order of filling
Why are chromium and copper exceptions of the rules.
In both cases the 4s subshell contains only one electron even though there are 3 electrons in the 3d subshell
The 3d subshell is more stable when it is either half full or completely full. In case of chromium by having only one electron in 4s subshell, it can have half full 3d subshell. In case of copper, by only having one electron in the 4s subshell, it can have completely full 3d subshell
