Alkanes (4c)
1/9
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
10 Terms
Alkanes
saturated hydrocarbons: only singe C-C bonds, no double
general formula is CnH2n+2
unreactive generally
can undergo combustion, can be cracked into smaller molecules, can react with halogens in the presence of light
ex: Methane
Methane
CH4
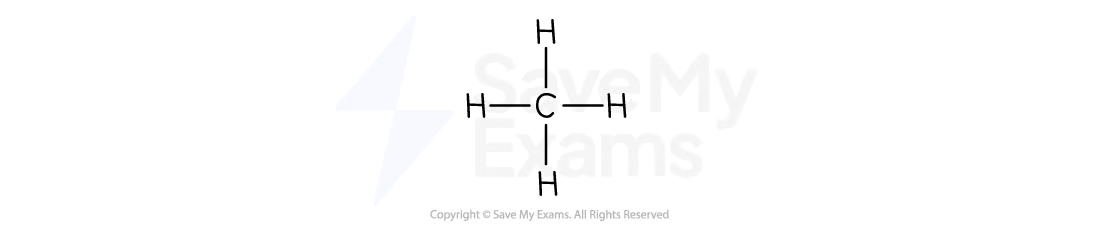
Ethane
C2H6
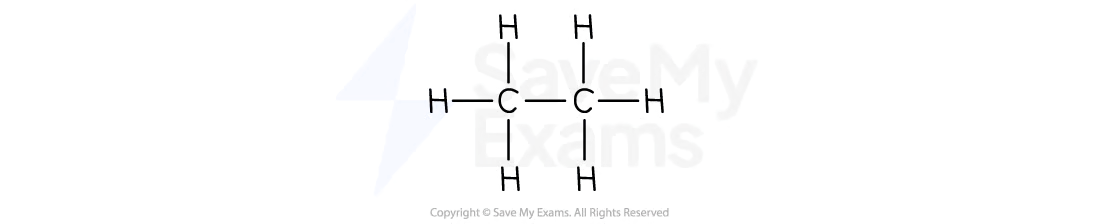
Propane
C3H8
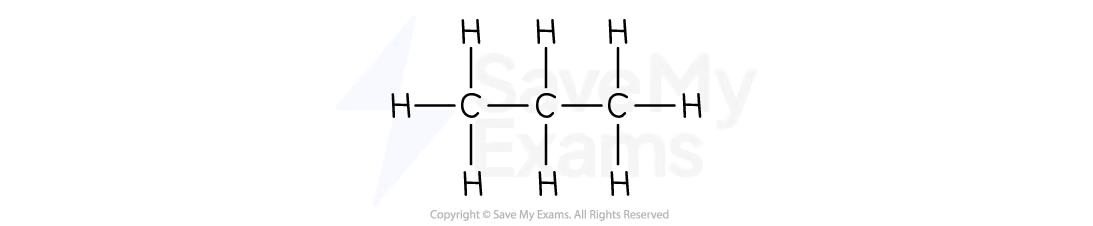
Butane
C4H10
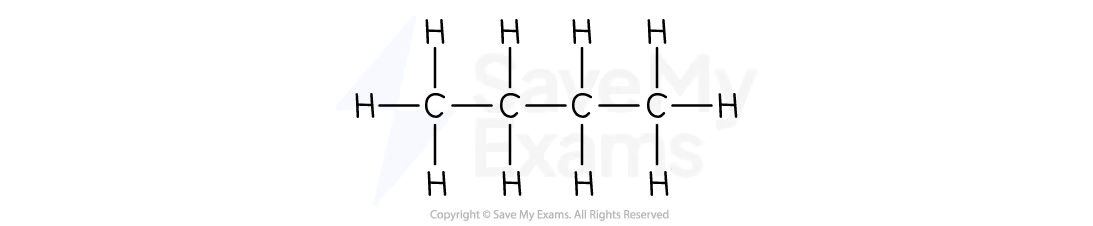
Pentane
C5H12
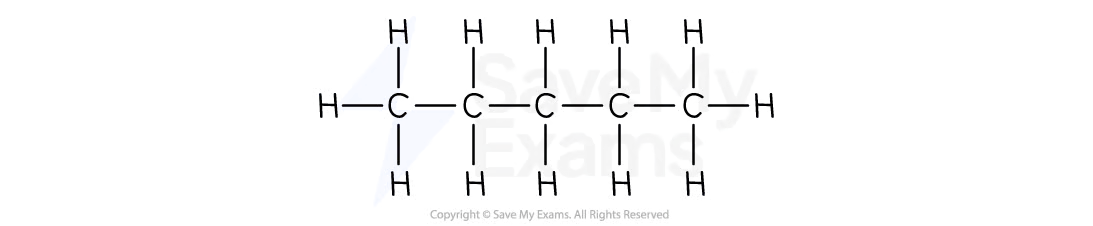
Alkanes’ Substitution Reaction
one atom is swapped with another
alkanes undergo substitution in the presence of UV radiation
Methane and Bromine reaction in UV Radiation
methane + bromine → bromomethane + hydrogen bromide
CH4 + Br2 → CH3Br + HBr
also reacts with Chlorine the same way
methane + chlorine → chloromethane + hydrogen chloride
CH4 + Cl2 → CH3Cl + HCl
Halogenalkanes
formed from halogen + alkane reactions in UV radiation
also called haloalkanes
used as solvents, refrigerants, propellants and pharmaceuticals
CFC
chlorofluorocarbon
important haloalkane
slowly phased out due to ozone damage
previously used in fridges and ACs