Biochem Chymotrypsin Mechanism (Final)
1/20
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
21 Terms
Step 1 of Chymotrypsin Mechanism
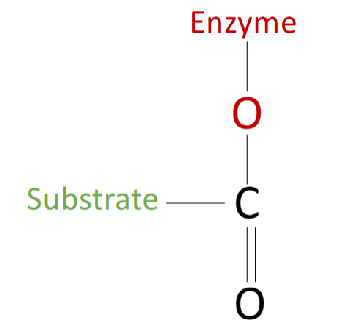
substrate binds to the enzyme
substrate has an aromatic side chain that rests in the specificity pocket, this places the bond to be cleaved near the catalytic serine, chymotrypsin cleaves the peptide bonds directly following (on the C-terminal side of) aromatic amino acids
Step 2 of Chymotrypsin Mechanism
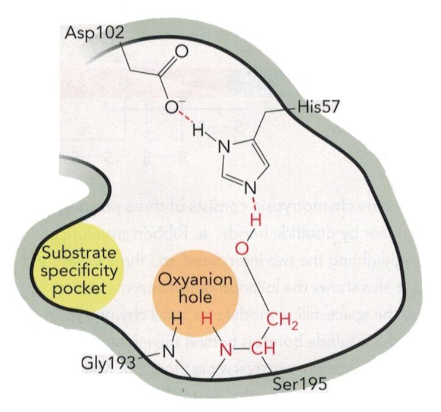
base catalysis by His 57 in the catalytic triad
deprotonates the catalytic serine (195) to create the strong nucleophile
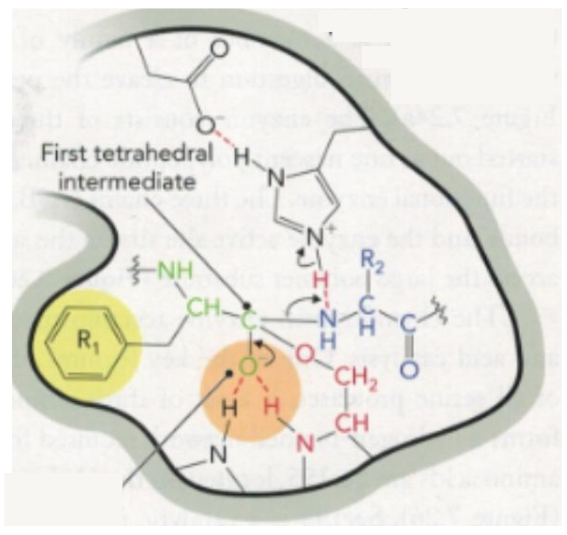
Step 3 of Chymotrypsin Mechanism
negatively charged oxygen of serine (nucleophile) attacks the C of the substrate peptide bond
nucleophilic attack step, electrons move to the oxygen of the substrate, creating a tetrahedral oxygen
Step 4 of Chymotrypsin Mechanism
oxyanion is stabilized by the partial positive charges in the oxyanion hole by interacting with the backbone amide hydrogens to form two H-bonds
this is a key step because it stabilizes the transition state
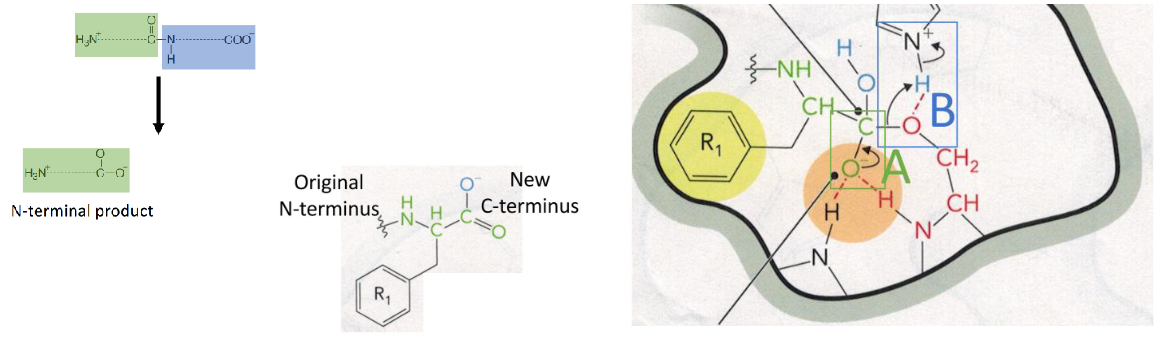
Step 5 of Chymotrypsin Mechanism
release of the first product- the C-terminal end of the original protein
proton from the His is transferred to the NH of the new product (new N-terminus), and the C-terminal half of the protein is released while the N-terminal half of the protein is still covalently attached to the enzyme
Step 6 of Chymotrypsin Mechanism
formation of second oxyanion intermediate
water enters the active site and is deprotonated by His to produce OH-, OH- attacks the C of the substrate, electrons move to the oxygen of the substrate which creates another oxyanion that is stabilized by the oxyanion hole
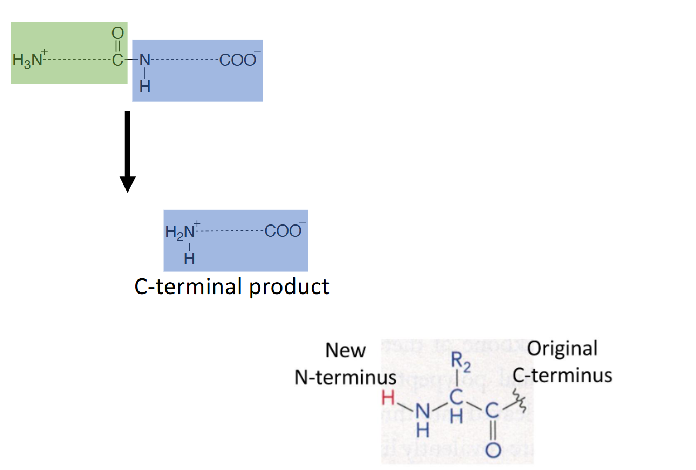
Step 7 of Chymotrypsin Mechanism
release of the second product- the N-terminal half of the substrate
enzyme is the leaving group in this case (ser detaches), serine is regenerated by transferring a proton from His
Step 8 of Chymotrypsin Mechanism
the catalytic triad is regenerated
active site is reset to catalyze again
Step 1 of Chymotrypsin Mechanism Diagram
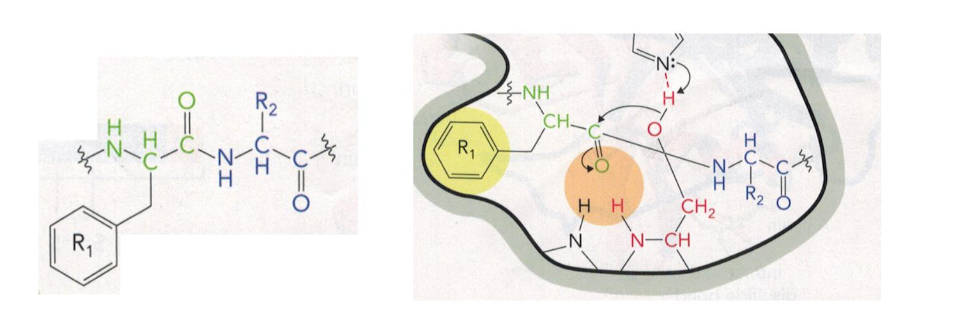
Chymotrypsin Diagram
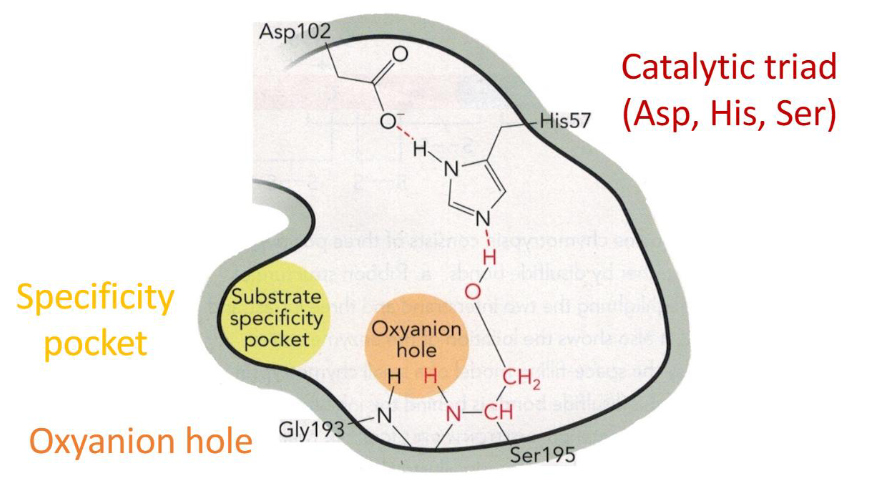
Step 2 of Chymotrypsin Mechanism Diagram
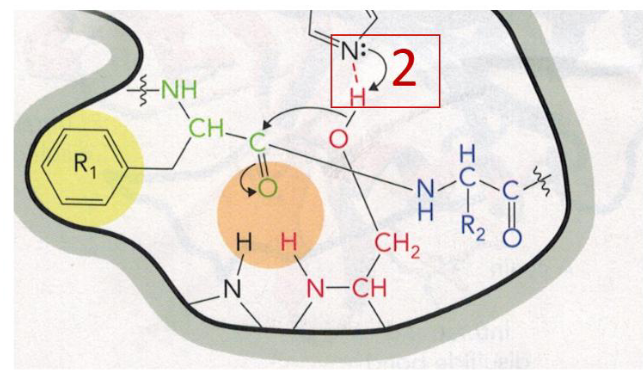
Step 3 of Chymotrypsin Mechanism Diagram
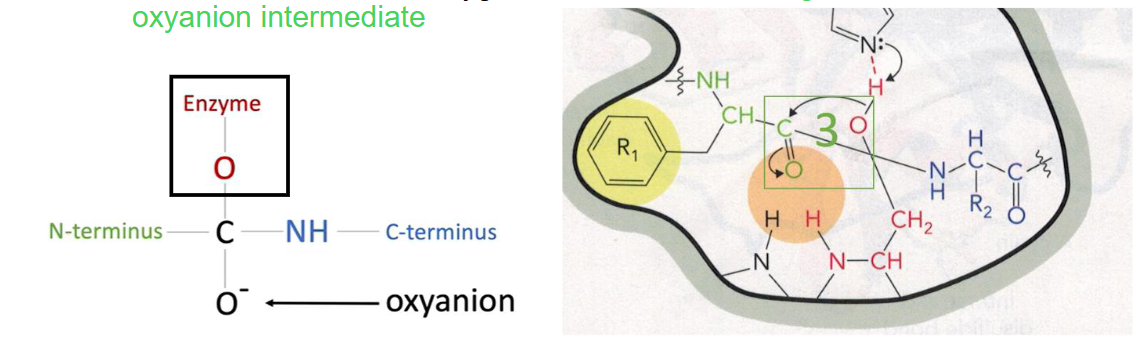
Step 4 of Chymotrypsin Mechanism Diagram
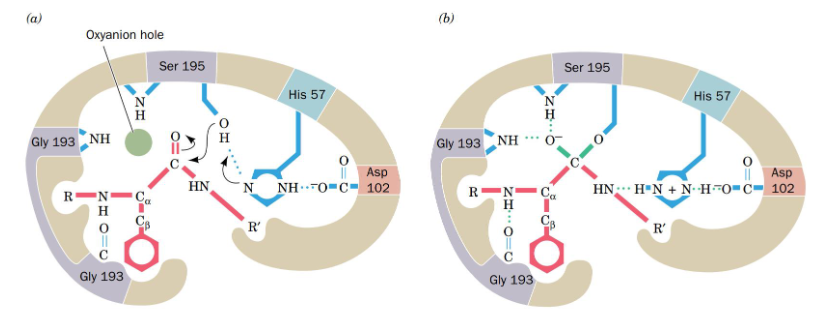
Step 5 of Chymotrypsin Mechanism Diagram
Step 6 of Chymotrypsin Mechanism Diagram
Step 7 of Chymotrypsin Mechanism Diagram
Step 8 of Chymotrypsin Mechanism Diagram
What is the first product released by chymotrypsin?
the c-terminal product (that has a new n-terminus)
First Chymotrypsin Product Diagram
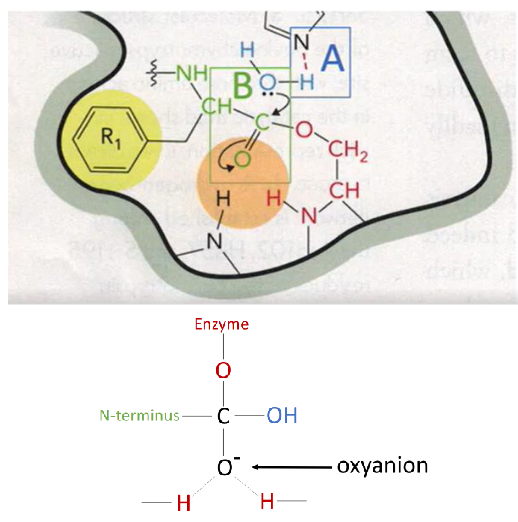
What is left on the enzyme after the first product (c-terminal end) is released?
acyl-enzyme intermediate, n-terminal half is still attached to the enzyme
N-Terminal Attachment to Enzyme After C-Terminal Release Diagram