BIOL-L 112 Chapter 3
1/57
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
58 Terms
Element
Pure substance made of only one type of atom, which cannot be broken down into simpler substance by ordinary chemical means
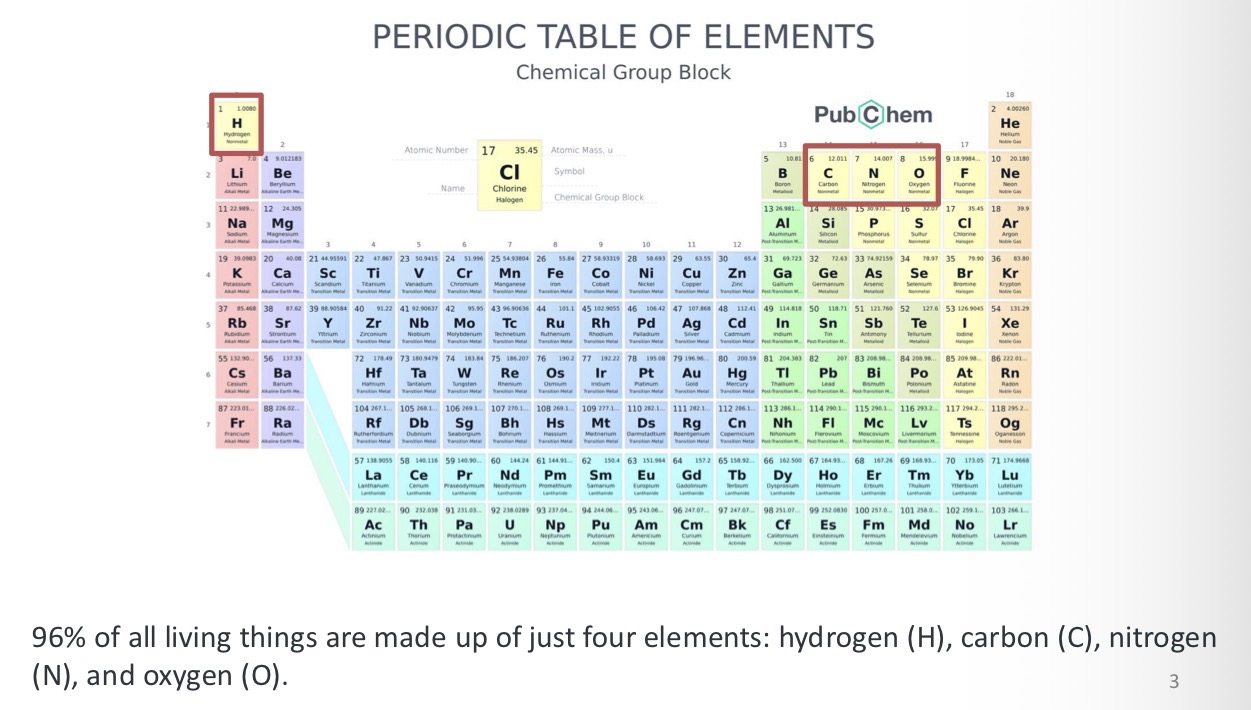
Atom
The fundamental building blocks of all matter, the smallest unit of an element that retains it chemical properties, consisting of a central nucleus of protons (positively charged) and neutrons (neutral) surrounded by a cloud of negatively charged electrons
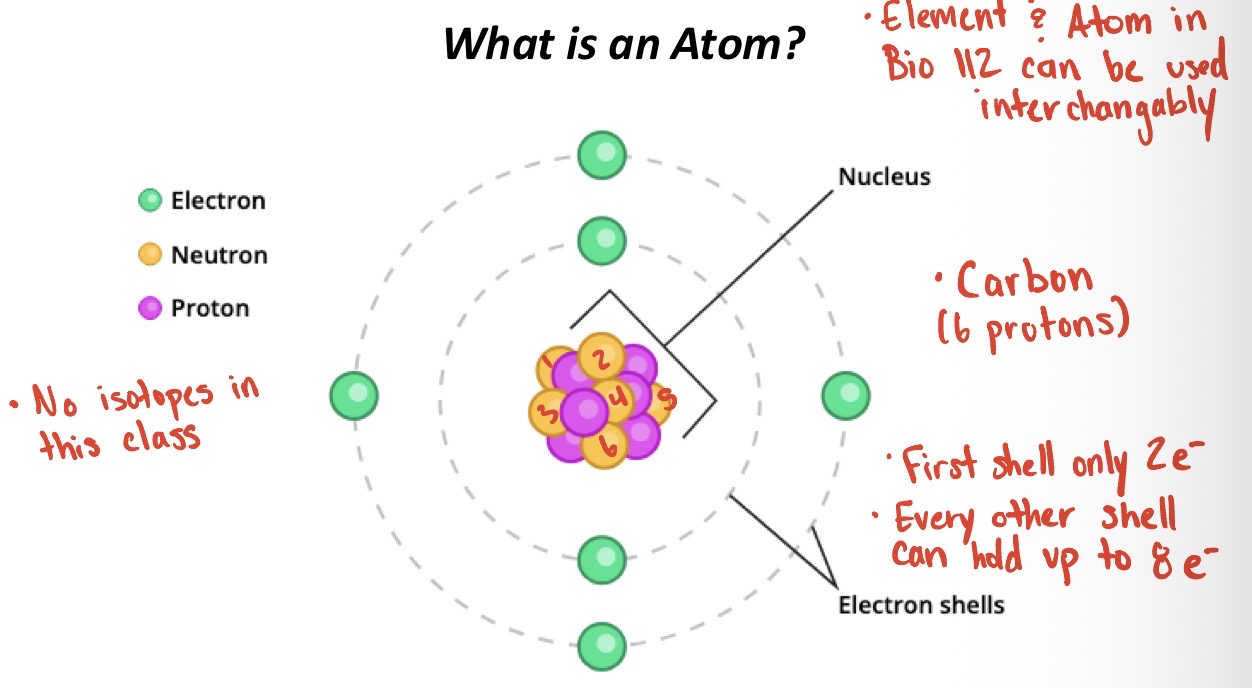
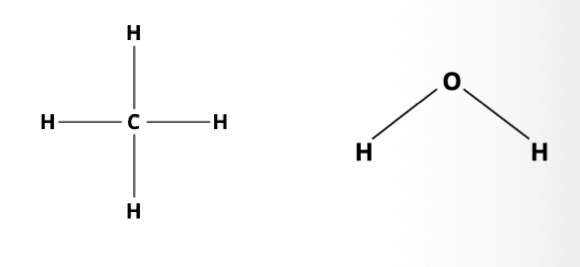
What is a molecule?
A molecule is a group of 2 or more atoms held together by chemical bonds, forming the smallest unit of a pure substance that still has the substance’s chemical properties
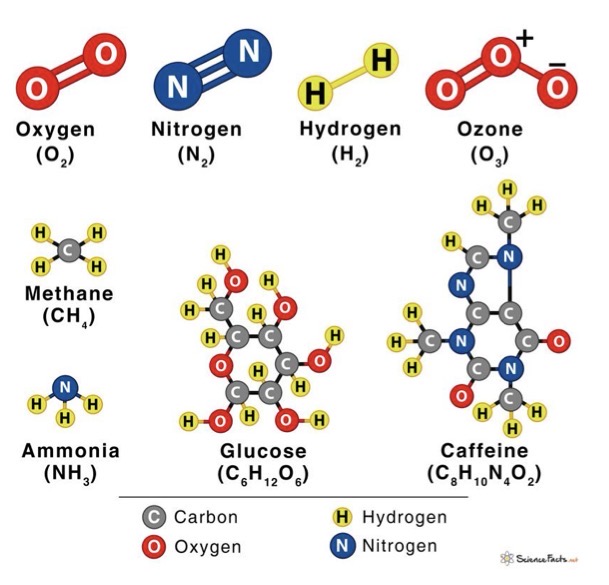
Molecular formulas
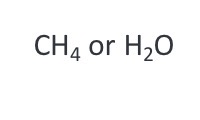
Structural formulas
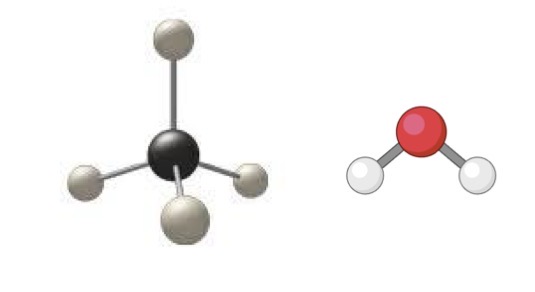
Ball and stick models
Space filling models
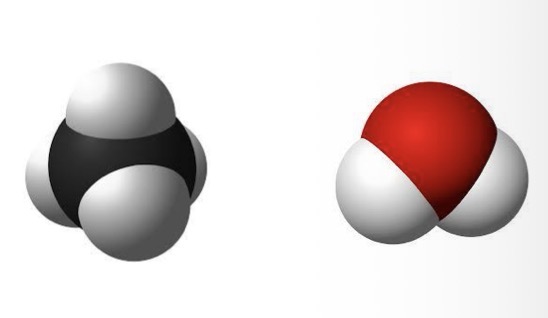
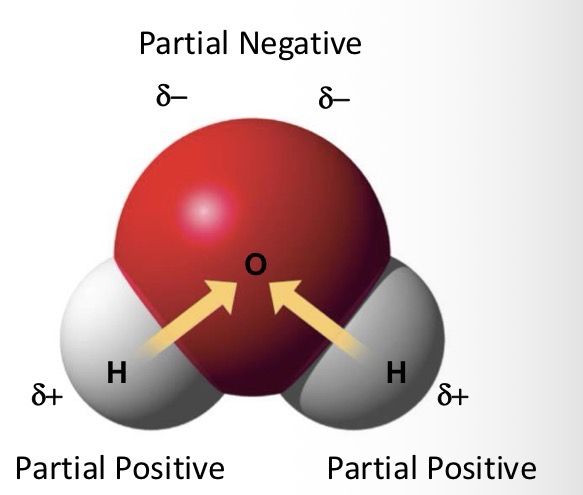
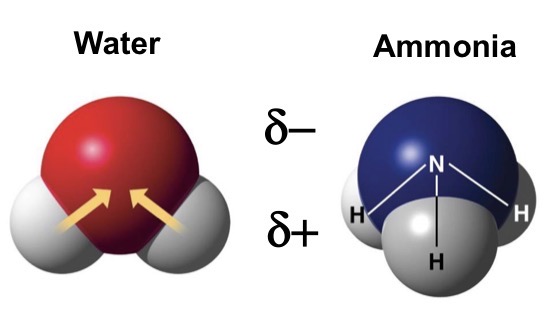
Is water a HIGHLY polar or non-polar molecule?
Polar
Electronegativity
A chemical property that describes the ability of an atom to attract electrons towards itself
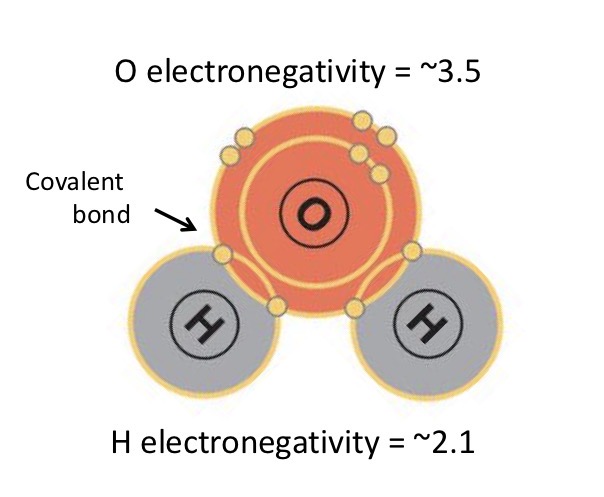
Which atom has more electronegativity and what does this mean?
Oxygen; shared electrons in covalent bond are pulled towards oxygen. This gives oxygen a partial negative charge, while H has a partial positive charge.
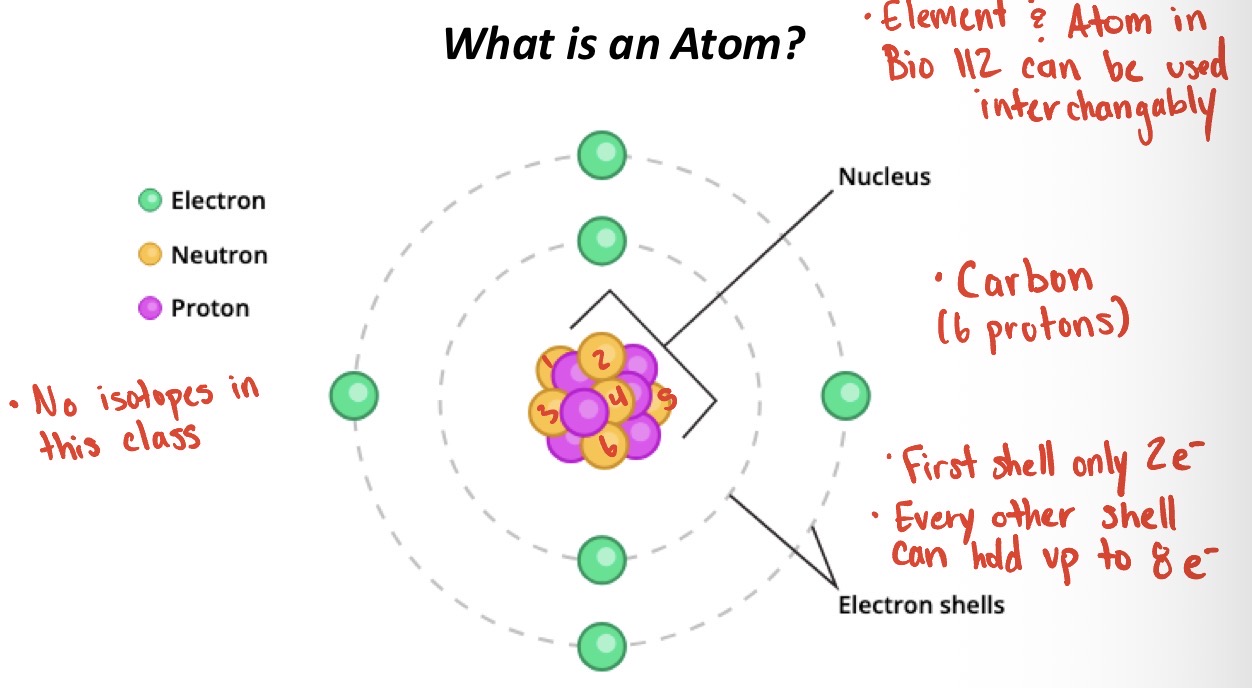
In an atom how many electrons are in each shell?
The first shell has only 2 electrons and every other shell can hold up to 8 electrons
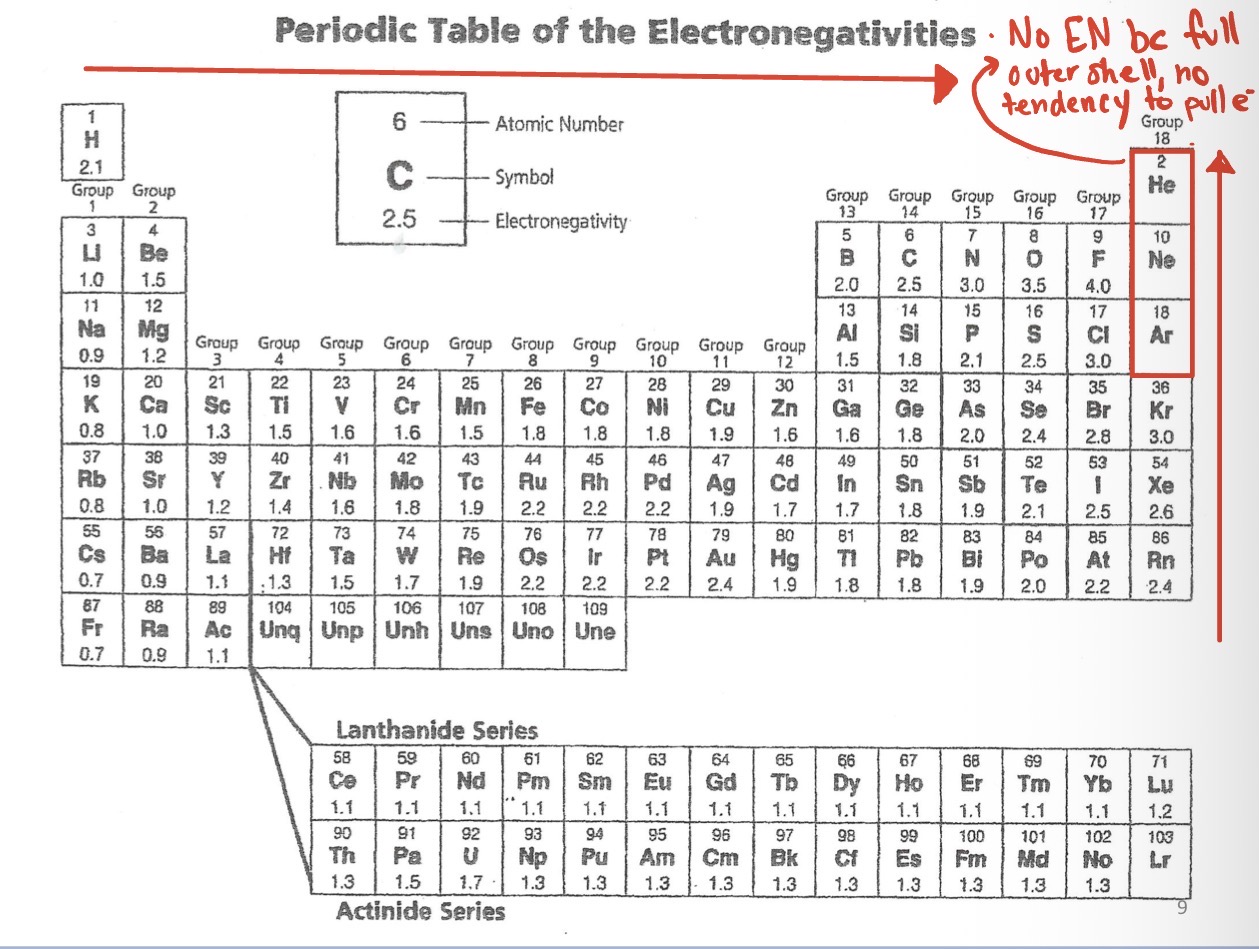
Why do Noble gases have no electronegativity?
Because their outer shell is already full, so it has no tendency to pull electrons.
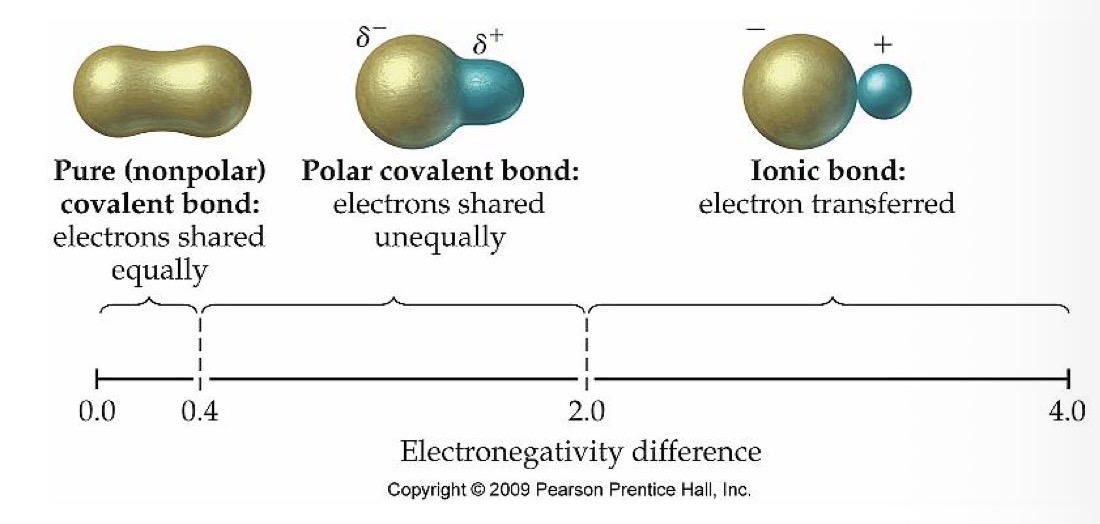
What type of bond do two atoms with an electronegativity of 0-0.4 have?
Nonpolar (pure) covalent bond
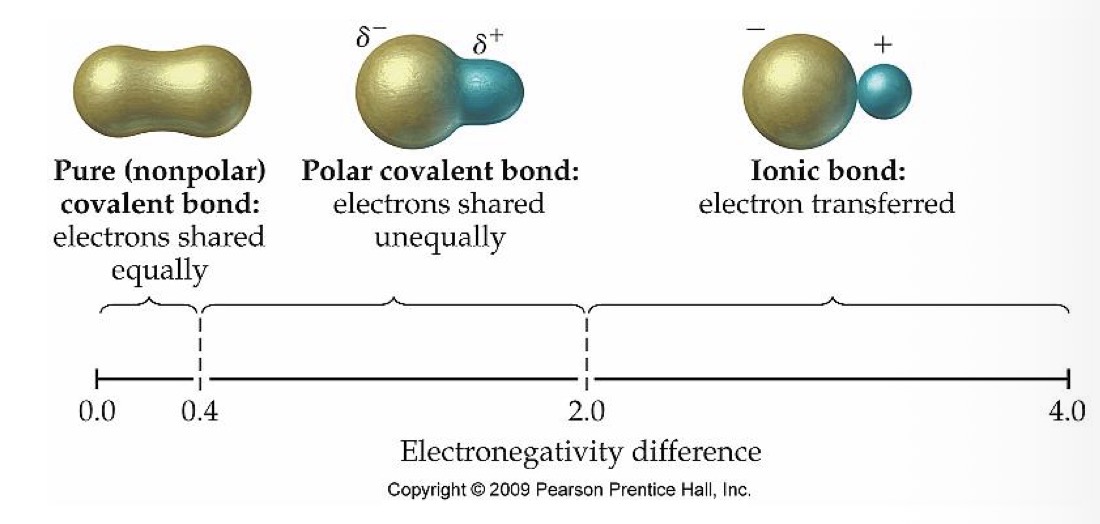
What type of bond do 2 atoms with an electronegativity of 0.4-2.0 have?
Polar covalent bond
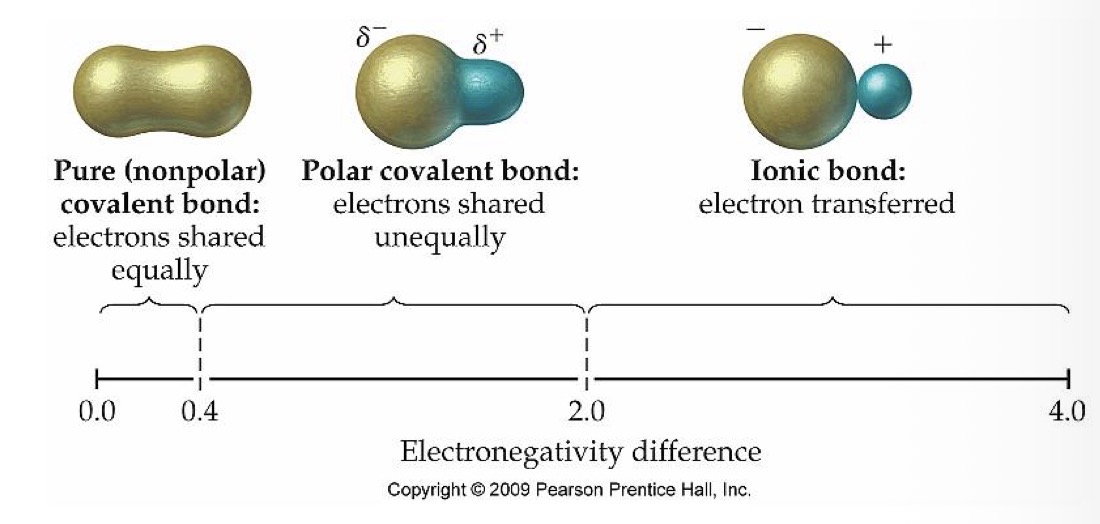
What is the relationship between the electrons in two atoms with a nonpolar (pure) covalent bond?
Electrons are shared equally
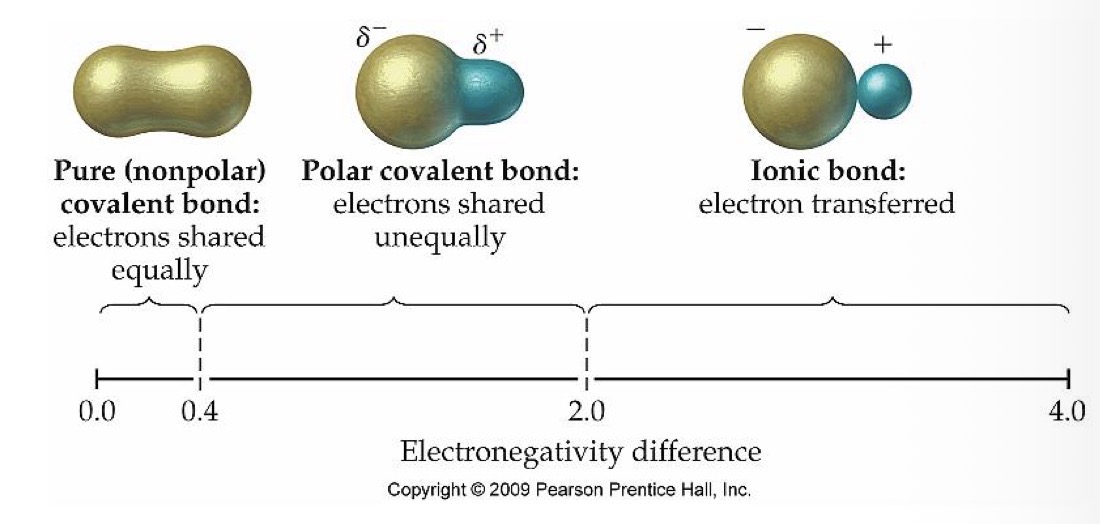
What is the relationship between the electrons in two atoms with a polar covalent bond?
Electrons are shared equally
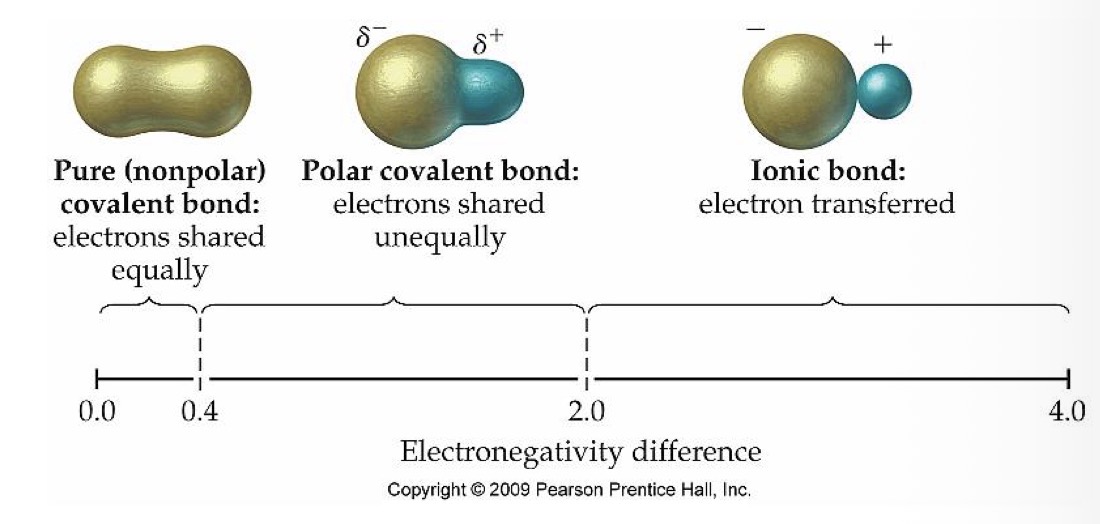
What is the relationship between the electrons in two atoms with an ionic covalent bond?
Electron transferred
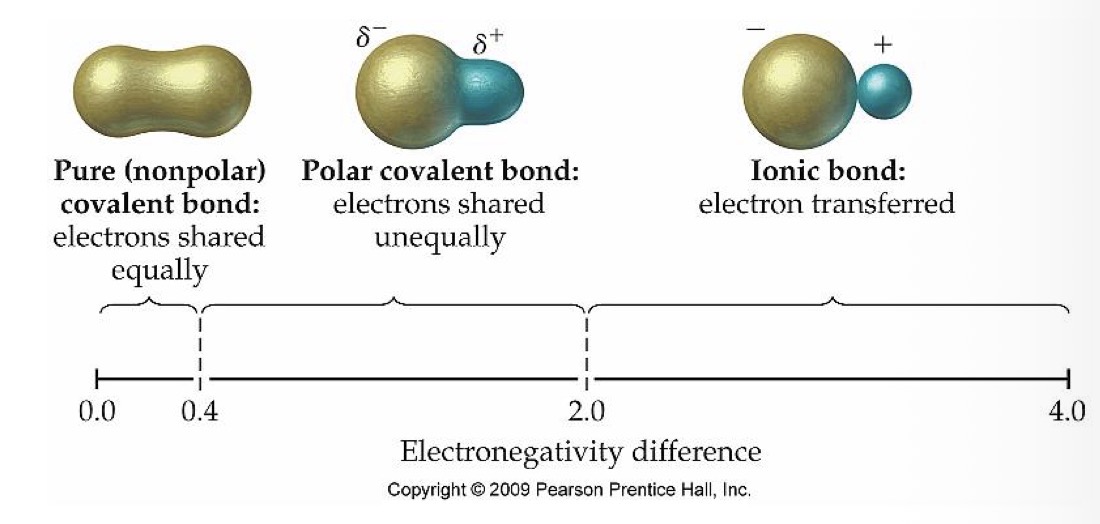
What type of bond do 2 atoms with an electronegativity of 2.0-4.0 have?
Ionic bond
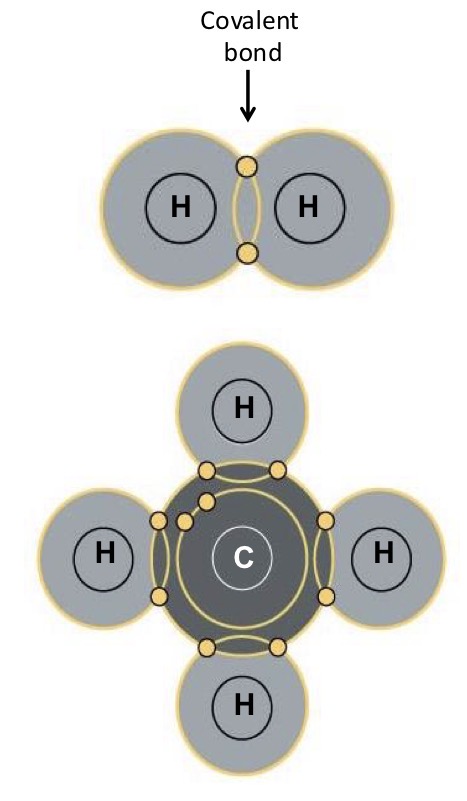
What kind of molecules are molecular hydrogen (H2) and methane (CH4)?
Hydrogen (H) has an electronegativity of ~2.1
Carbon (C) has an electronegativity of ~2.5
Nonpolar molecules. Electrons are shared relatively equally between the atoms.
Based on these characteristics is this a polar or nonpolar molecule?
unequal sharing of electrons
Frequently asymmetrical
Contain partial positive and partial negative charges
Polar Molecules
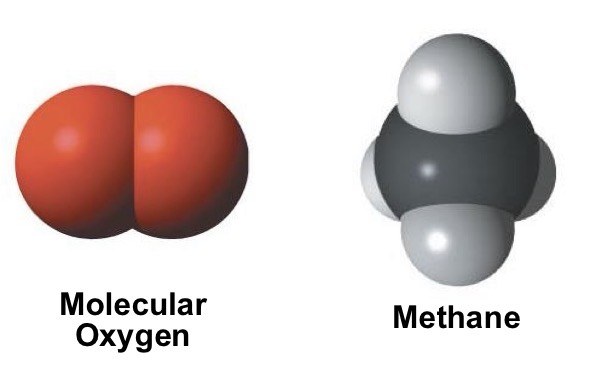
Based on these characteristics is this molecule polar or nonpolar?
equal sharing of electrons
Usually symmetrical
Have little or no partial charge
Nonpolar molecules
True or False: Many large biological molecules contain both polar and nonpolar components
True
What enables water to form hydrogen bonds?
Polarity. The partial positive charge of hydrogen atom in one polar molecule is attracted to the partial negative charged oxygen atom of another polar molecule to form hydrogen bond.
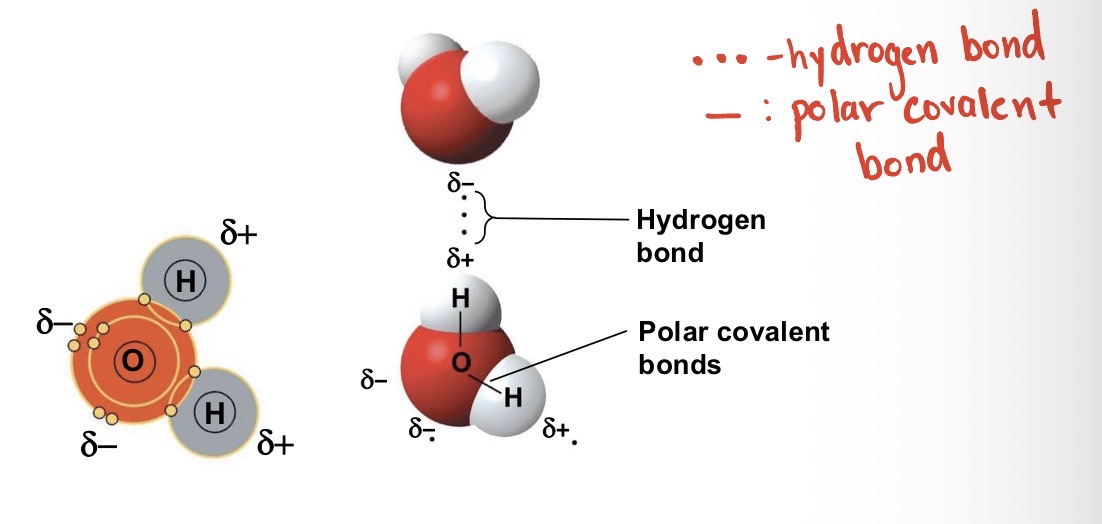
Polarity enables 1 water molecule to hydrogen bond with how many other water molecules?
4
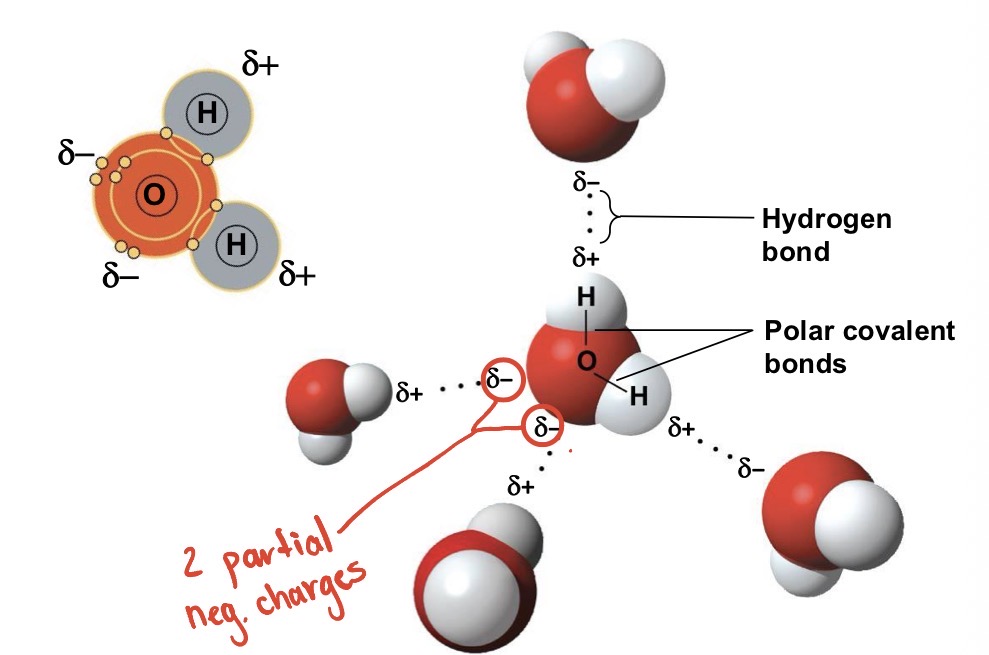
When in liquid form, is each hydrogen bond strong and long lived or fragile and short lived?
Fragile and short lived
Water’s unique ability to form hydrogen bonds lead to what 4 emergent properties of water? An why are these emergent properties important?
Cohesive behavior
Ability to moderate temperature
Expansion upon freezing
Versatility as a solvent
They help make Earth an environment that is suitable for life!
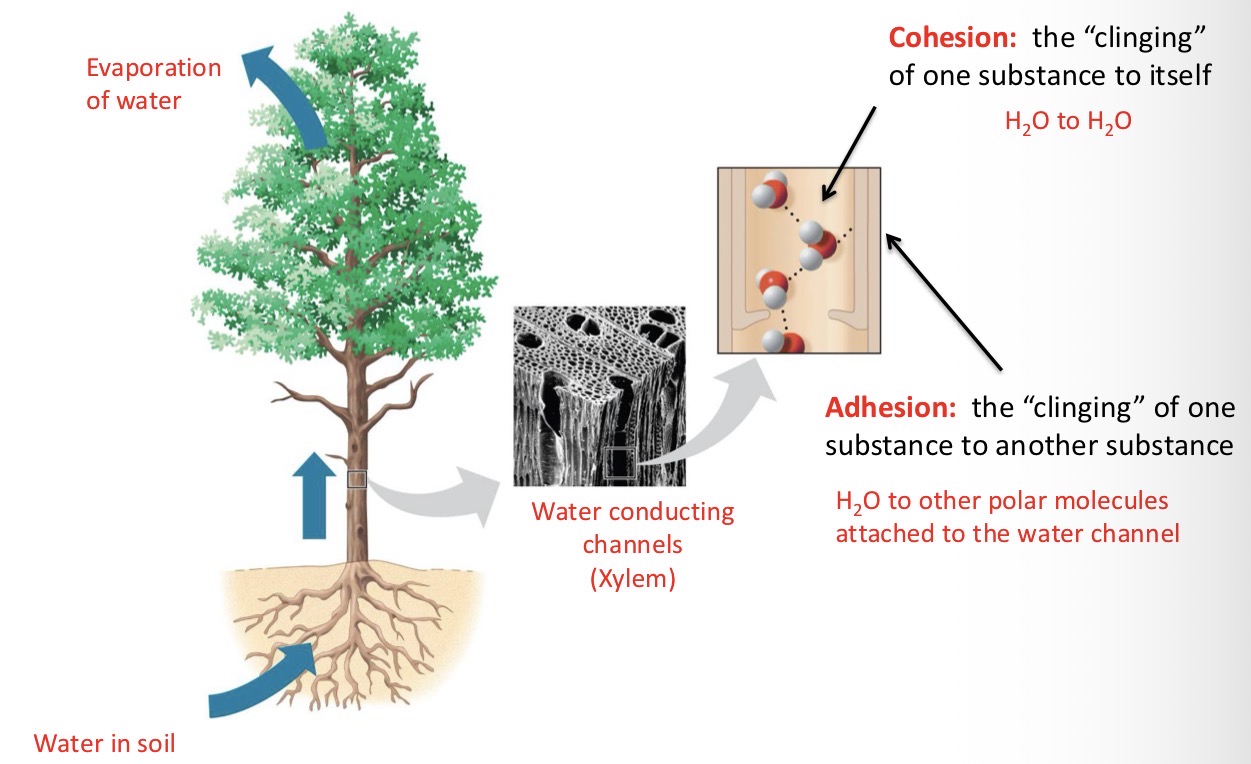
Explain Waters Cohesive Behavior
Hydrogen bonding holds water molecules together. Water’s cohesive and adhesive properties enable the transport of water and dissolved nutrients upwards in plants - against the force of gravity.
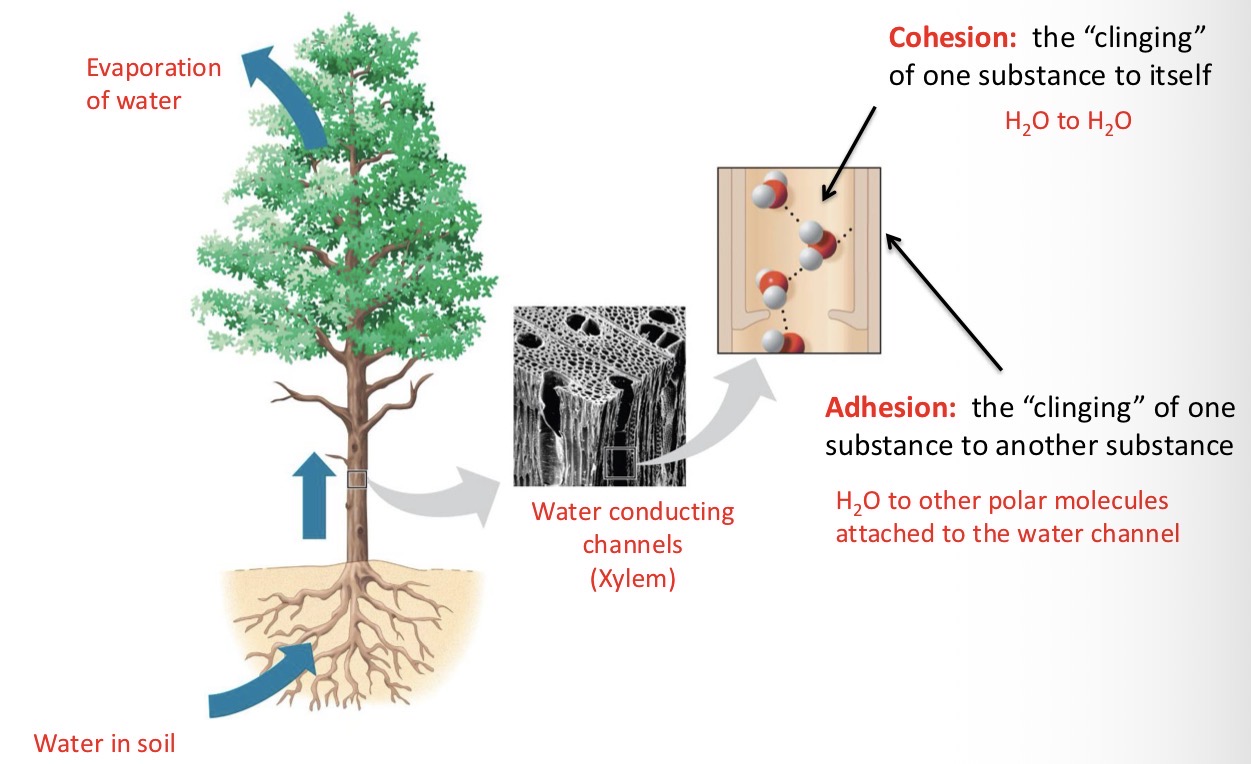
Cohesion
The “clinging” of one substance to itself.
Ex: H2O to H2O
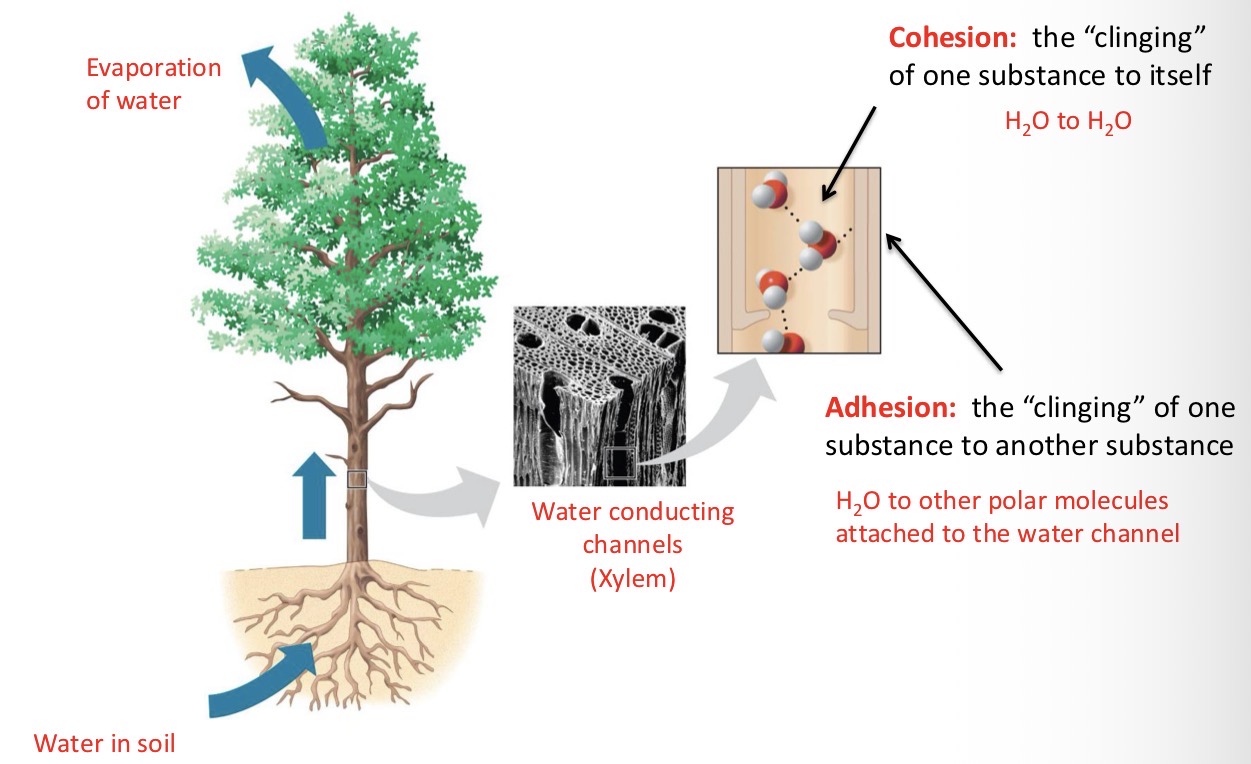
Adhesion
The “clinging” of one substance to another substance.
Ex: H2O to other polar molecules attached to the water channel
Explain waters ability to moderate temperature
water is a “heat bank”
Absorbs heat from air that is warmer
Releases heat to air that is cooler
Water absorbs and releases large amounts of heat energy with only slight changes in temperature
Anything that moves has potential or kinetic energy?
Kinetic energy: the energy of motion
Fill in the blank: Atoms and molecules have kinetic energy. The faster a molecules moves, the greater its _______ energy.
Kinetic
Temperature
the measure of energy that represents the average kinetic energy of molecules
Does not depend on matter’s volume
Measure directly with a thermometer
Units: °C, °F, K
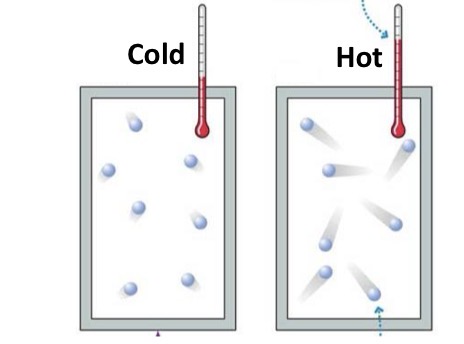
Heat
The total amount of kinetic energy due to random motion of atoms or molecules in a body of matter
Depends on matter’s volume
Cannot be measured with a device directly
Units: calorie= amount of energy of heat it takes to raise the temperature of 1g water 1°C
What is the difference between temperature and heat in terms of kinetic energy, volume, how it’s measured and units?
Temperature is the average kinetic energy of molecules. Heat is the total amount of kinetic energy.
Temperature does not depend on matter’s volume. Heat does depend on matter’s volume.
Temperature can be measured w/ a thermometer directly. Heat cannot be measured with a device directly.
Temperature units include: °C, °F, K. Heat units include calorie.
Water’s ability to moderate temperature stems from its high specific heat. Define specific heat:
Amount of heat absorbed or lost when 1 g of a substance changes its temperature by 1°C. Aka heat capacity.
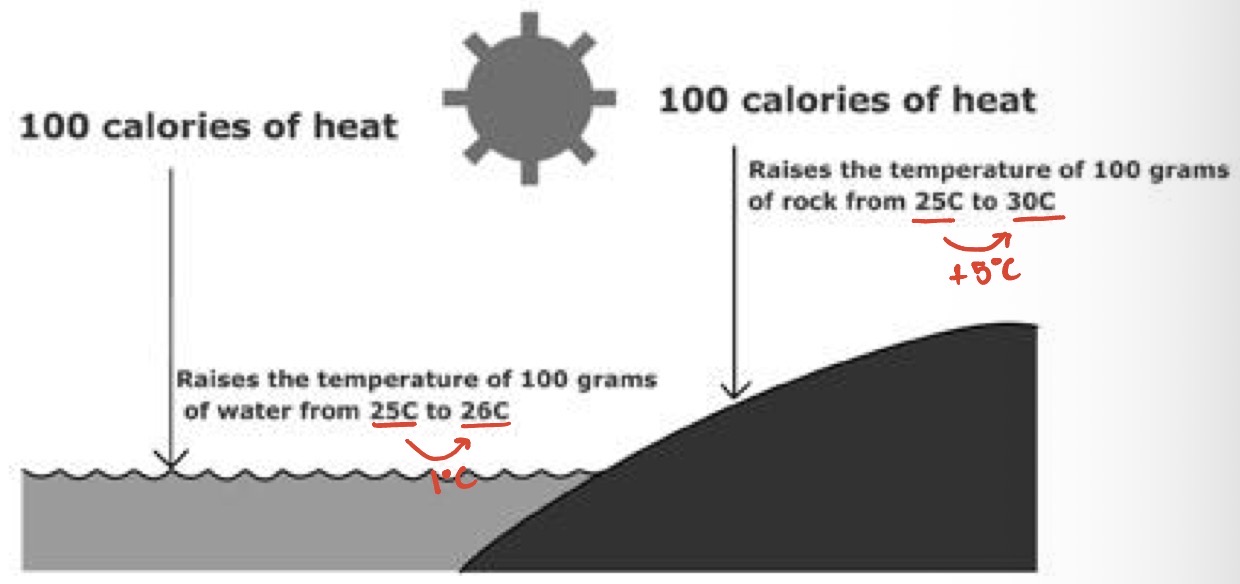
What should be done to increase the temperature of water?
the average kinetic energy of water must increase
Heat absorbed must be great enough to break the hydrogen bonds and increase the molecules kinetic energy
A calorie of heat cases a relatively small change in temperature. Why?
Because much of the heat is used to break hydrogen bonds before the molecules can move faster.
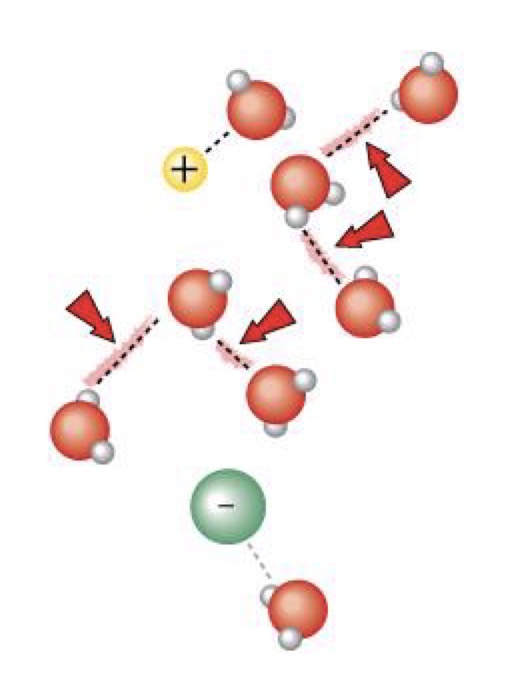
What is responsible for water’s high specific heat?
Hydrogen bonding
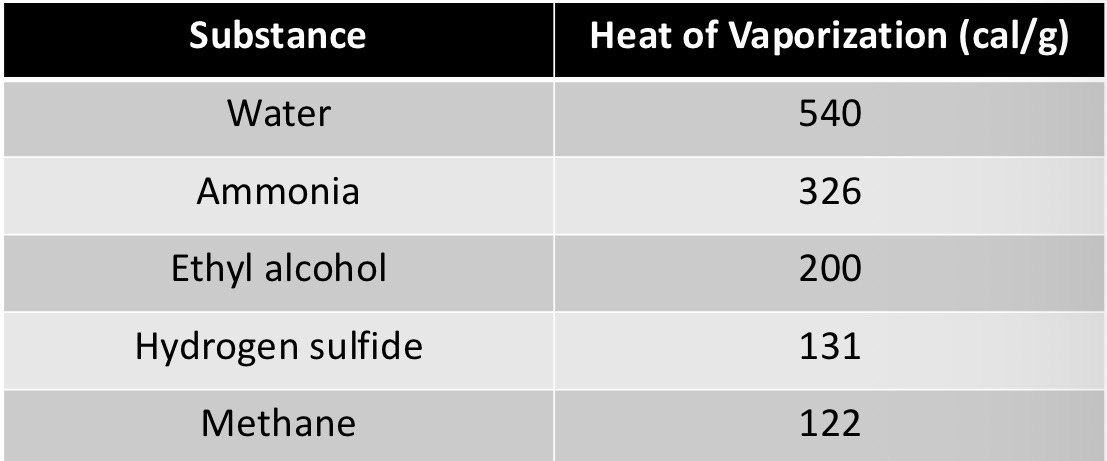
Water has a high heat of vaporization. Define heat of vaporization:
Amount of heat needed to convert 1 g of liquid to gaseous state
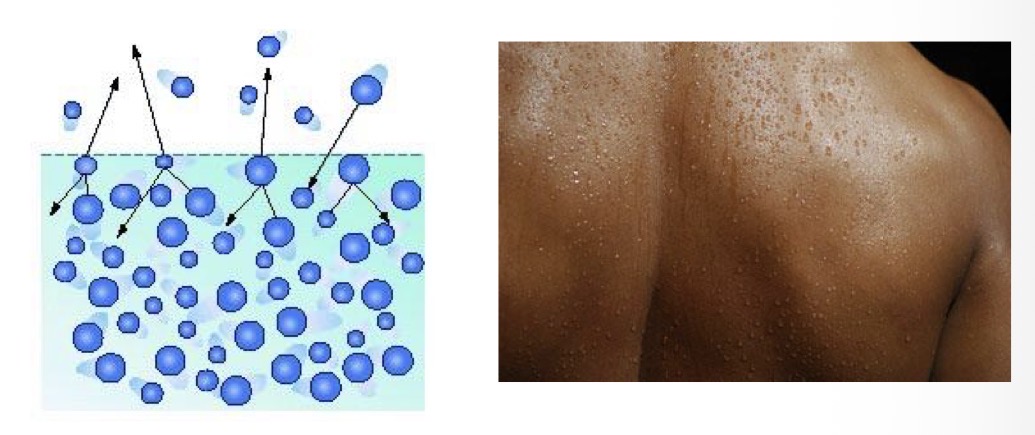
Water’s high heat of vaporization leads to what?
Evaporative cooling. “Hottest” molecules (most kinetic energy) are most likely to leave as gas, leaving behind “colder” molecules.
Explain 1 of the 4 emergent properties of water: Expansion upon freezing
Crystal ice:
each molecule of water is bound to 4 neighboring water molecules in a fixed structure
Liquid Water:
water molecules are closer together. Hydrogen bonds are unstable - will form and reform rapidly. Changes the arrangement of molecules from one instant to another.
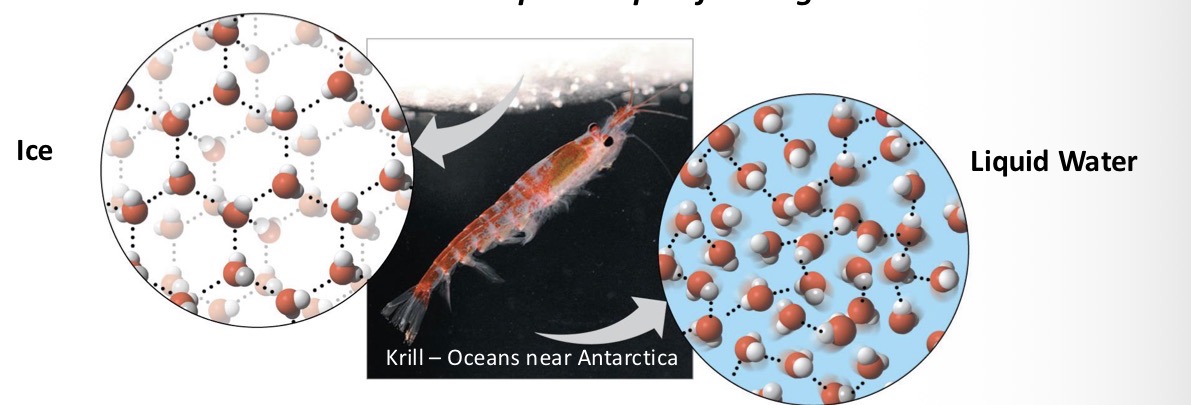
Which is greater in density ice or water?
Ice, because it is 10% less dense than liquid water at 4°C. That is the reason ice floats on water.
Why is it important that ice floats?
It allows life to exist under frozen surfaces
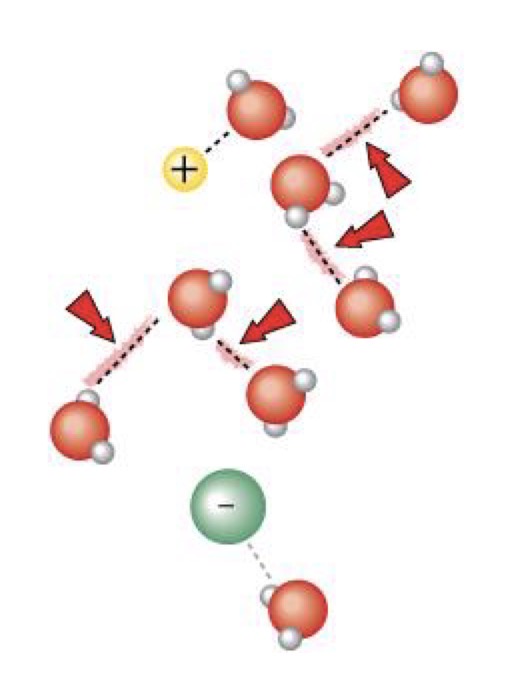
What makes water the solvent of life?
The polarity of water makes it versatile because it forms hydrogen bonds easily with other polar molecules and ionic compounds.
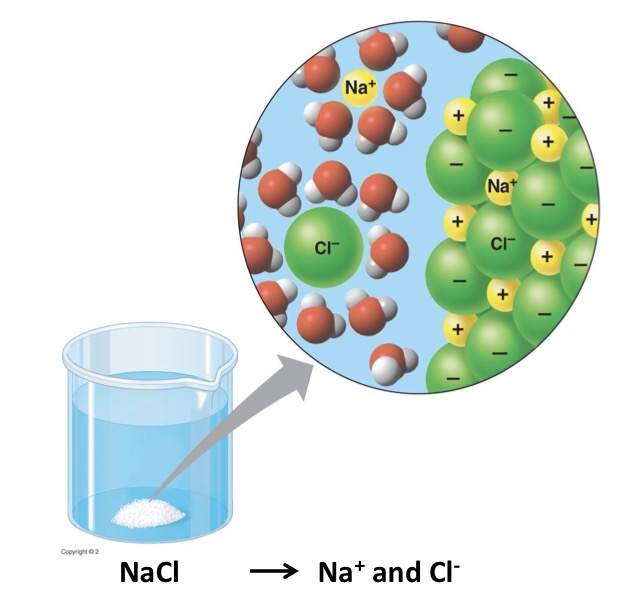
What is a hydration shell?
When water molecules surround solutes they dissolve in water. The hydration shell keeps the particles separated or dispersed in the water.

What is a hydrophilic molecule?
One that can interact readily with the partial charges on water. They are molecules that contain mostly polar covalent or ionic bonds.

What is a hydrophobic molecule?
One that cannot interact readily with the partial charges on water. They are molecules that contain mostly nonpolar covalent bonds.
Acid
Substances that increase the H+ concentration of a solution
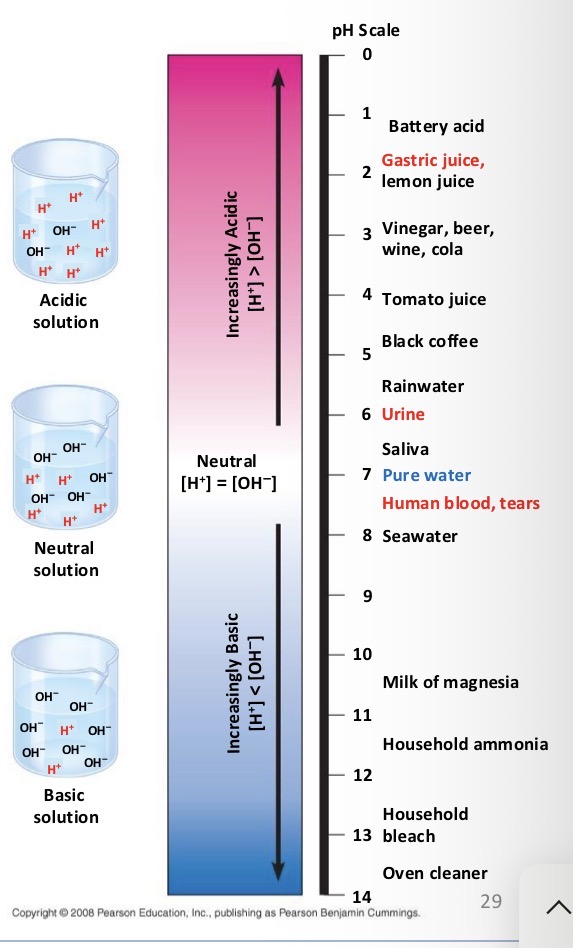
Base
Substance that reduce the H+ concentration of a solution
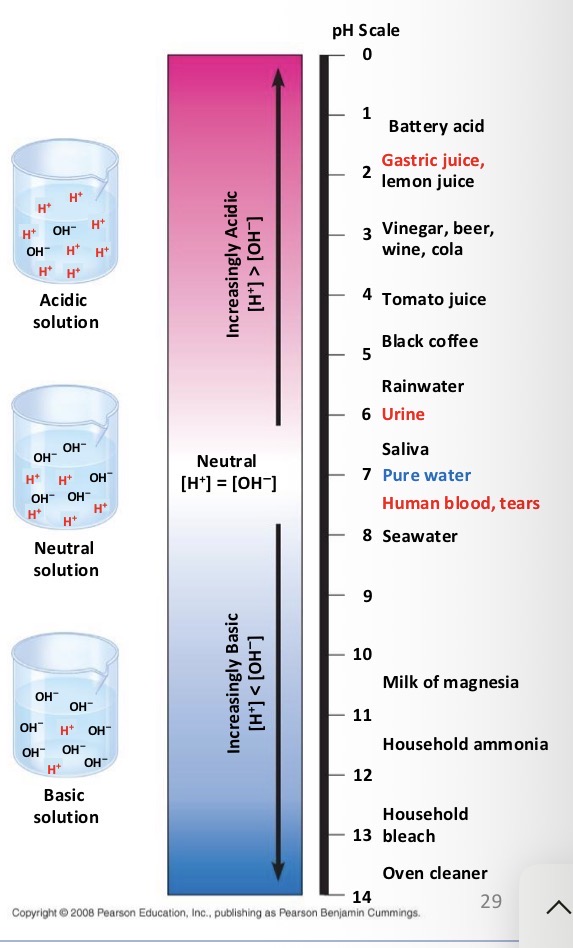
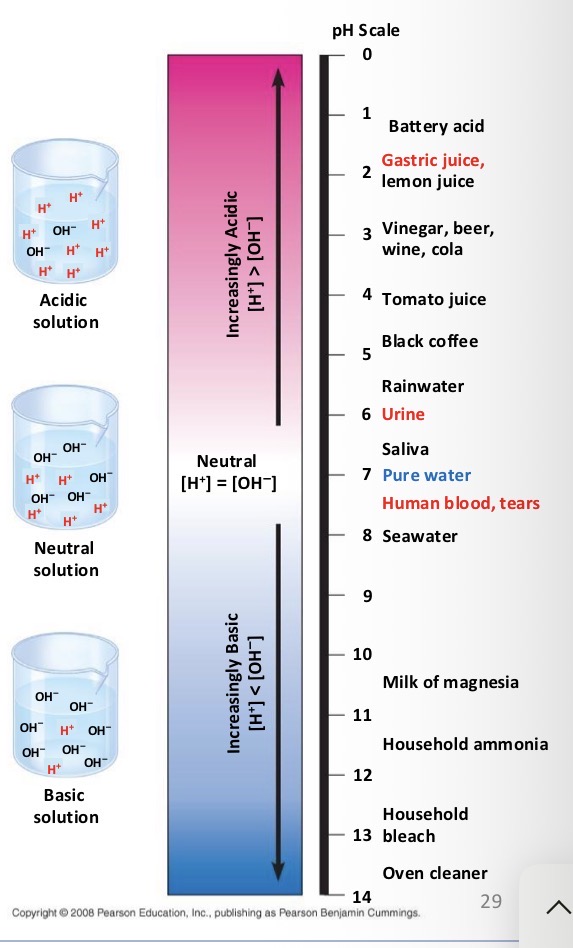
What do biologist use the pH scale for?
To describe whether a solution is acidic or basic
If you have a pH of 3 do you have more H+ or OH-?
H+
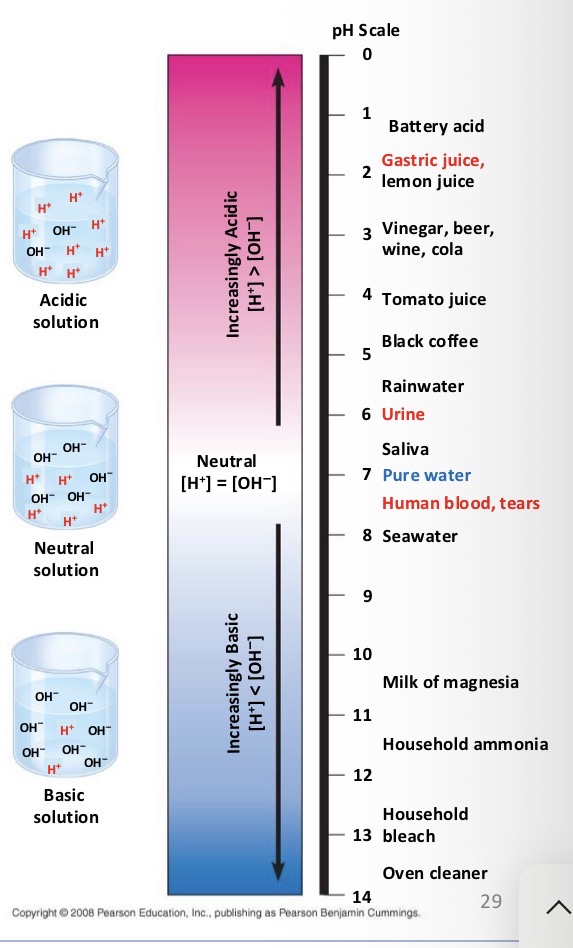
True or False: Acidity is a suitable environment for proteins.
False. Acidity functions to denature proteins and kill microorganisms
True or False: A slight change to the pH of an organism can have drastic effects on an organism.
True
Buffers
Substances that keep in the pH of biological fluids relatively constant
What does a buffer contain?
A weak acid and its corresponding weak base, which combine reversible with H+
Acidosis
pH of blood is less than 7.35
Reaction will proceed to the left to maintain pH homeostasis. More H2CO3 forms and [H+] decreases, pH increases
![<ul><li><p>pH of blood is less than 7.35</p></li><li><p>Reaction will proceed to the left to maintain pH homeostasis. More H2CO3 forms and [H+] decreases, pH increases</p></li></ul><p></p>](https://assets.knowt.com/user-attachments/eb683c13-7b84-4d11-b0e1-10d4e74156f3.jpg)
Alkalosis
pH of blood is greater than 7.45
Reaction will proceed to right to maintain pH homeostasis. More H2CO3 dissociates and [H+] increases, pH decreases.
![<ul><li><p>pH of blood is greater than 7.45</p></li><li><p>Reaction will proceed to right to maintain pH homeostasis. More H2CO3 dissociates and [H+] increases, pH decreases. </p></li></ul><p></p>](https://assets.knowt.com/user-attachments/b6a772a8-bb93-4ff7-9954-0ed09d685ba1.jpg)