B1.1 Carbohydrates and Lipids
1/27
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
28 Terms
Explain the versatility of organic molecules
Carbon is unparalleled in its ability to form large, complex, and diverse molecules
Carbon atoms can form diverse molecules by bonding to four other atoms
With four valence electrons, carbon can form four covalent bonds with a variety of atoms
This tetravalence makes large, complex molecules possible
In molecules with multiple carbons, each carbon bonded to four other atoms has a tetrahedral shape
carbon chains vary with branched or unbranched chains and single or multiple rings.
What are isomers
two or more compounds with the same formula but a different arrangement of atoms in the molecule, and different corresponding properties.
What are carbohydrates
Carbohydrates are a group of organic compounds, made up of the elements and carbon, hydrogen and oxygen.
Formula of carbohydrates
Carbohydrates have the chemical formula CnH2nOn (the ratio of H to O atoms is close to 2:1).
What are monosaccharides, examples, and their chemical formulas
Single sugar molecules
Quite soluble and sweet to taste
Examples
Glucose, galactose and fructose
These three sugars are hexoses - they have six carbon atoms
Isomers of C6H12O6
Ribose and deoxyribose (in nucleic acid)
These sugars are pentoses with five carbon atoms
C5H10O5 and C5H10O4
What are disaccharides, examples, and their monomers
Contain two monosaccharides joined by condensation reaction
Soluble and sweet to taste
Examples
Sucrose is composed of glucose and fructose
Lactose is composed of galactose and glucose
Maltose is composed of two glucose molecules
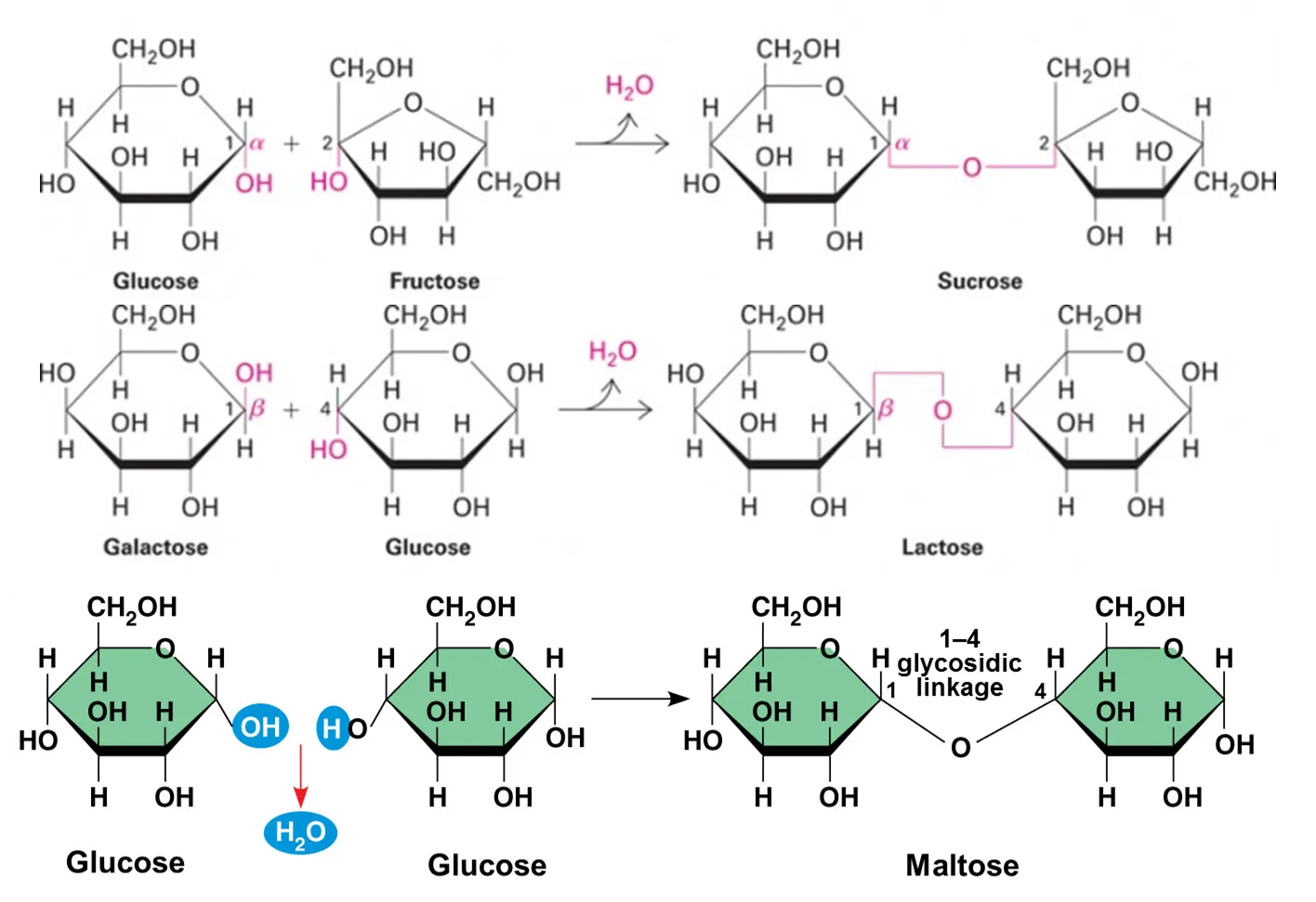
solubility, taste, and general function of polysaccharides
Low solubility; not sweet to taste
Polysaccharides are used as energy storage molecules
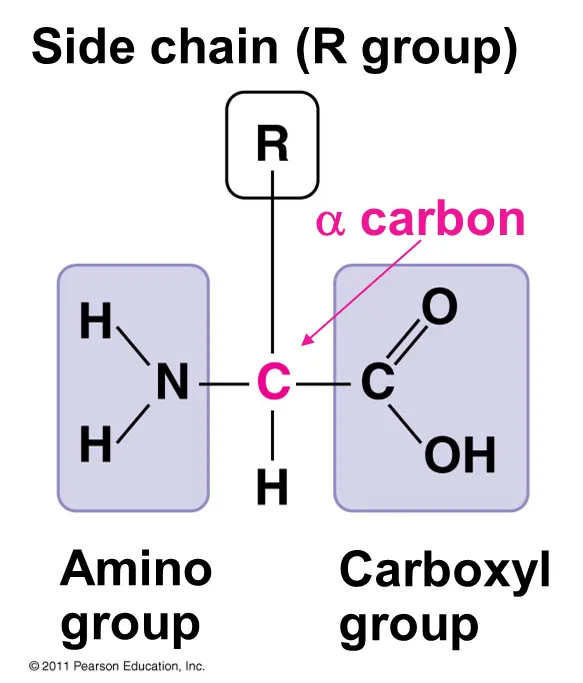
What is the structure of amino acids
Each amino acid has a central carbon atom (the alpha carbon) to which are attached
A hydrogen atom
An amine group –NH2
A carboxylic acid group –COOH
And one of 20 different types of –R (remainder) groups
There are 20 different standard amino acids that make up proteins (There are some rare non-standard amino acids in various organisms.)
Amino acids differ in their properties according to their particular R group (side chains)
What are nucleic acids
Consists of nucleic acid monomers
DNA is a polymer of nucleic acids
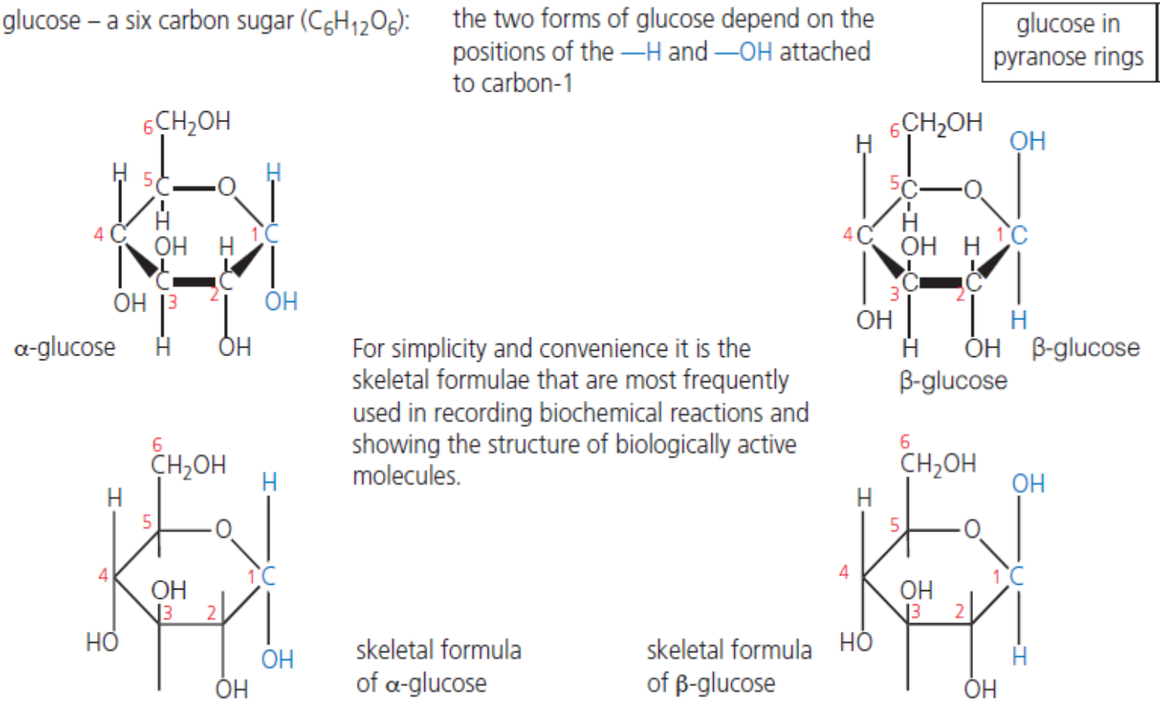
What is the structure of glucose and its properties
It is a hexose ring comprising of 6 carbon atoms (note that 1 of the ring position is occupied by O atom), giving the chemical formula C6H12O6
Molecular stability
Bonds within glucose are stable covalent bonds that do not break easily
Soluble in water
Glucose is polar and so dissolves readily in polar solvent
Easily transportable
Glucose can easily circulate in blood and in fluids between cells due to its solubility
Yields a great deal of chemical energy when covalent bonds are broken
Thus, its high energy yield makes it a good energy store
Sweet
Reducing sugar
Monomer of starch/ glycogen/cellulose
Procedure for test for reducing sugars
To test for the presence of monosaccharides and reducing disaccharide sugars in food, the food sample is dissolved in water, and a small amount of Benedict's reagent is added.
During a water bath, which is usually 4–10 minutes, the solution should progress in the colours of blue (with no reducing sugar present), green, orange, yellow, red, and then brick red precipitate or brown (with high reducing sugar present).
A colour change would signify the presence of a reducing sugar. The common disaccharides lactose and maltose are directly detected by Benedict's reagent because each contains a glucose with a free reducing aldehyde moiety, after isomerization.
Why is sucrose not a reducing sugar?
Sucrose (table sugar) contains two sugars (fructose and glucose) joined by their glycosidic bond in such a way that there is no free aldehyde or ketone group to be oxidised.
Sucrose is thus a non-reducing sugar which does not react with Benedict's reagent.
Sucrose indirectly produces a positive result with Benedict's reagent if heated with dilute hydrochloric acid prior to the test, although after this treatment it is no longer sucrose.
The acidic conditions and heat break the glycosidic bond in sucrose through hydrolysis.
The products of sucrose decomposition are glucose and fructose, both of which can be detected by Benedict's reagent, as described above.
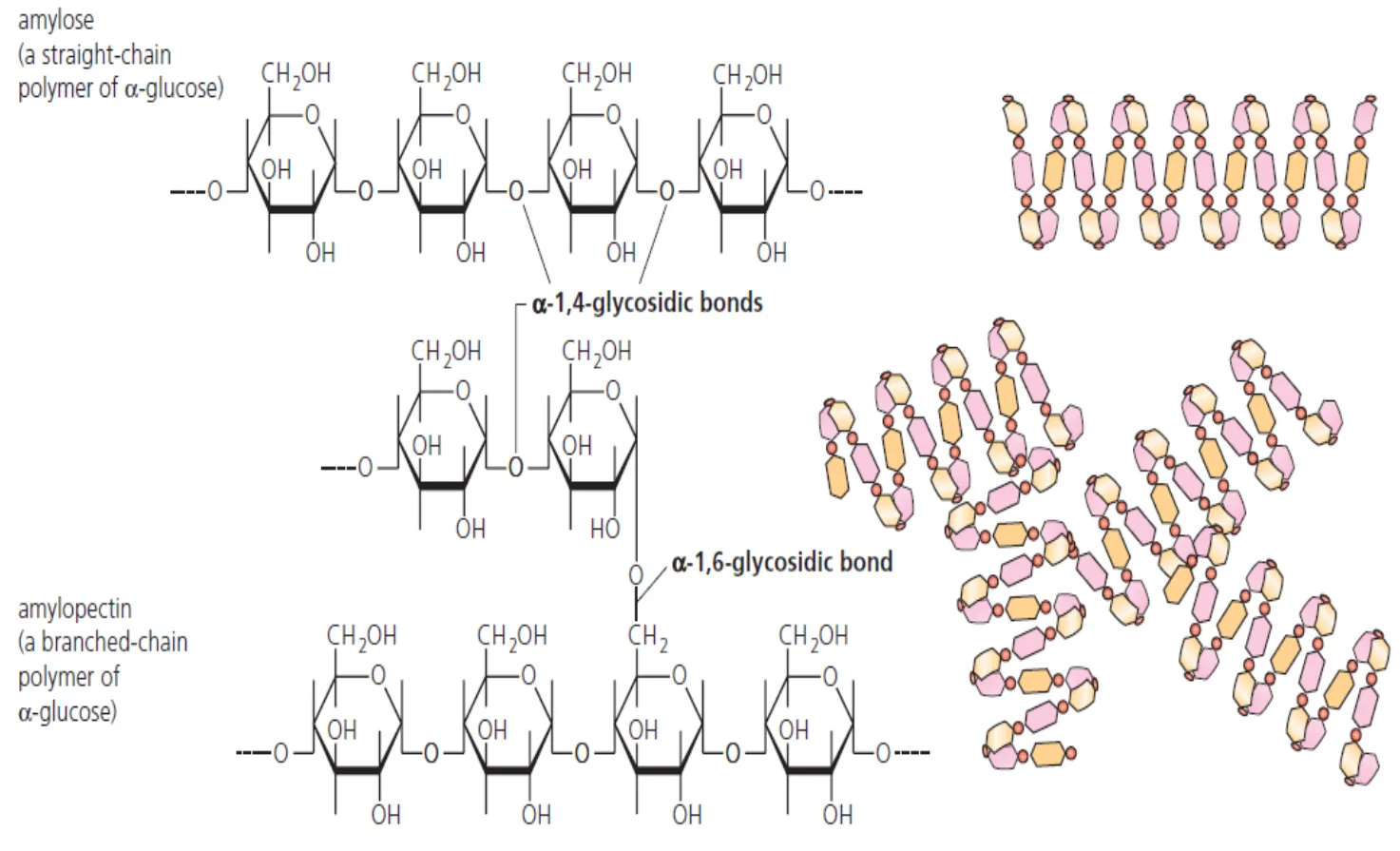
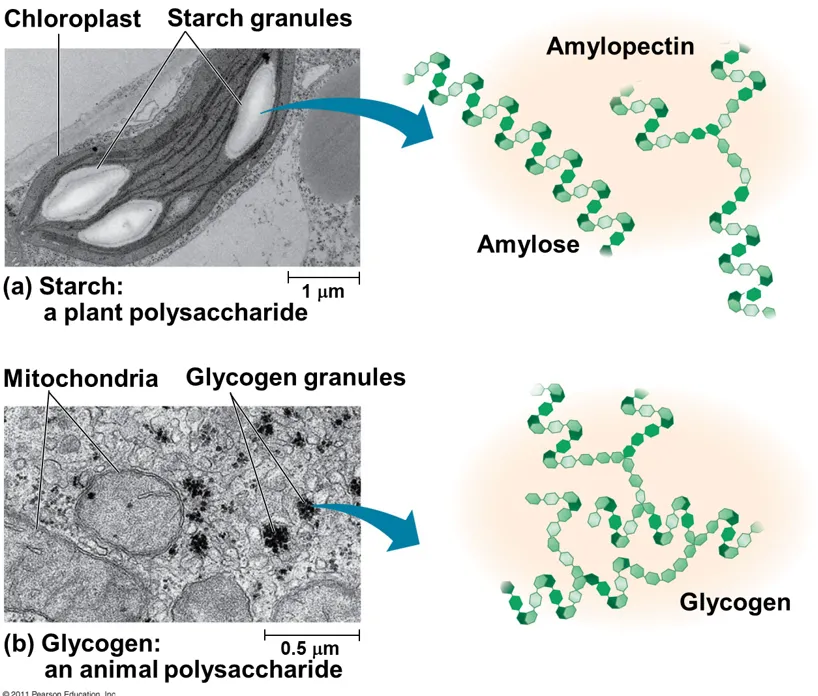
What is the structure of starch and its function
Polymer of alpha glucose with few side branches
Starch is a mixture of amylose and amylopectin.
Amylose is an unbranched chain of several thousand 1,4 linked α-glucose units.
Amylopectin has shorter chains of 1,4 linked α-glucose units but, in addition, there are branch points of 1,6 links along its chains.
In starch, the bonds between glucose residues bring the molecules together as a helix.
The whole starch molecule is stabilized by countless hydrogen bonds between parts of the component glucose molecules
Used for short-term energy storage, often found in plants as it does not draw water.
What is the structure of glycogen and its function
how does the structure of glycogen contribute to its function
A polymer of α-glucose, chemically very similar to amylopectin, although larger and more highly branched.
Glucose is stored as glycogen in liver and muscles
Glycogen is branched in order to increase the release of glucose from glycogen as there is a higher surface area for enzymes to react
What is the structure of cellulose and its uses
Cellulose is a polymer of β glucose which forms microfibrils
alternating orientation of beta-glucose monomers, giving straight chains that can be grouped in bundles and cross-linked with hydrogen bonds.
Primary constituent of plant cell walls
Main component of wood and many natural fibers
Why is cellulose indigestible by most animals
Enzymes that digest starch by hydrolyzing a linkages can’t hydrolyze b linkages in cellulose
Cellulose in human food passes through the digestive tract as insoluble fiber
Some microbes use enzymes to digest cellulose
Many herbivores, from cows to termites, have symbiotic relationships with these microbes
What are the function of carbohydrates in humans
Monosaccharides | Function |
Glucose | Transported to cells in blood plasma |
Galactose | Used in the production of lactose (milk sugar) |
Disaccharides | Function |
Lactose | Produced in mammary glands and secreted into the milk as an important component in the diet of very young mammals |
Polysaccharides | Function |
Glycogen | Storage carbohydrate formed from glucose in the liver and other cells (but not in brain cells) when glucose is not immediately required for cellular respiration |
What is the function of carbohydrates in plants
Monosaccharides | Function |
Glucose | A first product of photosynthesis |
Fructose | Produced in cellular respiration as an intermediate of glucose breakdown |
Disaccharides | Function |
Sucrose | Produced in green leaves from glucose and fructose |
Maltose | Breakdown product in the hydrolysis of starch |
Polysaccharides | Function |
Cellulose | Manufactured in cells and laid down externally, in bundles of fibres, as the main component of the cell walls |
Starch | Storage carbohydrate |
What is the structure of glycoproteins, their function in cell recognition, and an example
Glycoproteins are composed of polypeptides with carbohydrate attached.
In most cases, the carbohydrate is an oligosaccharide — a short chain of monosaccharides linked by glycosidic bonds.
Cellular recognition
Glycoproteins are a component of plasma membranes in animal cells and are positioned with the attached carbohydrate facing outwards.
By displaying distinctive glycoproteins, cells allow other cells to recognize them.
The glycoprotein on the surface of one cell is recognized by receptors on the surface of another cell
Cell-to-cell recognition helps with the organization of tissues and can also allow foreign cells or infected body cells to be identified and destroyed.
e.g. ABO glycoproteins
The ABO antigens in red blood cells are an example of glycoproteins providing the means of cell–cell recognition
Red blood cells have glycoproteins in their membranes that do not have a known function, but that affect blood transfusion.
Lipids and their functions
Type | Function |
|---|---|
Fats | Long-term energy storage and insulation in animals |
Oils | Long-term energy storage in plants and their seeds |
Phospholipids | Component of plasma membranes |
Steriods | Component of plasma membrane (cholesterol), sex hormones |
Waxes | Protection, prevent water loss (cuticle of plant surfaces), beeswax, earwax |
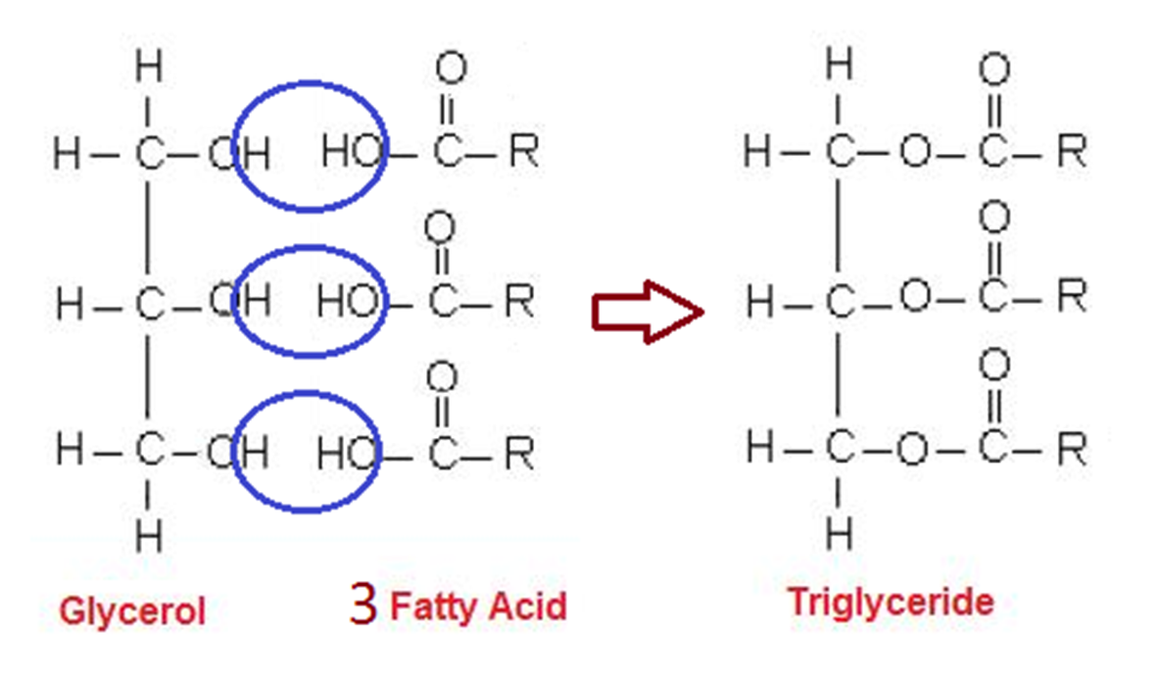
What are triglycerides and their structure
One of the principal groups of lipids
Consists of glycerol bonded to 3 fatty acids through ester bonds (formed by condensation reaction)
Saturation of fatty acids
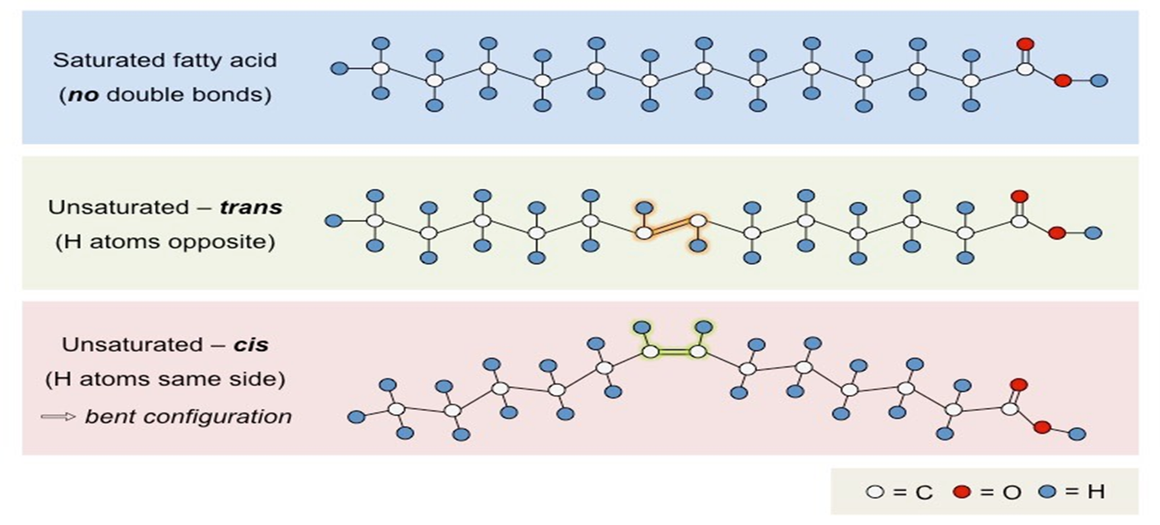
Saturated - no double bonds between carbons e.g. in fats (butter)
Unsaturated - 1 (monounsaturated) or more than1 (polyunsaturated) double bonds between carbons e.g. in oils
Depending on where the hydrogen atoms are on each side of the double bond, unsaturated fatty acids can either be cis or trans isomers.
Difference between length of fatty acid tails in oil and fats
Oils with unsaturated fatty acids have bends in the hydrocarbon tails which reduces the density of the molecule and lower the melting point. Oils tend to have short fatty acid tails.
Conversely fats tend to have longer fatty acid tails with saturated bonds which makes their structure densely packed and increases the melting point.
Most plant fatty acids are unsaturated and most animal fatty acids are saturated.
What is adipose tissue
Triglycerides are used for energy storage in plants and animals.
In animals, the triglycerides are fats and are stored in specialized groups of cells called adipose tissue.
In humans, adipose tissue is located immediately beneath the skin and also around some organs including the kidneys.
Properties of triglycerides that make them suitable for energy storage and thermal insulation
Chemically very stable, so energy is not lost over time.
Immiscible with water, so they naturally form droplets in the cytoplasm which do not have osmotic or other effects on the cell.
Release twice as much energy per gram in cell respiration as carbohydrate, so enough energy can be stored in half the body mass. This is important for animals that move and especially for birds and bats that fly.
Poor conductors of heat, so they can be used as a thermal insulator in animals that need to conserve body heat.
Advantages of lipids vs carbohydrates
Advantage | Lipids | Carbohydrates |
|---|---|---|
Energy released | Lipids contain more energy per gram than carbohydrates. Therefore lipid is a lighter store than carbohydrates for the same amount of energy. | Carbohydrates are more easily digested than lipids so the energy stored by them can be released more rapidly. |
Solubility | Lipids are insoluble in water and therefore do not affect the osmotic potential in the cell. | Carbohydrates are soluble in water and therefore easily move to and from the store. |
Duration | Long term energy storage | Short term energy storage |
Respiration | Used only in aerobic respiration | Used both in anaerobic and aerobic respiration |
Why does the phospholipid bilayer form
Most cells have watery environment on both sides of membrane
Water attracts the polar phosphate ends of the phospholipids
Phospholipids align to form double layer membrane, with polar ends on outside of each layer of the membrane
Non-polar tails are inside the bilayer
What are fats, oils, waxes, steriods, and phospholipids
fats are lipids which are in solid state at 20°C.
Oils are lipids which are liquid state at 20°C.
waxes are ong-chain fatty acid bonded to a long-chain alcohol
High melting point
Waterproof
Resistant to degradation
steriods are skeletons of four fused carbon rings
Non-polar steroids (meaning they are fat soluble) can pass through the phospholipid bilayer
E.g. cholesterol, testosterone, estrogen
phospholipids consist of a glycerol backbone bonded to two fatty acids and one phosphate group
Phospholipids are amphipathic and have hydrophilic and hydrophobic properties.
Phosphate end of the phospholipid molecule is a hydrophilic polar molecule.
The other end of the molecule made up of two fatty acids tails is hydrophobic and non-polar.